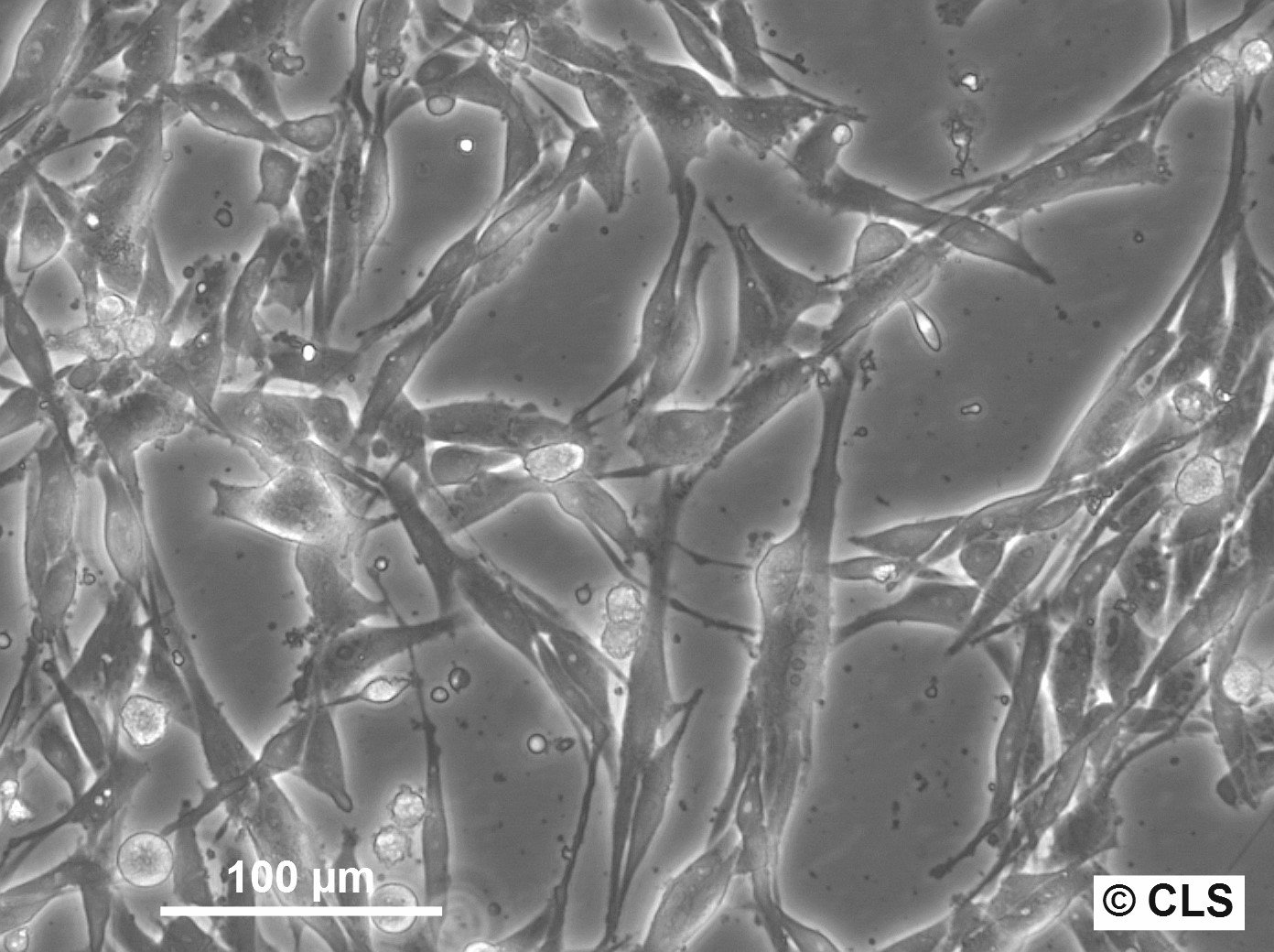
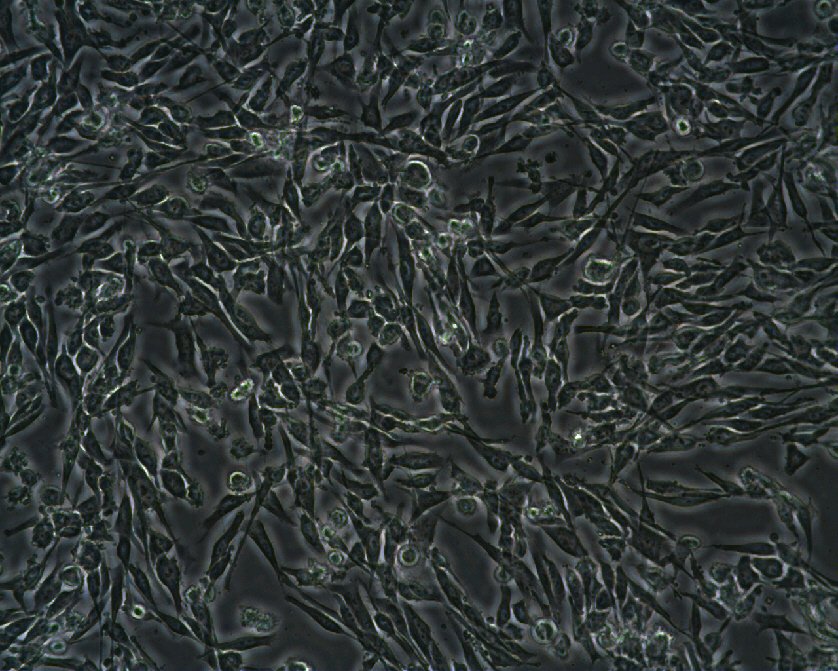
MML-1 Cells






















€800.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The MML-1 cell line is a melanoma cell line derived from malignant melanoma. This cell line is used primarily to study melanoma biology, particularly the role of extracellular vesicles (EVs) in cell-to-cell communication and tumor progression. MML-1 cells release various subtypes of EVs, including exosomes, microvesicles, and apoptotic bodies, each carrying distinct RNA cargos, such as microRNAs (miRNAs) and other non-coding RNAs. Studies using MML-1 cells have demonstrated that exosomes released from these cells contain specific miRNAs like miR-214-3p, miR-199a-3p, and miR-155-5p, which are closely associated with melanoma progression and metastasis. These miRNAs are enriched in exosomes compared to other EV types and have been linked to important melanoma-related pathways, such as the regulation of the MAPK signaling pathway and tumor microenvironment interactions. Interestingly, comparisons of miRNA profiles from MML-1-derived exosomes with clinical samples of melanoma show a significant overlap, indicating the clinical relevance of this cell model in understanding melanoma progression. In addition to miRNAs, MML-1 cells also release other non-coding RNAs such as small nucleolar RNAs (snoRNAs) and mitochondrial-associated transfer RNAs (mt-RNAs), which are differentially distributed among the EV subtypes. These findings highlight the MML-1 cell line's utility in studying the molecular mechanisms of melanoma, particularly how tumor cells communicate via EVs and influence their microenvironment. |
|---|---|
| Organism | Human |
| Tissue | Skin |
| Disease | Melanoma |
| Synonyms | MML1 |
Characteristics
| Age | Unspecified |
|---|---|
| Gender | Unspecified |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | MML-1 (Cytion catalog number 300288) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_6004 |
Biomolecular Data
| Protein expression | P53 positive |
|---|---|
| Tumorigenic | Yes, in nude mice |
| Reverse transcriptase | Negative |
| Mutational profile | V600E type BRAF Mutation was determined by DNA based methods (sequencing, RT-PCR) and protein based methods (Western Blot). |
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Material Transfer Agreement
If you intend to use Cytion cell lines solely for internal research at a single research site, please complete and sign our Material Transfer Agreement (MTA) and submit it along with your order.
For any commercial applications - including but not limited to fee-for-service work, quality control testing, product release, diagnostic use, or regulatory studies - please complete the Intended Use Form so we can prepare a suitable agreement tailored to your project.
Please note: The MTA applies only to certain cell lines. If this notice and the MTA document appear on a product page, the agreement is applicable. For cell lines not covered by the MTA, no reference to the agreement will be shown. The MTA is not valid for customers in the Americas, China, or Taiwan. Please contact our U.S. entity to receive the appropriate agreement.
