H9c2(2-1) Cells
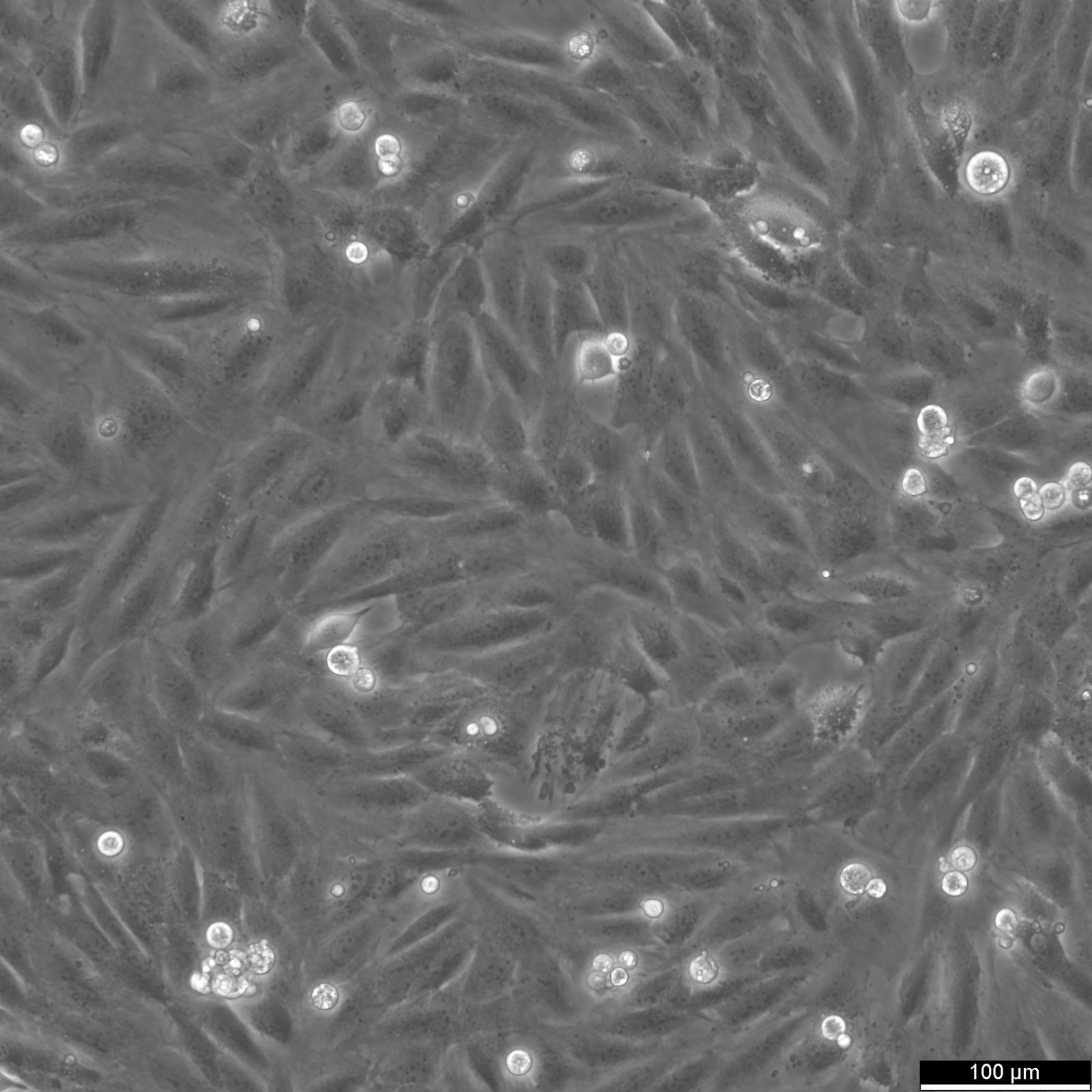

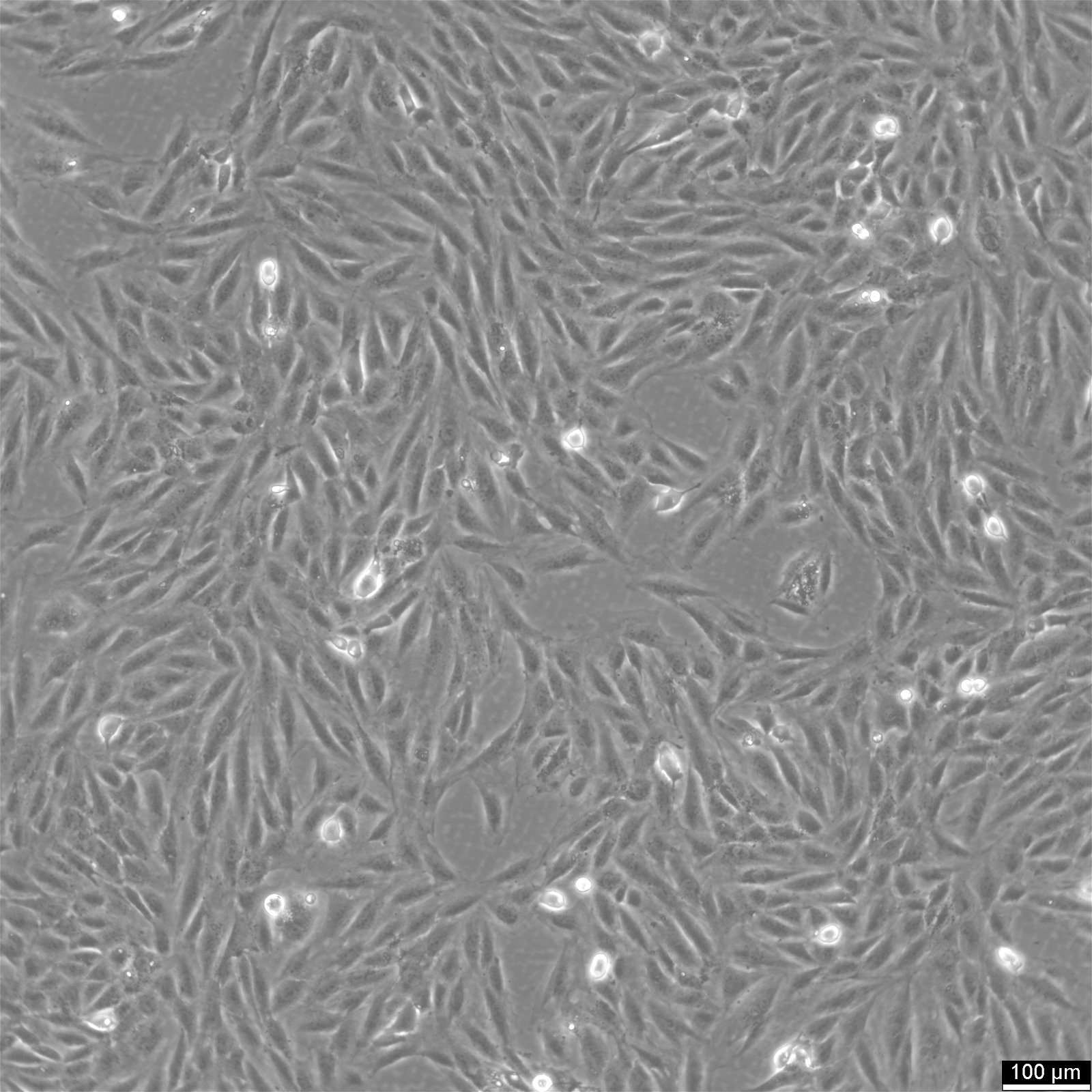

































General information
| Description | H9c2(2-1) cells, derived from the ventricular myoblasts of embryonic BD1X rat hearts, are a subclone of the original H9 cell line established in the early 1990s. These cells are immortalized myoblasts that are commonly used in vitro to study cardiac metabolism, physiology, and pathophysiology, including myocardial ischemia, hypertrophy, and apoptosis mechanisms. Phenotypically, H9c2 cells exhibit characteristics of skeletal muscle but retain the ability to adopt a cardiac muscle phenotype under specific experimental conditions, such as differentiation induced by retinoic acid or other agents. This flexibility makes them a valuable model for investigating cardiac muscle behavior in response to various physiological and pharmacological stimuli. Genetically, H9c2 cells are diploid, facilitating their use in genetic studies, where maintaining a stable karyotype is crucial. Research employing H9c2(2-1) cells has contributed significantly to understanding cellular responses to oxidative stress, mitochondrial dysfunction, and the protective roles of various pharmacological agents against cardiotoxicity. This cell line remains a cornerstone in cardiomyocyte-related research, offering a reproducible, controlled model to elucidate the complex biological and molecular mechanisms underlying cardiac function and diseases. |
|---|---|
| Organism | Rat |
| Tissue | Heart, myocardium |
| Synonyms | H9c2 (2-1), H9c2, H9C2 |
Characteristics
| Breed/Subspecies | BDIx |
|---|---|
| Age | Embryo |
| Morphology | Myoblast |
| Growth properties | Adherent |
Regulatory Data
| Citation | H9c2(2-1) (Cytion catalog number 305203) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 10116 |
| CellosaurusAccession | CVCL_0286 |
Biomolecular Data
| Receptors expressed | Acetylcholine, expressed |
|---|---|
| Protein expression | Myokinase, Creatine Phosphokinase, Myosin |
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 3.7 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:2 to 1:4 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305203-250325 | Certificate of Analysis | 23. May. 2025 | 305203 |
| 305203-271222 | Certificate of Analysis | 23. May. 2025 | 305203 |
