Vero Cells: Pioneering Paths in Viral Studies and Vaccine Innovation
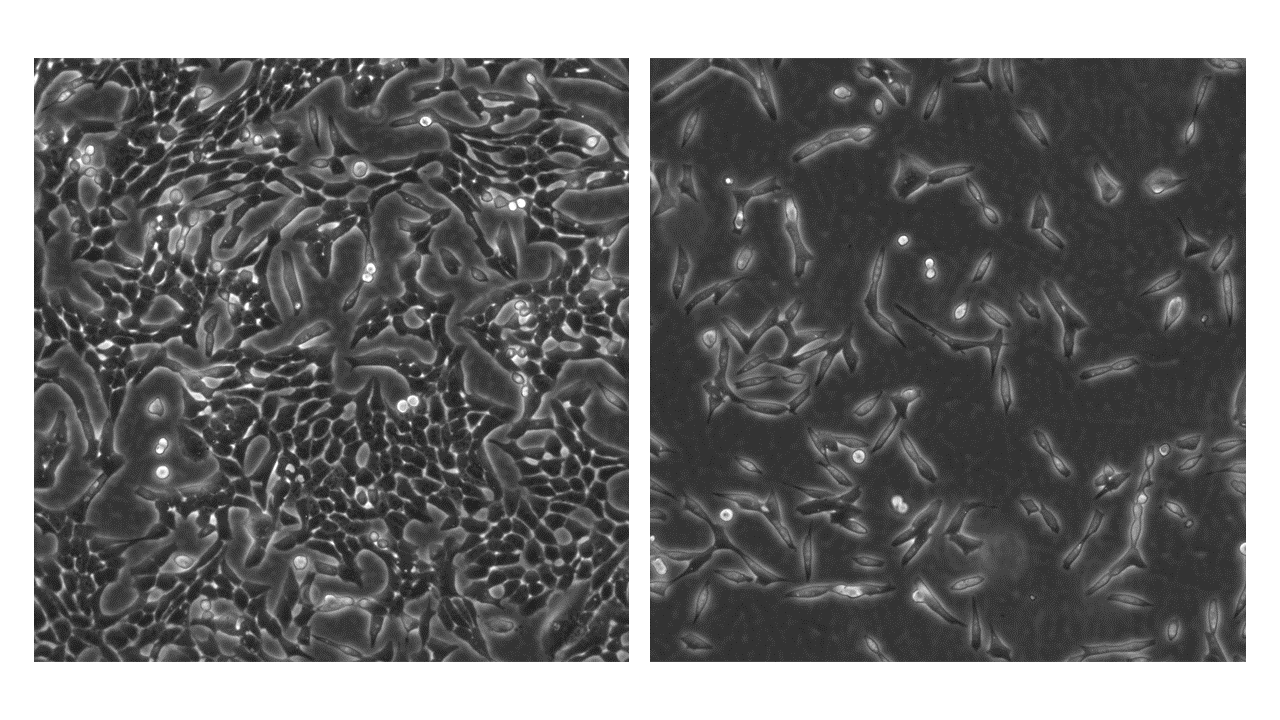
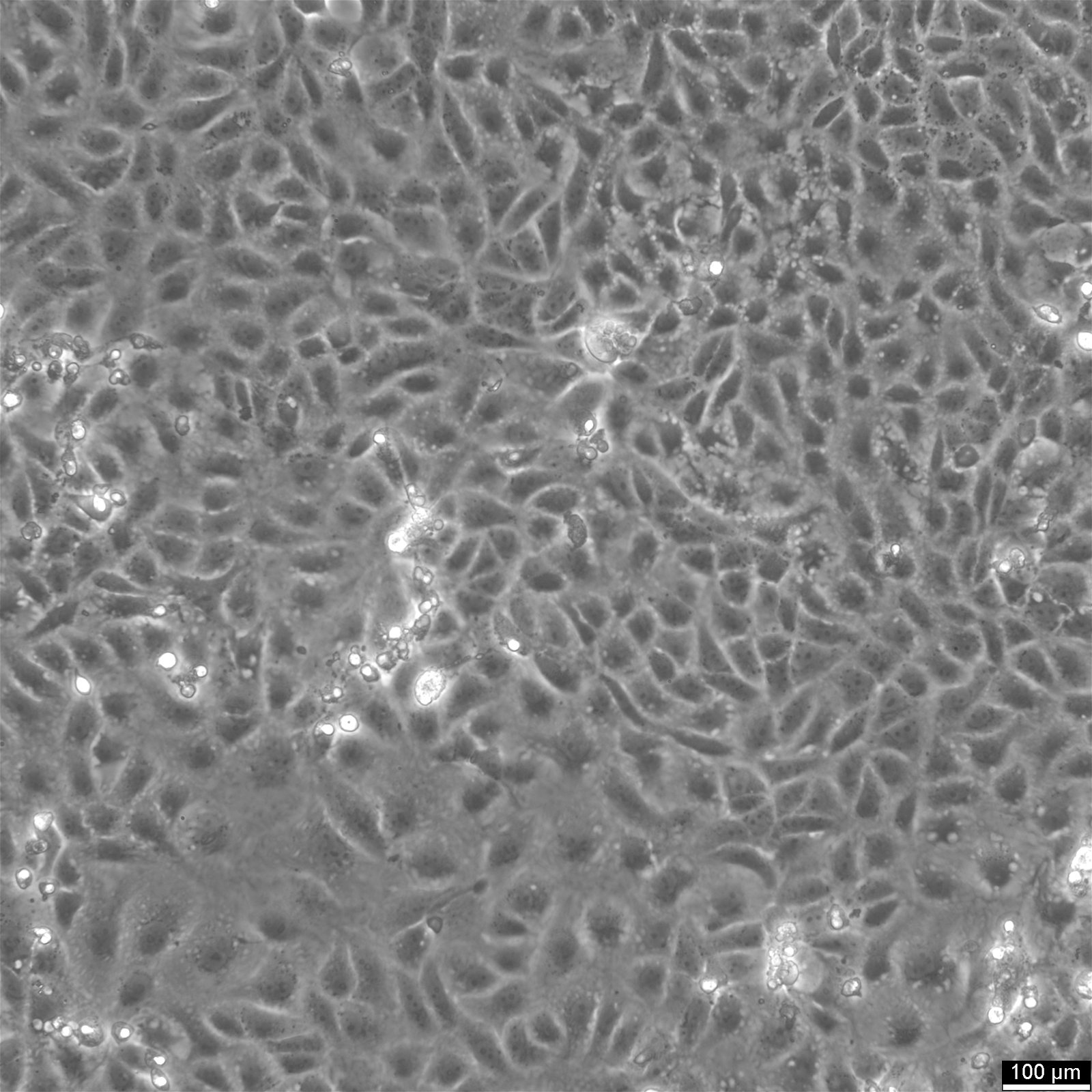
The Vero cell line, an immortalized mammalian cell model derived from the kidney epithelial cells of the African green monkey, stands at the forefront of research in virology, microbiology, and cellular and molecular biology. Their widespread application spans vaccine development, pharmaceutical screenings, and the exploration of virus and parasite biology, tumor immunology, and immunotherapeutic strategies.
- Growth Medium
- See product page
- Doubling Time
- See product page
- Growth Type
- Adherent
- Biosafety Level
- BSL-1
- Available from
- Cytion — Order Vero
- Origin and Key Features of Vero Cells
- Culturing information
- Applications of Vero cell line in research
- Explore our Vero cells and derivatives for groundbreaking research applications
- Research publications
- Resources for Vero cells: Protocols, videos, and more
- FAQs for Vero cells
- References
- Frequently asked questions
Origin and Key Features of Vero Cells
Delving into a cell line such as Vero prompts several inquiries: What exactly are Vero cells? How was the Vero cell line established? What's the story behind the name "Vero"? This segment aims to elucidate the genesis and primary attributes of Vero cells.
The inception of the Vero cell line dates back to 1962, originating from the kidney epithelial cells of the African green monkey. This line was cultivated by Y. Kawakita and Yasumura at Chiba University in Japan. The term "Vero" is derived from "Verda reno" in Esperanto, translating to "green kidney," though "Vero" also resonates with the notion of "truth."
Vero cells typically form monolayers but can adapt to suspension cultures, displaying an epithelial-like structure. These cells are characterized by their rounded to elongated shape and an average diameter of about 17 µm. Notably, Vero cells exhibit a hypodiploid chromosome count, with the modal chromosome number being 58 in roughly 66% of the cell population, though variations with higher ploidies exist in a small fraction (1.7%) of cells.
Vero Cell Clones and Their Unique Properties
Distinct clones derived from the Vero cell lineage exhibit unique features that distinguish them from the original line. Among these, two notable Vero cell clones are:
Vero E6 Cell Line: Also known as Vero C1008, this clone stems from Vero 76 cells and was isolated in 1979 by P.J. Price using a dilution technique on microtiter plates. Vero E6 cells are particularly suited for cultivating viruses that replicate slowly.
Vero 76 Cells: Originating from the kidney of an African green monkey in 1968, these cells maintain the epithelial morphology characteristic of Vero cells.
These variants of Vero cells, alongside the parent line, continue to be instrumental in the advancement of virological research and the development of medical interventions, marking their significance in the scientific community.
Culturing information
Culturing Vero cells, a monkey cell line, requires familiarity with specific parameters such as doubling time, seeding density, and the appropriate growth medium.
Population Doubling Time: The doubling time for Vero cells is around 24 hours.
Adherence: Vero cells adhere to surfaces and typically form monolayers when cultured.
Seeding Density: It is advisable to start with a seeding density of 1 x 10^4 cells/cm^2. To culture adherent Vero cells, wash them with PBS and treat them with Accutase to detach them. After detaching, centrifuge the cells, resuspend them in fresh medium, and transfer them to new culture flasks.
Growth Medium: Both Ham’s F12 and DMEM are suitable media for cultivating Vero cells. These should be supplemented with 2.5 mM L-glutamine and 5% fetal bovine serum (FBS) to support optimal growth. The medium should be refreshed two to three times per week.
Growth Conditions: Vero cells thrive at a temperature of 37°C in a humidified atmosphere supplied with 5% CO2.
Storage: For long-term storage, Vero cells should be kept at temperatures below -150°C, either in an ultra-low temperature freezer or in the vapor phase of liquid nitrogen.
Freezing Process and Medium: For cryopreservation, use CM-1 or CM-ACF, or the growth medium with FBS and DMSO, as the freezing medium. Employ a slow freezing technique, decreasing the temperature gradually by 1°C per minute.
Thawing Process: Thaw Vero cells by immersing the container in a 37°C water bath and gently agitate it for 40-60 seconds. Then, dilute the cells in fresh medium, centrifuge to remove the cryoprotectant, resuspend in fresh growth medium, and place them in a new flask to recover and grow.
Biosafety Level: Vero cells should be handled in a laboratory that meets the requirements for biosafety level 1.
Applications of Vero cell line in research
The Vero cell line has numerous research applications in the cell biology and virology fields. Here, we have discussed some specific ones.
Vero Cells in Viral Research and Vaccine Production
Vero cells, derived from African green monkey kidney cells, have become a staple in the bioprocess development of vaccines for various viruses, including poliovirus and Japanese encephalitis virus. Their adaptability in both adherent and suspension cultures and their broad virus support capability, including for pathogens like the des petits ruminants virus, highlight their significance in virus isolation and vaccine development.
Numerous studies have utilized Vero cells for the production of human vaccines. For instance, a notable study published in 2019 demonstrated the use of Vero cells in the development of an inactivated vaccine for the yellow fever virus [2].
Vero cells are commonly used in viral infection studies, such as a study conducted in 2020 that infected Vero cells with different isolates of the SARS-CoV-2 virus to study the growth properties of the virus [4]. Similarly, another study investigated cell responses to SARS-CoV-2 infection using Vero cell culture [5].
The Role of Vero Cells in Tissue Engineering and Upstream Bioprocess Development
Beyond vaccine production, Vero cells contribute to tissue engineering and the broader field of bioprocess development, underscoring the need for ongoing research into their properties and applications. The selection of appropriate Vero cell sublines is key to maximizing their potential in the biotechnology and pharmaceutical industries.
Application of Vero Cells in Drug Efficacy and Safety Testing
Vero cells are frequently employed in drug testing to assess the effectiveness and safety of pharmaceutical compounds. These cells are often considered a standard model for normal kidney cells in studies investigating the cytotoxic effects of various drugs and therapeutic agents. For example, research comparing the impact of Terminalia avicennioides plant root extracts on both HepG2, a liver cancer cell line, and Vero cells, derived from monkey kidney epithelium, found that the extracts were more detrimental to the cancerous cells than to the normal cells.
Limitations of Vero Cells
While Vero cells are extensively used, they have their limitations, such as vero toxin production and genomic alterations that may impact certain applications. Understanding the specific sublines and genomic features of Vero cells, including the Vero lineage F6, is essential for optimizing their use in various bioprocesses.
Explore our Vero cells and derivatives for groundbreaking research applications
Research publications
The following are some recent and most cited research publications featuring Vero cells.
This study published in the Vaccine Journal (2019) adapted Vero cells to grow in suspension cultures to produce rabies virus at high titer using various serum-free media.
Susceptibility of Toxoplasma gondii to Ethanolic Extract of Tinospora crispa in Vero Cells
This paper was published in the Evidence-Based Complementary and Alternative Medicine journal in 2019. The study proposed that the ethanolic extract of the plant Tinospora crispa exerts a detrimental effect on the Toxoplasma gondii parasite. However, it is safe for the host cells (the Vero cell line).
This article was published by the Journal of the Balkan Union of Oncology in 2021. In this stidy, Ozlem Dagdeviren Ozsoylemez and Gul Ozcan explored the cytotoxic effect of Colchicum baytopiorum leaf extract on C-4I and Vero cell lines.
This study in Phytotherapy Research (2021) used SARS-CoV-2-infected Vero cells to investigate the therapeutic effect of resveratrol on virus replication.
Lipophilic statins inhibit Zika virus production in Vero cells
This paper in Nature Scientific Reports (2019) proposes that lipophilic statins, i.e., cerivastatin, lovastatin, fuvastatin, simvastatin, and mevastatin, can impede the production of Zika virus in Vero cells.
Resources for Vero cells: Protocols, videos, and more
- Transfection of Vero cells: This link will guide you through a transfection protocol for Vero cells.
- Transfecting Vero cell line: This video explains a transfection protocol for the Vero cell line.
- Subculture Vero cells: This video describes general subculturing recommendations for adherent cell lines.
Cell culture protocols
- Culturing Vero cells: This website contains a well-explained protocol for culturing Vero cells.
- Vero cell culture: This document may help you learn the protocol for propagating, maintaining, and freezing Vero cells.
FAQs for Vero cells
Vero cell sublines are specialized strains of the original Vero cell line, each with unique characteristics for different research needs. Examples include:
- Vero 76: Adapted for growth in suspension, used in vaccine production.
- Vero E6: Highly susceptible to the Ebola virus, used in hemorrhagic fever research.
References
- Ammerman, N.C., M. Beier‐Sexton, and A.F. Azad, Growth and maintenance of Vero cell lines. Current protocols in microbiology, 2008. 11(1): p. A. 4E. 1–A. 4E. 7.
- Pato, T.P., et al., Purification of yellow fever virus produced in Vero cells for inactivated vaccine manufacture. Vaccine, 2019. 37(24): p. 3214–3220.
- Aliyu-Amoo, H., et al., Antiproliferative effect of extracts and fractions of the root of Terminalia avicennioides (Combretaceae) Guill and Perr. on HepG2 and Vero cell lines. Clinical Phytoscience, 2021, 7(1): p. 1–7.
- Yao, P., et al., Isolation and growth characteristics of SARS-CoV-2 in Vero cells. Virologica Sinica, 2020. 35(3): p. 348–350.
- Park, B.K., et al., Differential signaling and virus production in Calu-3 cells and Vero cells upon SARS-CoV-2 infection. Biomolecules & Therapeutics, 2021. 29(3): p. 273.