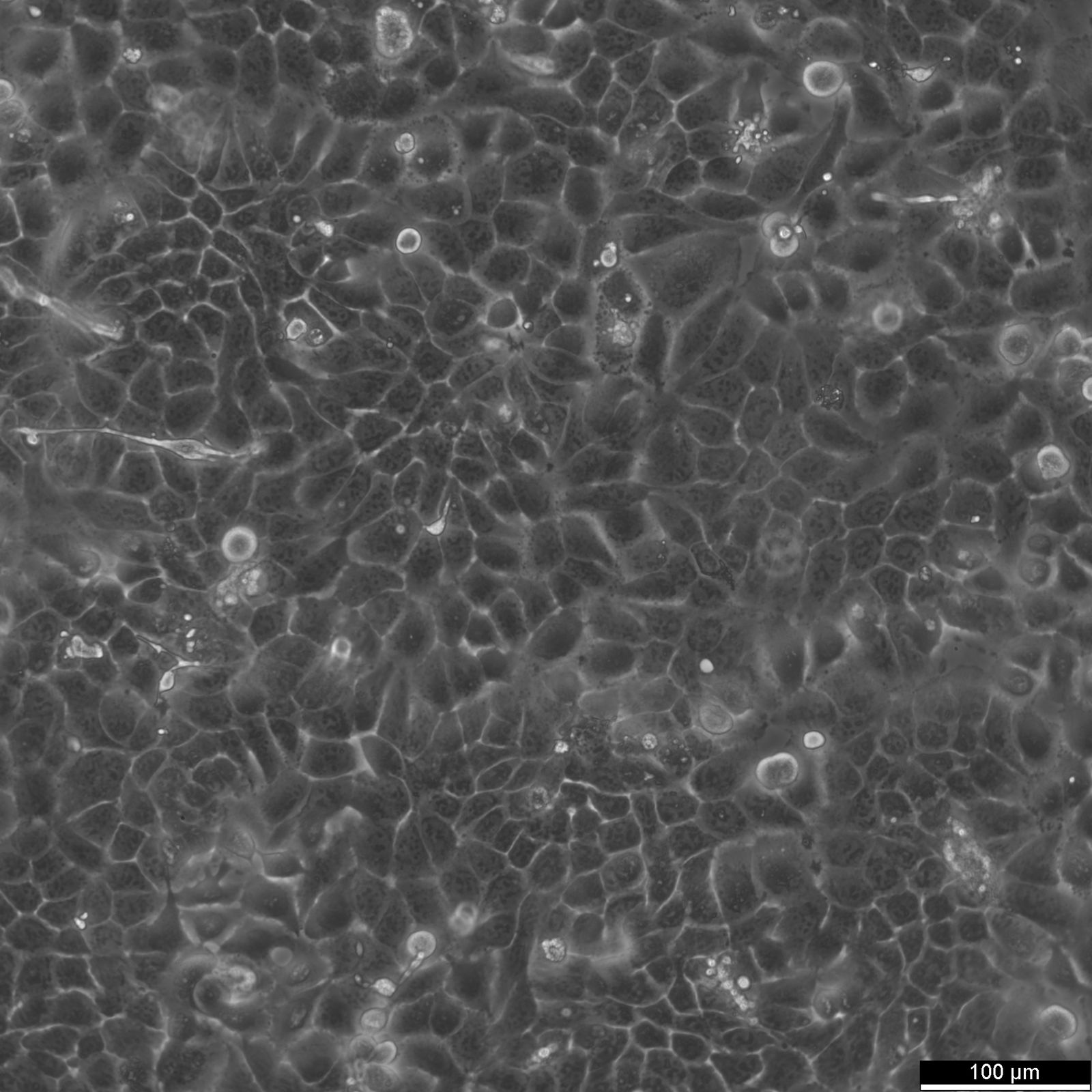
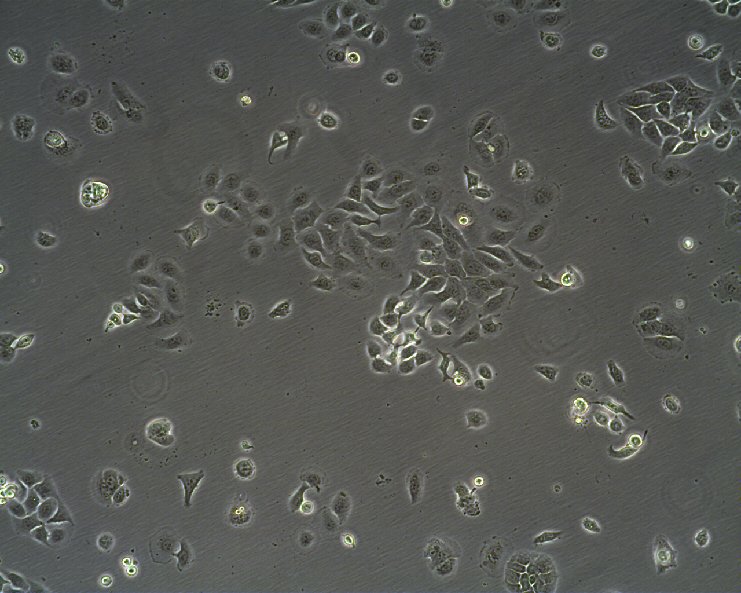
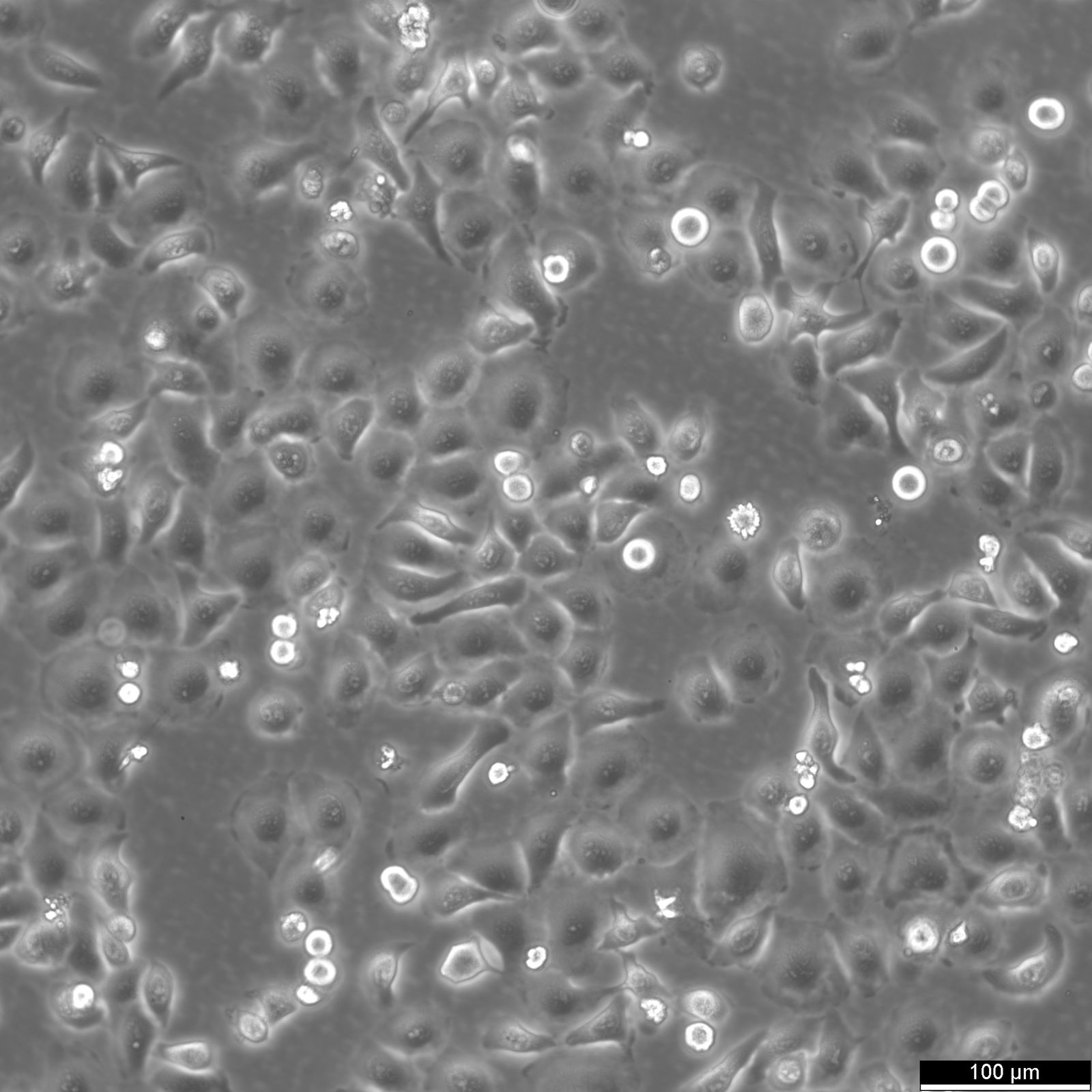
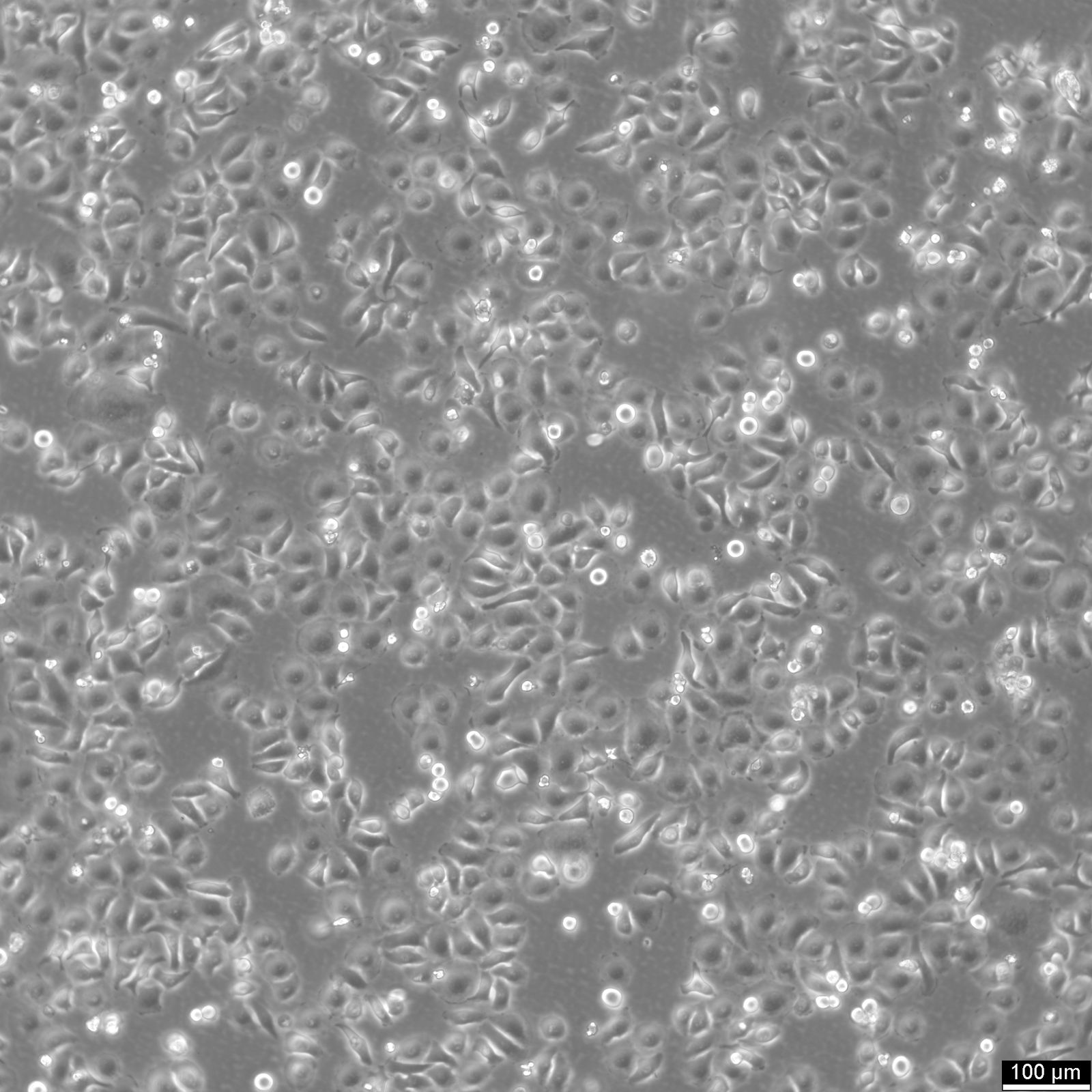
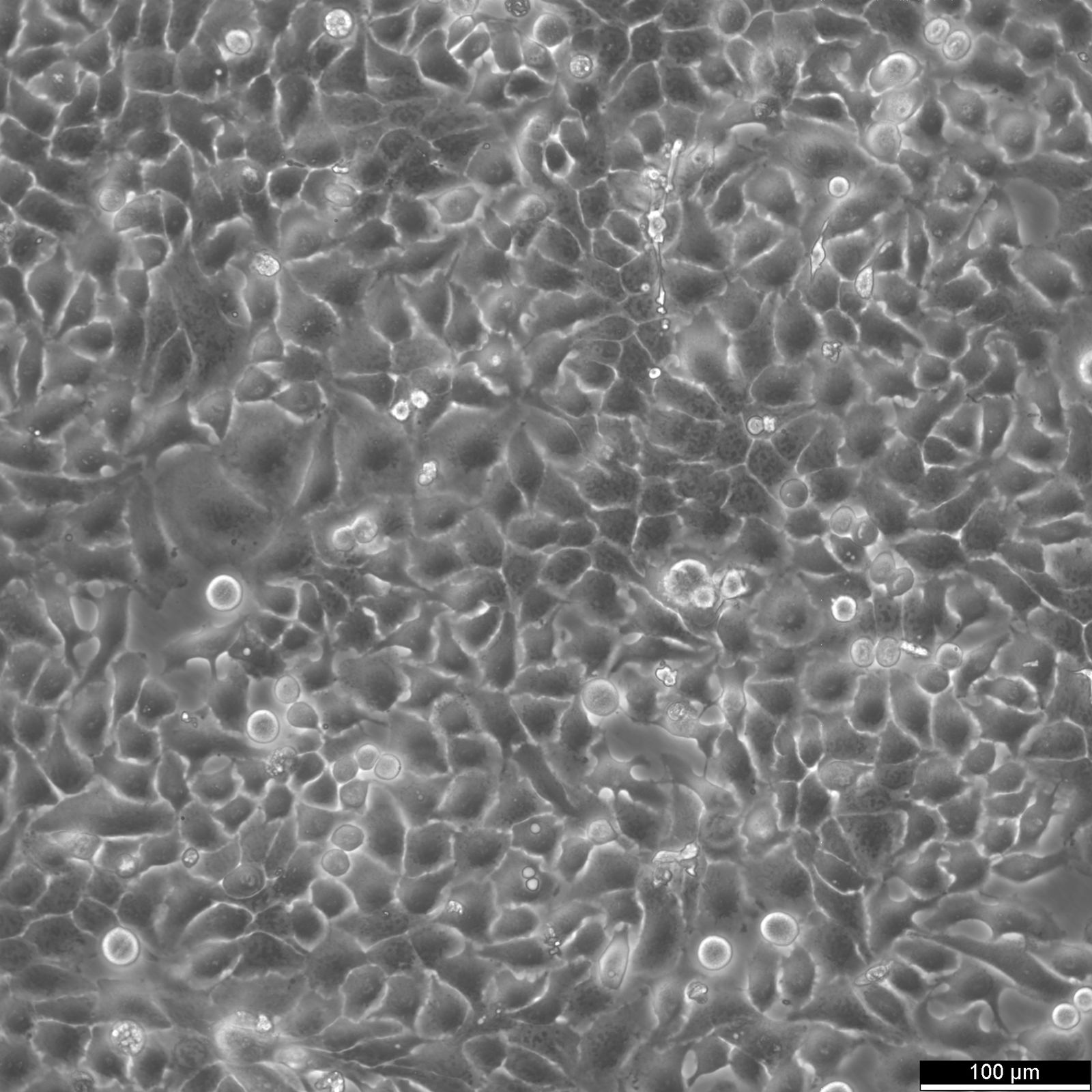
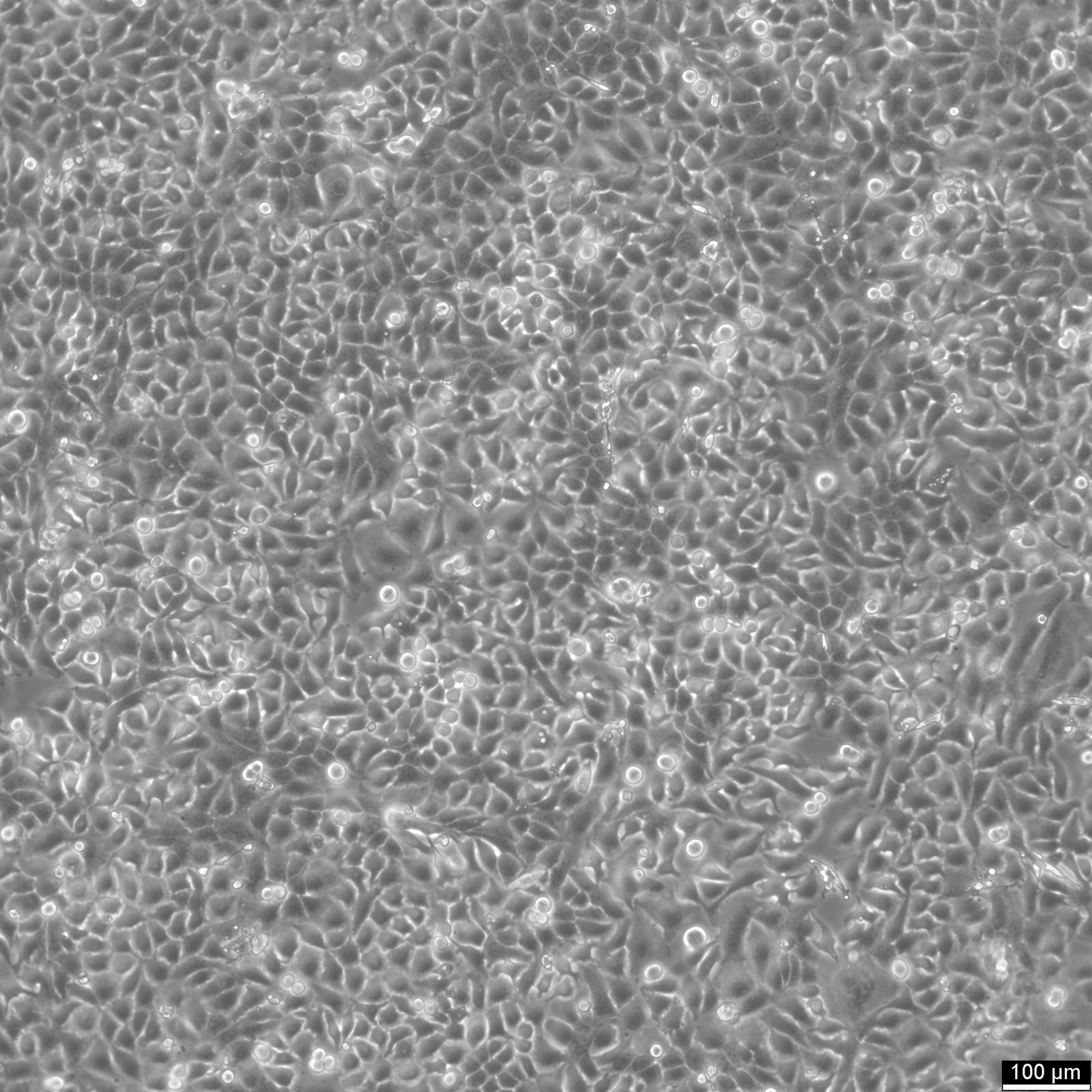
RT-112 Cells


















Product number:
300324
General information
| Description | This cell line was established by Dr. Carol Rigby, St. Paul?S Hospital, London as described by Benham et al. in 1976 from a human bladder carcinoma. |
|---|---|
| Organism | Human |
| Tissue | Bladder |
| Disease | Carcinoma |
| Synonyms | RT 112, RT112 |
Characteristics
| Age | Unspecified |
|---|---|
| Gender | Female |
| Ethnicity | Caucasian |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | RT-112 (Cytion catalog number 300324) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_1670 |
Biomolecular Data
| Protein expression | p53 positive, Cytokeratine (4),5,(6), 7, 8, 13, 17, 18, 19, Desmoplakin |
|---|---|
| Isoenzymes | Yes, in nude mice |
| MSI-status | Stable (MSS) |
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | A ratio of 1:4 to 1:8 is recommended |
| Seeding density | Start culture at 2 to 3 x 10^4 cells/cm^2 and continue with a seeding density of 1 x 10^4 cells/cm^2. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|---|
| STR profile |
Amelogenin: x,y
CSF1PO: 10,11
D13S317: 13,14
D16S539: 11,13
D5S818: 10,13
D7S820: 12,11
TH01: 7
TPOX: 8,11
vWA: 14,17
D3S1358: 15
D21S11: 27,3
D18S51: 15
Penta E: 12,16
Penta D: 10,11
D8S1179: 13,15
FGA: 23
|
| HLA alleles |
A*: '26:01:01
B*: '27:05:02
C*: '01:02:01
DRB1*: '01:01:01
DQA1*: '01:01:01
DQB1*: '05:01:01
DPB1*: '01:01:01
E: '01:01:01
|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300324-040425 | Certificate of Analysis | 23. May. 2025 | 300324 |
| 300324-070923 | Certificate of Analysis | 23. May. 2025 | 300324 |
