Stem Cell Portfolio
Explore a curated range of human stem cell models designed for regenerative medicine, tissue engineering, and advanced biological research.

Core Applications
- Regenerative medicine
- Tissue engineering
- Vascular and metabolic studies
- Dental and craniofacial research
- Neonatal and developmental biology
Quality Control
Stem cells are sourced from consented donors and undergo rigorous testing to ensure viability, sterility, and research suitability.
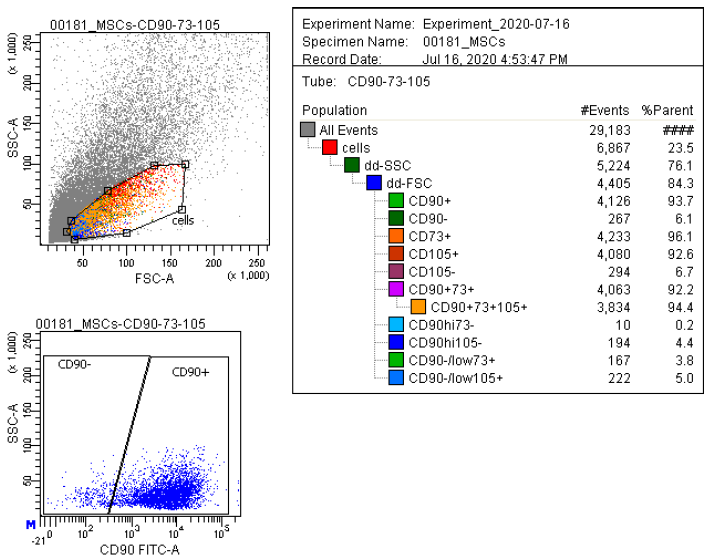
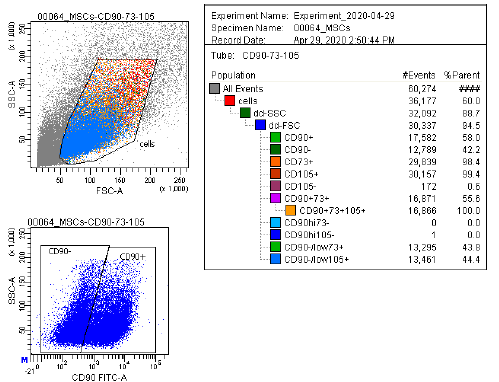
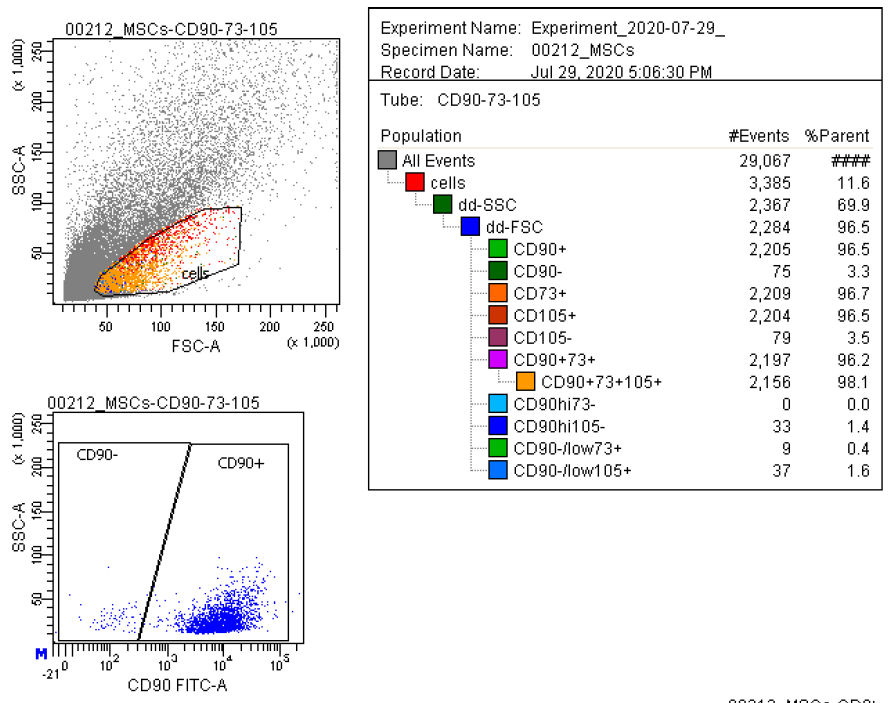
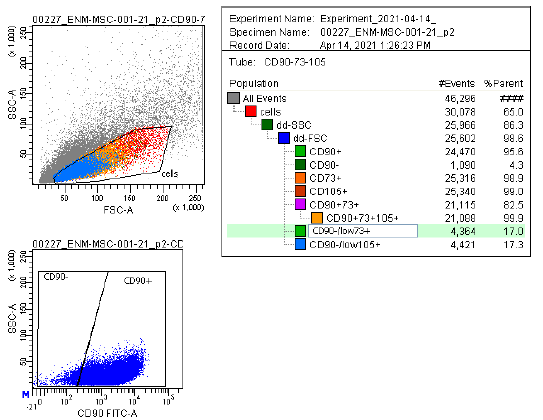
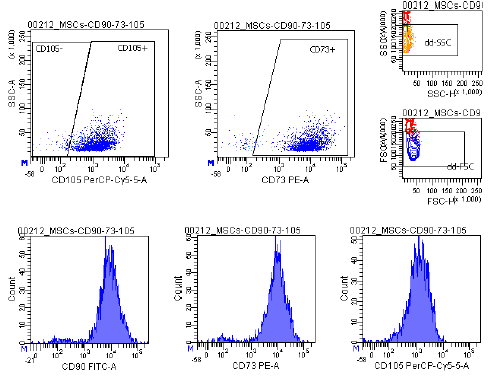
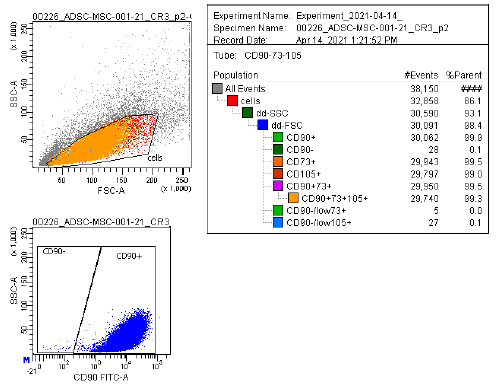
Marker Profile
Characterized using established marker panels in accordance with ISCT guidelines to confirm identity and purity.
Safety and Purity
Each batch is screened for major human pathogens and contaminants, ensuring reliable and safe research use.
| Organism | Human |
|---|---|
| Tissue | Umbilical Cord - Artery |
| Organism | Human |
|---|---|
| Tissue | Umbilical Cord - Whartons Jelly |