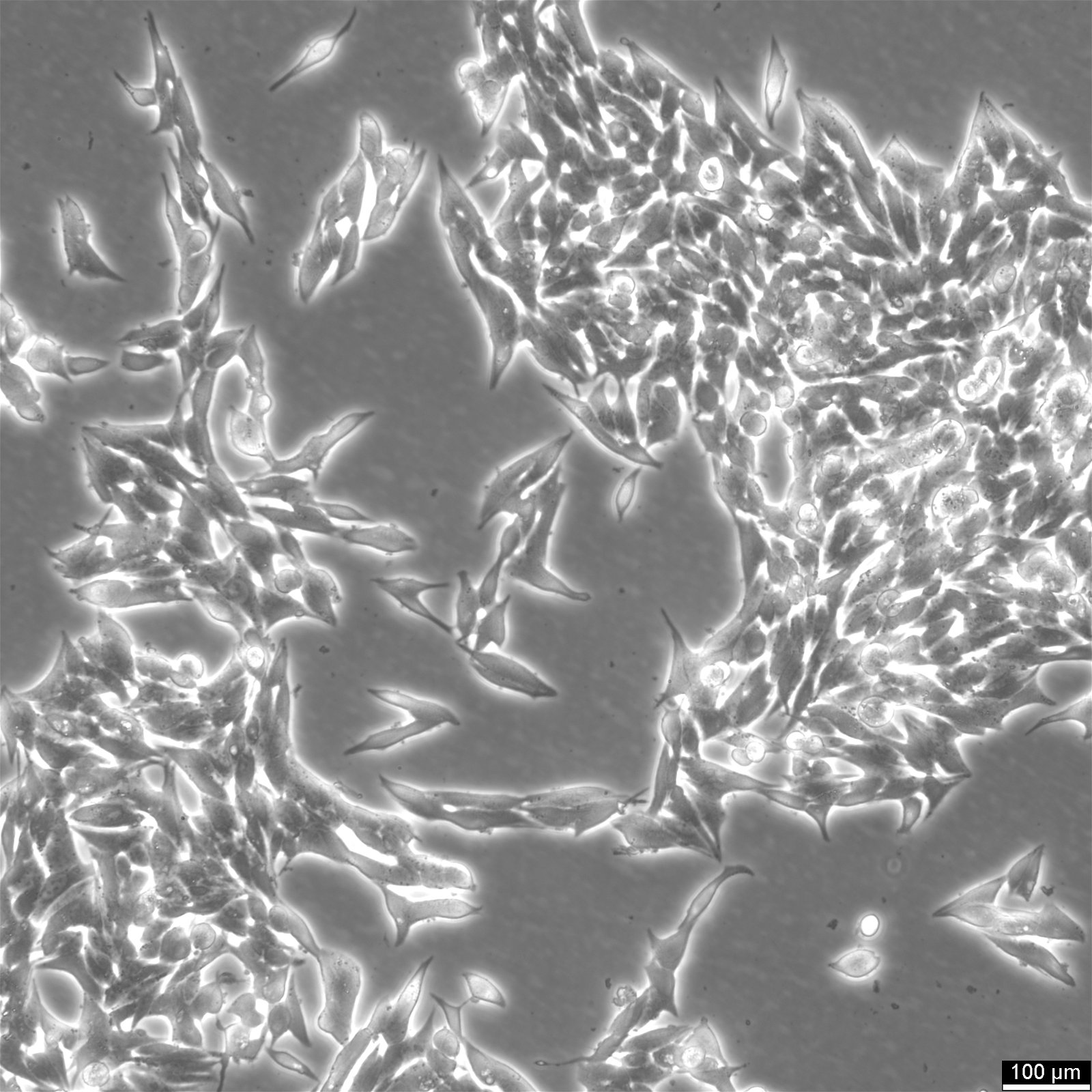
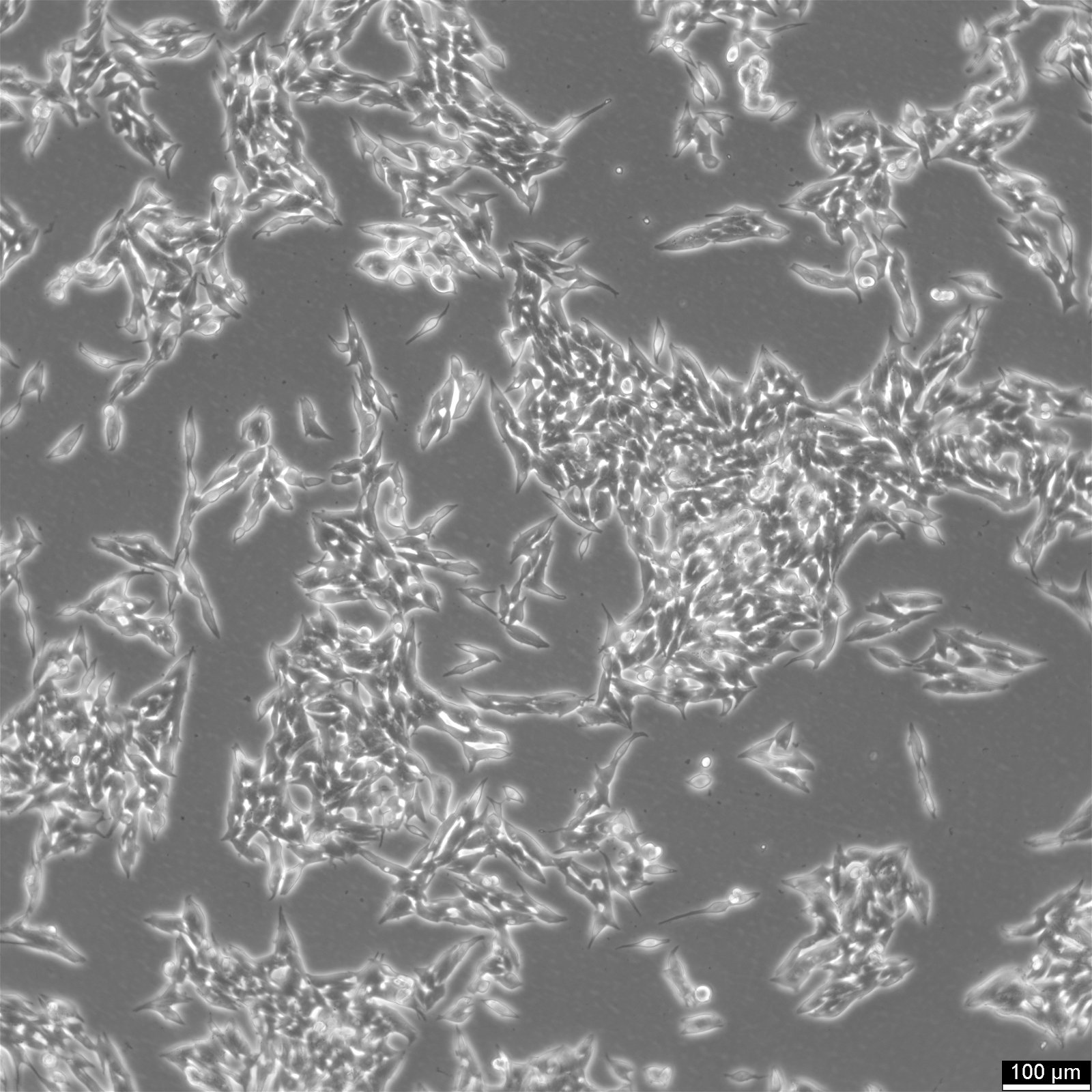
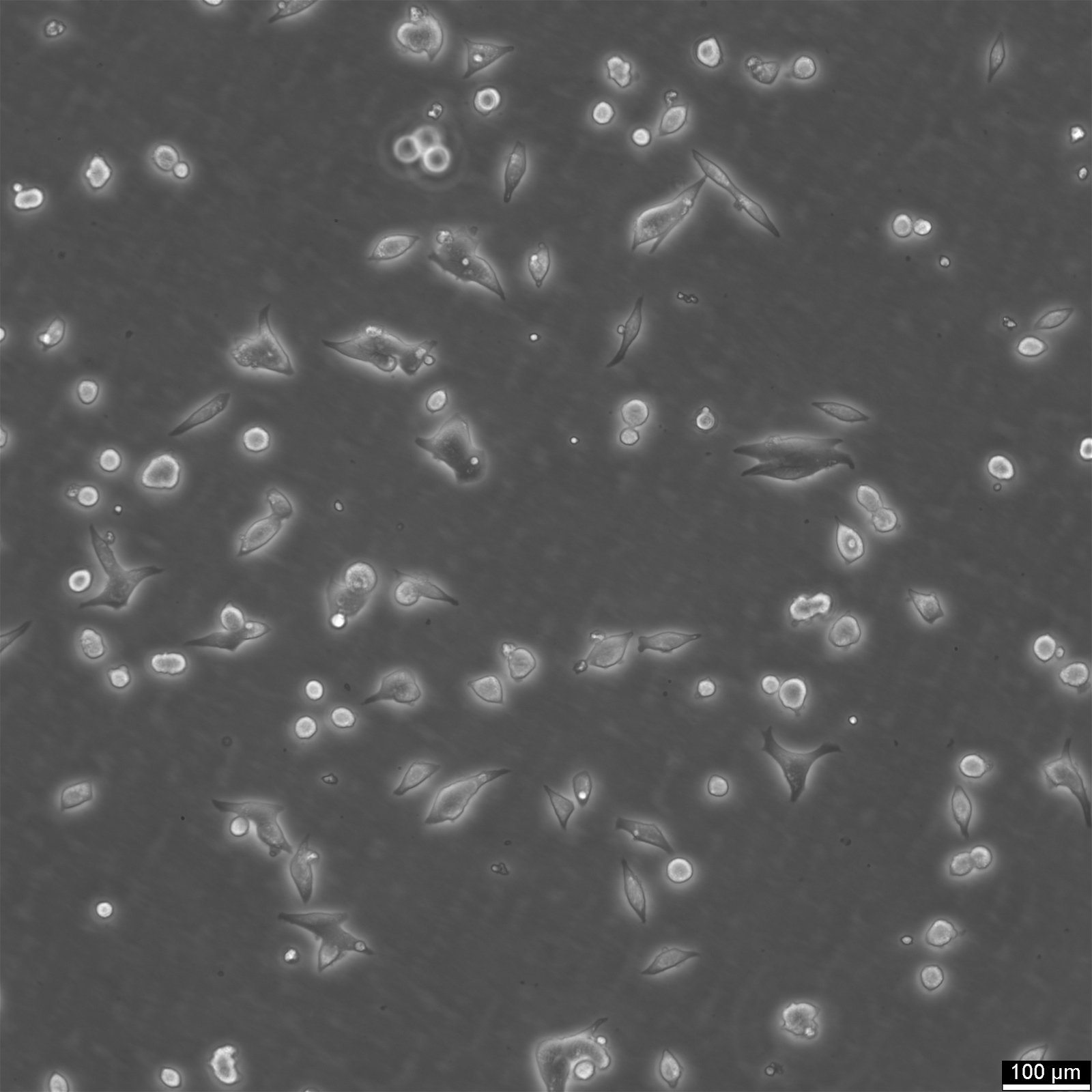
TT Cells









€430.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | TT cells continuously produce high levels of calcitonin and CEA.Immunoreactive calcitonin was found to be produced in cell culture at levels of 3900 pg/million cells and 7700 pg/million cells 24 and 72 hours respectively, after a medium change.CEA was found to accumulate to greater than 27 ng/million cells over a 72 hours period.Chromosomal analysis of the cell line and tumors induced in nude mice reveal an aneuploid human karyotype with several marker chromosomes.The initial characterization studies of the TT cell line were conducted using early passage TT cells cultivated in RPMI 1640 medium supplemented with 15% fetal bovine serum and 1mM L-glutamine.It is not known if the neuropeptides reported to be produced by this cell line when it was grown in RPMI 1640 medium are also produced by the cells when they are cultured in Ham's F-12K medium.Chromosomal analysis of the cell line and tumors induced in nude mice reveal an aneuploid human karyotype with several marker chromosomes. |
|---|---|
| Organism | Human |
| Tissue | Thyroid, medulla |
| Disease | Hereditary thyroid gland medullary carcinoma, Multiple endocrine neoplasia type 2 |
| Synonyms | MTC-TT |
Characteristics
| Age | 77 years |
|---|---|
| Gender | Female |
| Ethnicity | European |
| Morphology | Epithelial |
| Growth properties | Adherent |
Regulatory Data
| Citation | TT (Cytion catalog number 305027) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_1774 |
Biomolecular Data
| Protein expression | Calcitonin, Carcinoembryonic Antigen(CEA) |
|---|---|
| Tumorigenic | Yes |
Handling
| Culture Medium | Ham's F12K Medium, w: 2.0 mM L-Glutamine, w: 2.0 mM Sodium pyruvate, w: 2.5 g/L NaHCO3 (Cytion article number 820608a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS, 1% NEAA and 1mM Sodiumpyruvat |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305027-120925 | Certificate of Analysis | 05. Dec. 2025 | 305027 |
| 305027-141022 | Certificate of Analysis | 23. May. 2025 | 305027 |
Material Transfer Agreement
If you intend to use Cytion cell lines solely for internal research at a single research site, please complete and sign our Material Transfer Agreement (MTA) and submit it along with your order.
For any commercial applications - including but not limited to fee-for-service work, quality control testing, product release, diagnostic use, or regulatory studies - please complete the Intended Use Form so we can prepare a suitable agreement tailored to your project.
Please note: The MTA applies only to certain cell lines. If this notice and the MTA document appear on a product page, the agreement is applicable. For cell lines not covered by the MTA, no reference to the agreement will be shown. The MTA is not valid for customers in the Americas, China, or Taiwan. Please contact our U.S. entity to receive the appropriate agreement.
-
Required products
Required products
Freeze Medium CM-1 - 50 mlCryopreservation Media Variants: 50 mlCytion’s Freeze Medium CM-1 is a state-of-the-art cryopreservation medium designed to ensure the highest level of cell viability and functionality post-thaw. This versatile medium is suitable for a broad spectrum of cell types, including both human and animal cells, making it an essential tool for diverse research applications. Formulated with a meticulously balanced combination of cryoprotectants and essential nutrients, Freeze Medium CM-1 minimizes ice crystal formation and cellular stress during the freezing process, thus preserving cellular integrity.
Key features of Freeze Medium CM-1 include:
Broad Compatibility: Effective for a wide range of cell types, including primary cells, stem cells, and established cell lines.
High Viability: Optimized to maximize post-thaw cell recovery and viability, ensuring reliable experimental outcomes.
Ready-to-Use: Conveniently prepared and sterilized for immediate application, reducing preparation time and risk of contamination.
Enhanced Stability: Maintains consistent performance under standard cryopreservation conditions, ensuring reproducible results.
Long Shelf Life: CM-1 is a serum-containing, ready-to-use cryopreservation medium that can be stored in the refrigerator for up to one year.
Using CM-1 for Freezing Cells
To use CM-1 for freezing both adherent and suspension cells, follow these steps:
For adherent cells, wash and dissociate them from the culture substrate. For suspension cells, proceed directly to the next step.
Count the cells to ensure they are at the proper concentration.
Centrifuge the cells to pellet them, then resuspend in CM-1 freeze medium.
Transfer the resuspended cells into cryovials.
Use a slow-freezing method before transferring the cells to long-term storage.
Method
Description
Steps
❄️
Manual Freezing
A step-by-step method involving gradual temperature reduction to ensure cell viability.
1️⃣ Place cells in freeze medium in a 4°C freezer for 40 minutes.
2️⃣ Transfer to a -80°C freezer for 24 hours.
3️⃣ Store cells in liquid nitrogen for long-term preservation.
❄️
Using Mr. Frosty
A convenient device that allows for controlled freezing rates without electrical power.
1️⃣ Prepare cells in cryovials with freeze medium.
2️⃣ Place cryovials in Mr. Frosty container.
3️⃣ Store at -80°C for 24 hours before transferring to liquid nitrogen.
❄️
Controlled-Rate Freezer
A high-precision freezer by Thermo Fisher or other manufacturers designed for controlled temperature reduction.
1️⃣ Program the device to gradually decrease the temperature.
2️⃣ Place prepared cells in the freezer.
3️⃣ After the freezing cycle, transfer cells to liquid nitrogen.
Store the cryovials at temperatures below -130°C or in liquid nitrogen for long-term preservation.
Ingredients
Contains FBS, DMSO, Glucose, Salts
Buffering capacity: pH = 7.2 to 7.6
Cytion’s Freeze Medium CM-1 offers a reliable solution for cryopreservation, ensuring high cell viability and functionality post-thaw for a wide range of research applications.€59.00*Ham's F12K Medium, w: 2.0 mM L-Glutamine, w: 2.0 mM Sodium pyruvate, w: 2.5 g/L NaHCO3Introducing Ham's F-12K (Kaighn's) Medium, a specialized modification of Ham's F-12 medium designed to meet the unique requirements of biological research. This advanced medium offers distinct advantages, enhancing the cultivation of primary human hepatocytes, as well as rat and chicken liver cells, particularly in reduced serum conditions.
Ham's F-12K (Kaighn's) Medium is carefully formulated to optimize cell culture conditions. It features an enriched composition, providing elevated levels of essential components such as amino acids and sodium pyruvate, as well as additional elements including putrescine, thymidine, hypoxanthine, and zinc. These additions enable researchers to supplement the medium with minimal serum or defined components for specific cell types, facilitating precise experimental conditions.
Notably, Ham's F-12K (Kaighn's) Medium does not contain proteins or growth factors. Consequently, supplementation with growth factors and Fetal Bovine Serum (FBS) is often necessary, allowing researchers to tailor the medium to the requirements of their specific cell lines. For optimal performance, the concentration of FBS must be carefully optimized for each cell line, ensuring optimal growth and functionality.
To maintain physiological pH, Ham's F-12K (Kaighn's) Medium employs a sodium bicarbonate buffer system (2.5 g/L), necessitating a controlled 5-10% CO2 environment during cultivation. This ensures the medium's pH remains within the ideal range for cell growth and viability.
Quality control
pH = 7.2 +/
- 0.02 at 20-25°C.
Each lot has been tested for sterility and absence of mycoplasma and bacteria.
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Freezing as well as warming up to +37°C minimize the quality of the product.
Do not heat the medium to more than 37°C or use uncontrollable sources of heat (e.g., microwave appliances).
If only a part of the medium is to be used, remove this amount from the bottle and warm it up at room temperature.
Shelf life for any medium but the basic medium is 8 weeks from the date of manufacture.
Composition
Components
mg/L
Inorganic Salts
Calcium chloride x 2H2O
135,24
Copper(II) sulfate x 5H2O
0,00
Iron (II) sulfate x 7H2O
0,83
Magnesium chloride x 6H2O
105,72
Magnesium sulfate x 7H2O
394,49
Potassium chloride
283,29
Potassium dihydrogen phosphate
58,52
Sodium chloride
7597,20
di-Sodium hydrogen phosphateanhydrous
115,02
Zinc sulfate x 7H2O
0,14
Other Components
D(+)-Glucose anhydrous
1260,00
Hypoxanthine
4,08
DL-α-Lipoic acid
0,21
Phenol red
3,00
Putrescine x 2HCl
0,32
Sodium pyruvate
220,00
NaHCO3
2500,00
Thymidine
0,73
Amino Acids
L-Alanine
17,82
L-Arginine x HCl
421,40
L-Asparagine x H2O
30,02
L-Aspartic acid
26,62
L-Cysteine x HCl x H2O
70,24
L-Glutamine
292,20
L-Glutamic acid
29,42
Glycine
15,01
L-Histidine x HCl x H2O
41,92
L-Isoleucine
7,87
L-Leucine
26,24
L-Lysine x HCl
73,04
L-Methionine
8,95
L-Phenylalanine
9,91
L-Proline
69,06
L-Serine
21,02
L-Threonine
23,82
L-Tryptophan
4,08
L-Tyrosine
10,87
L-Valine
23,42
Vitamins
D(+)-Biotin
0,07
D-Calcium pantothenate
0,48
Choline chloride
13,96
Folic acid
1,32
myo-Inositol
18,02
Nicotinamide
0,04
Pyridoxine x HCl
0,06
Riboflavin
0,04
Thiamine x HCl
0,34
Vitamin B12
1,36€108.00*AccutaseVariants: 100 mlAccutase Cell Dissociation Reagent
- A Gentle Alternative to Trypsin
Accutase is a cell detachment solution that is revolutionizing the cell culture industry. It is a mix of proteolytic and collagenolytic enzymes that mimics the action of trypsin and collagenase. Unlike trypsin, Accutase does not contain any mammalian or bacterial components and is much gentler on cells, making it an ideal solution for the routine detachment of cells from standard tissue culture plasticware and adhesion coated plasticware. In this blog post, we will explore the benefits and uses of Accutase and how it is changing the game in cell culture.
Advantages of Accutase
Accutase has several advantages over traditional trypsin solutions. Firstly, it can be used whenever gentle and efficient detachment of any adherent cell line is needed, making it a direct replacement for trypsin. Secondly, Accutase works extremely well on embryonic and neuronal stem cells, and it has been shown to maintain the viability of these cells after passaging. Thirdly, Accutase preserves most epitopes for subsequent flow cytometry analysis, making it ideal for cell surface marker analysis.
Additionally, Accutase does not need to be neutralized when passaging adherent cells. The addition of more media after the cells are split dilutes Accutase so it is no longer able to detach cells. This eliminates the need for an inactivation step and saves time for cell culture technicians. Finally, Accutase does not need to be aliquoted, and a bottle is stable in the refrigerator for 2 months.
Applications of Accutase
Accutase is a direct replacement for trypsin solution and can be used for the passaging of cell lines. Additionally, Accutase performs well when detaching cells for the analysis of many cell surface markers using flow cytometry and for cell sorting. Other downstream applications of Accutase treatment include analysis of cell surface markers, virus growth assay, cell proliferation, tumor cell migration assays, routine cell passage, production scale-up (bioreactor), and flow cytometry.
Composition of Accutase
Accutase contains no mammalian or bacterial components and is a natural enzyme mixture with proteolytic and collagenolytic enzyme activity. It is formulated at a much lower concentration than trypsin and collagenase, making it less toxic and gentler, but just as effective.
Efficiency of Accutase
Accutase has been shown to be efficient in detaching primary and stem cells and maintaining high cell viability compared to animal origin enzymes such as trypsin. 100% of cells are recovered after 10 minutes, and there is no harm in leaving cells in Accutase for up to 45 minutes, thanks to autodigestion of Accutase.
In summary
In conclusion, Accutase is a powerful solution that is changing the game in cell culture. With its gentle nature, efficiency, and versatility, Accutase is the ideal alternative to trypsin. If you are looking for a reliable and efficient solution for cell detachment, Accutase is the solution for you.€75.00*Antibiotic/Antimycotic Solution (100x)Product Overview
Volume: 100 ml
Storage: ≤-15°C
Sterility: Sterile-filtered
Antibiotic/Antimycotic Solution (100x) is a sterile, ready-to-use concentrate designed to reduce microbial contamination risks in cell culture and related laboratory applications. This 100x solution contains a well-established combination of penicillin, streptomycin, and amphotericin B—providing broad-spectrum antimicrobial activity against Gram-positive and Gram-negative bacteria, yeasts, and filamentous fungi. The formulation is suitable for use in eukaryotic cell cultures, bacterial media, and other contamination-sensitive systems, supporting clean and consistent lab operations.
Application and Benefits Optimized for routine research protocols, this solution is widely used to maintain aseptic conditions in cell culture workflows. It offers reliable performance in contamination-sensitive environments, helping researchers reduce the risk of microbial overgrowth without compromising cell health or experimental reproducibility. The sterile-filtered formulation eliminates the need for additional solubilization steps, supporting streamlined media preparation and reducing variability in daily lab procedures.
Usage and Compatibility To achieve standard working concentrations, dilute the solution 1:100 into your complete culture medium. The product is compatible with a broad range of mammalian cell lines and basal media. With consistent stock availability, researchers benefit from dependable supply continuity and simplified logistics planning. The solution should be stored at ≤ –15 °C and protected from repeated freeze-thaw cycles to maintain stability. For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.€45.00*PBSPhosphate-Buffered Saline (PBS) Solution
Phosphate-buffered saline (PBS) is a widely used buffer solution in biological and chemical research. It plays a crucial role in maintaining the pH balance and osmolarity during various experimental procedures, including tissue processing and cell culture. Our PBS solution is meticulously formulated with high-purity ingredients to ensure stability and reliability in every experiment. The osmolarity and ion concentrations of our PBS closely mimic those of the human body, making it isotonic and non-toxic to most cells.
Composition of Our PBS Solution
Our PBS solution is a pH-adjusted blend of ultrapure-grade phosphate buffers and saline solutions. At a 1X working concentration, it contains:
8000 mg/L Sodium chloride (NaCl)
200 mg/L Potassium chloride (KCl)
1150 mg/L Sodium phosphate dibasic anhydrous (Na2HPO4)
200 mg/L Potassium phosphate monobasic anhydrous (KH2PO4)
This composition ensures an optimal pH and ionic balance, suitable for a wide range of biological applications.
Applications of Our PBS Solution
Our PBS solution is ideal for various applications in biological research. Its isotonic and non-toxic properties make it suitable for substance dilution and cell container rinsing. PBS solutions containing EDTA are effective for disengaging attached and clumped cells. However, divalent metals such as zinc should not be added to PBS, as this can cause precipitation. In such cases, Good's buffers are recommended. Additionally, our PBS solution is an acceptable alternative to viral transport medium for the transport and storage of RNA viruses, including SARS-CoV-2.
Quality Control
Sterile-filtered
Storage and Shelf Life
Store at +2°C to +25°C, protected from light.
Once opened, store at 2°C to 25°C and use within 24 months.
Shipping Conditions
Ambient temperature
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Avoid freezing and frequent warming to +37°C, as it reduces product quality.
Do not heat the medium beyond 37°C or use uncontrolled heat sources such as microwave appliances.
If only part of the medium is to be used, remove the required amount and warm it to room temperature before use.
Composition
Category
Components
Concentration (mg/L)
Salts
Potassium chloride
200
Potassium phosphate monobasic anhydrous
200
Sodium chloride
8000
Sodium phosphate dibasic anhydrous
1150€20.00*





