Wilms8 Cells












USD$800.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
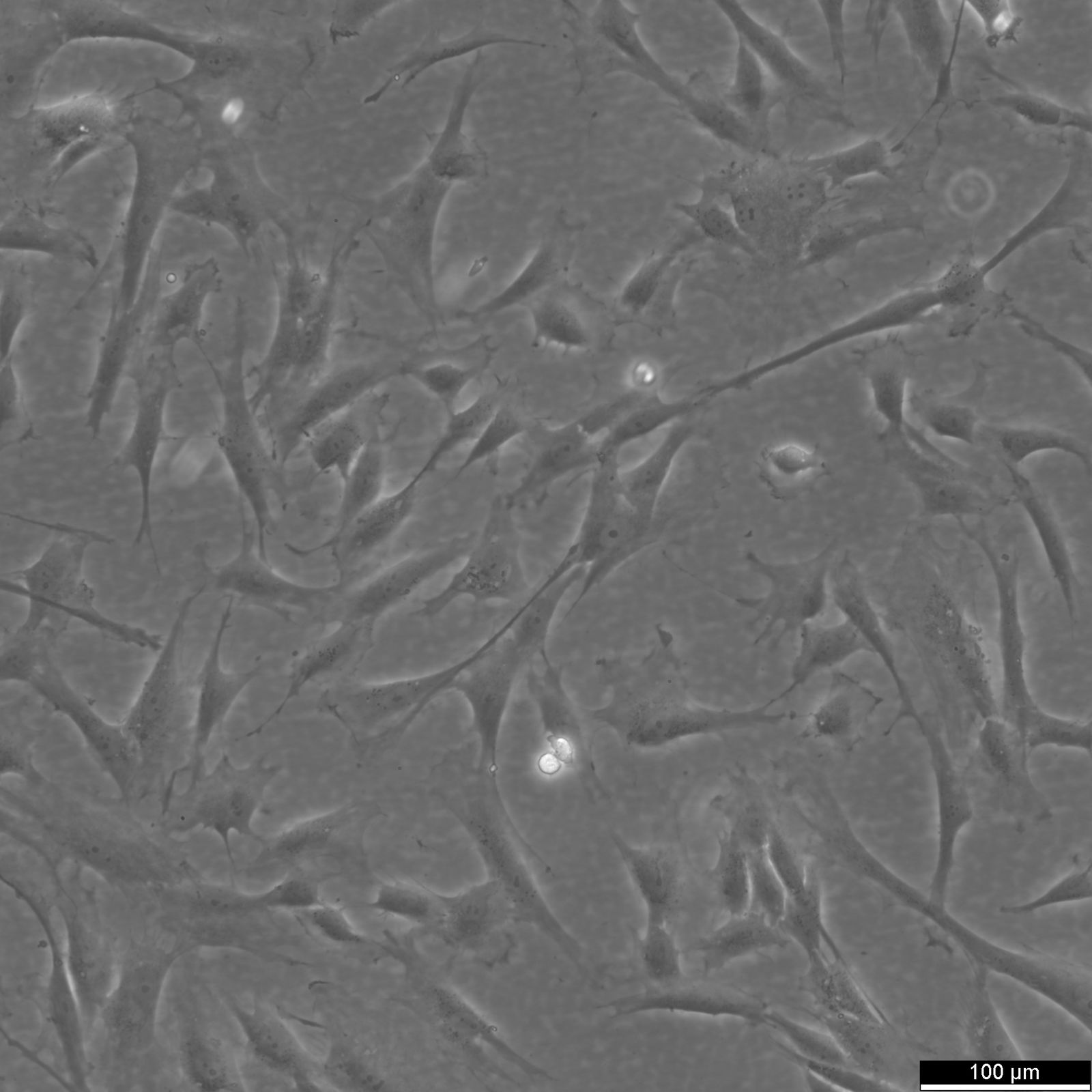
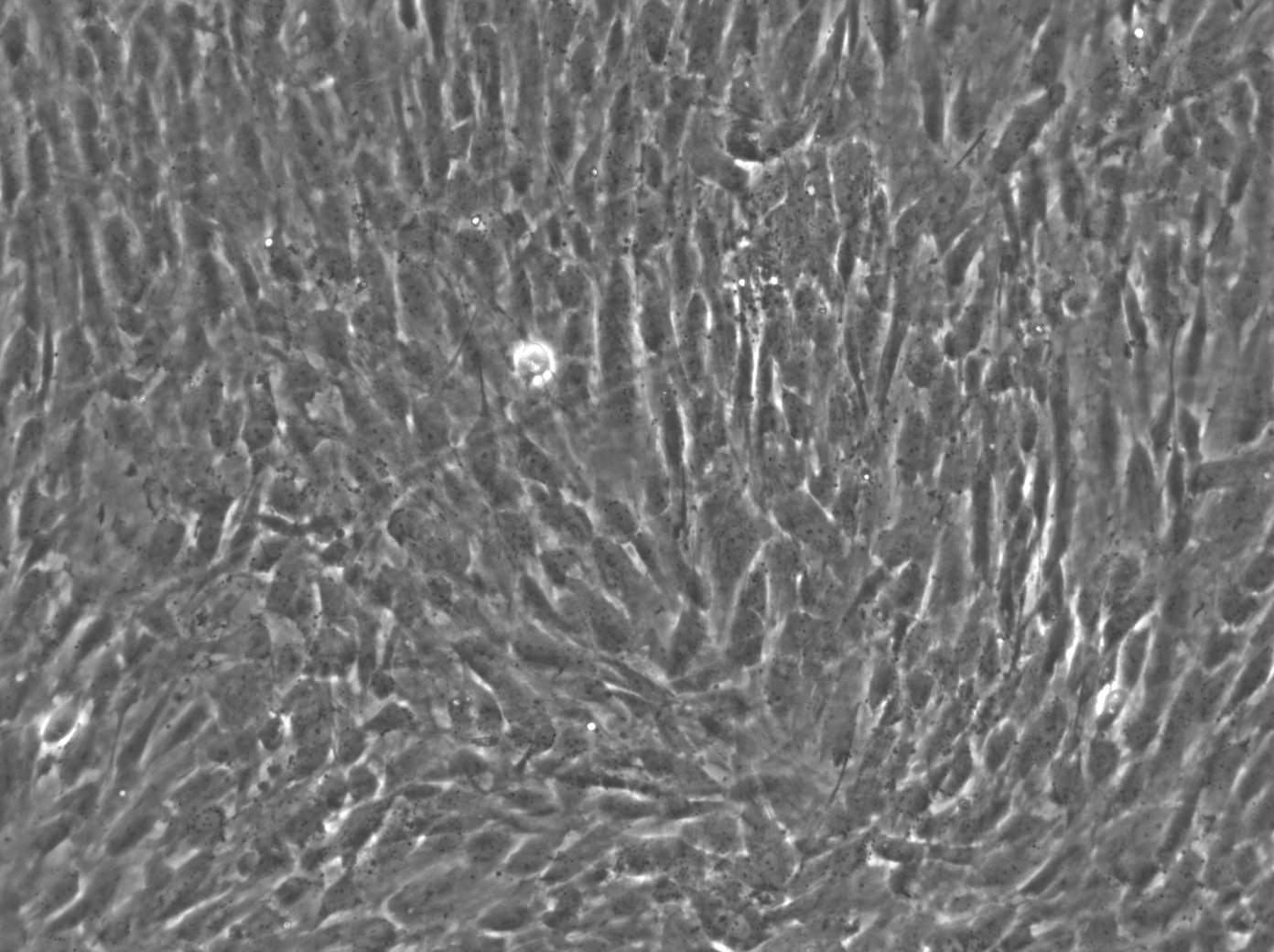
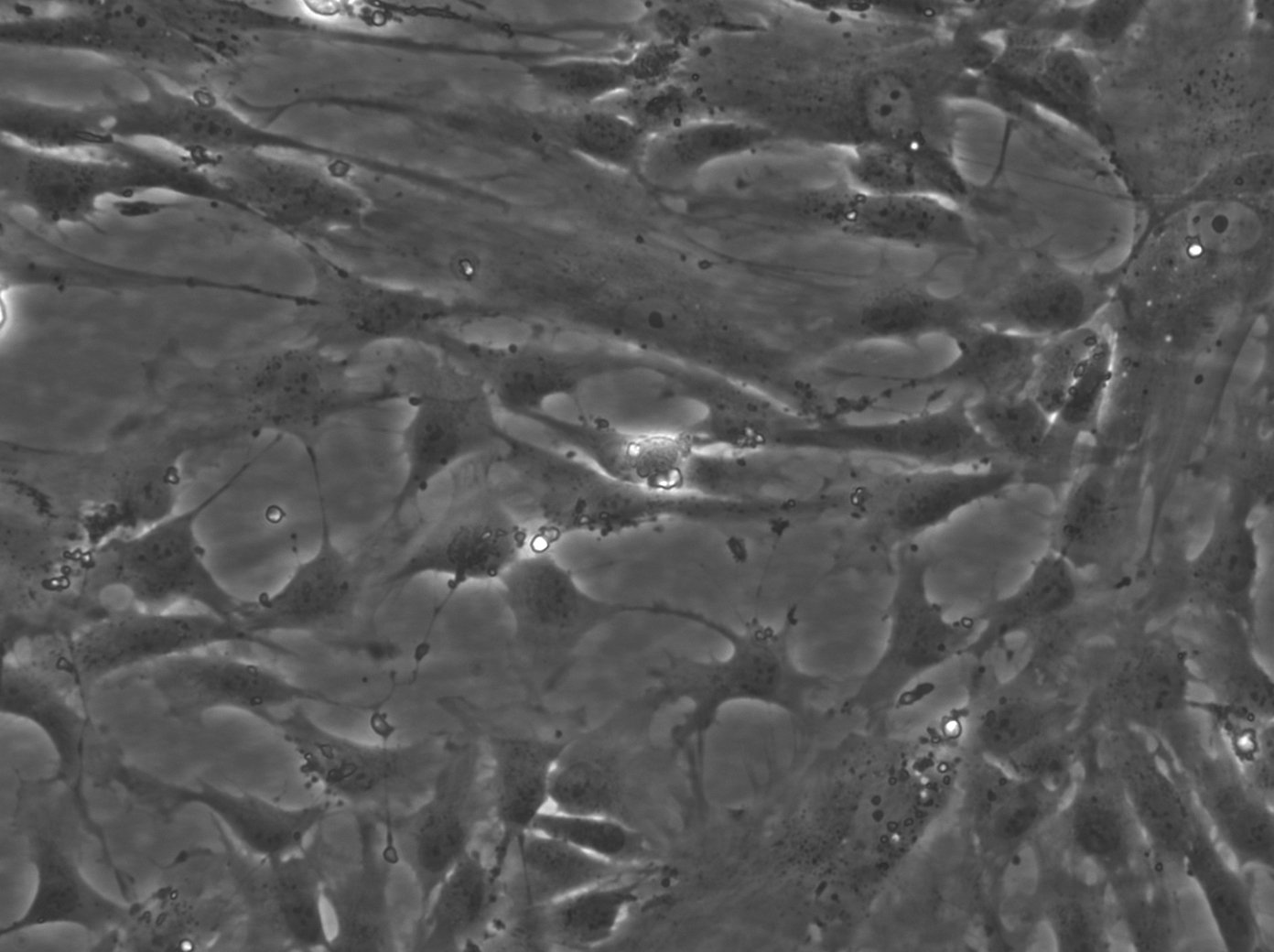
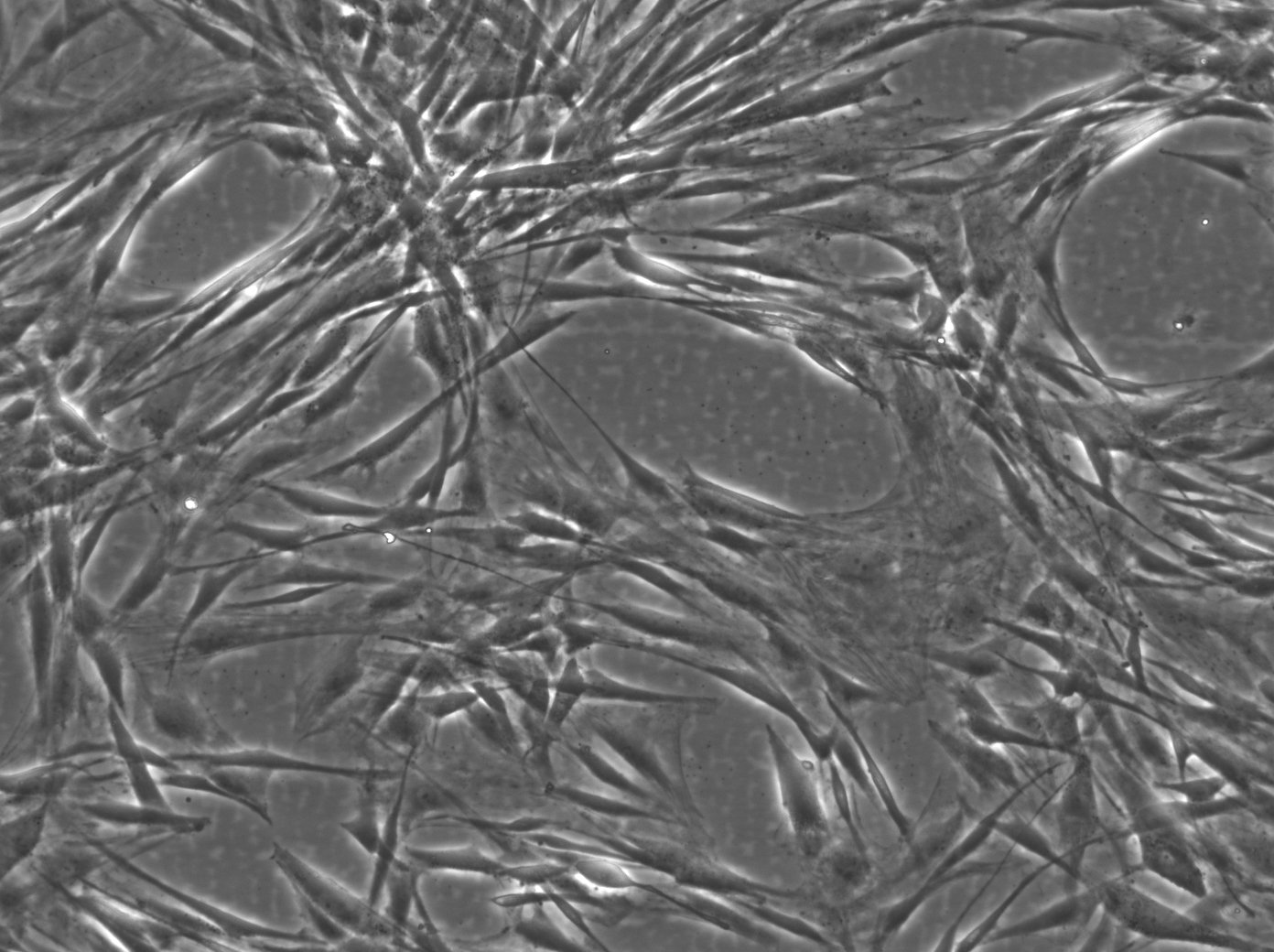
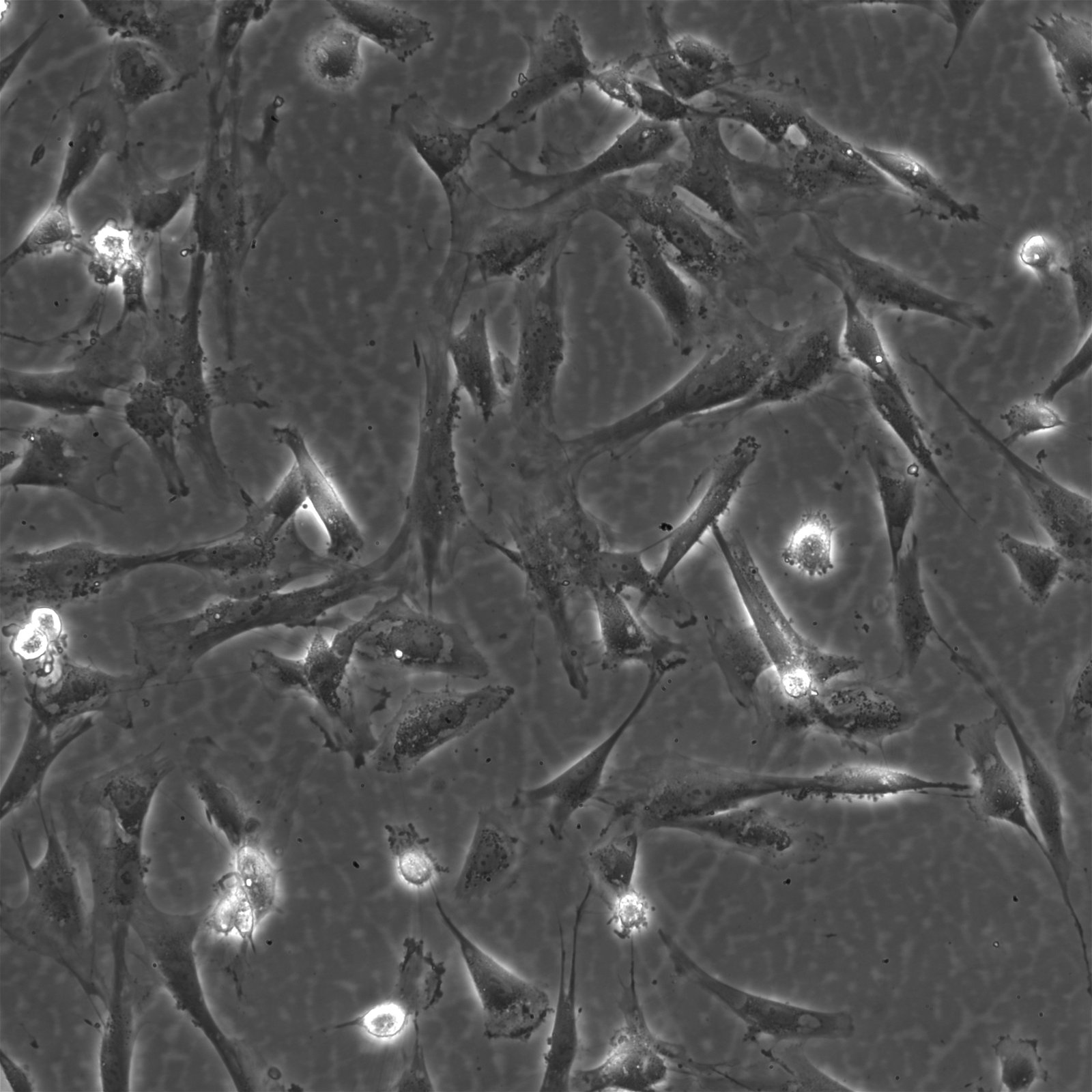
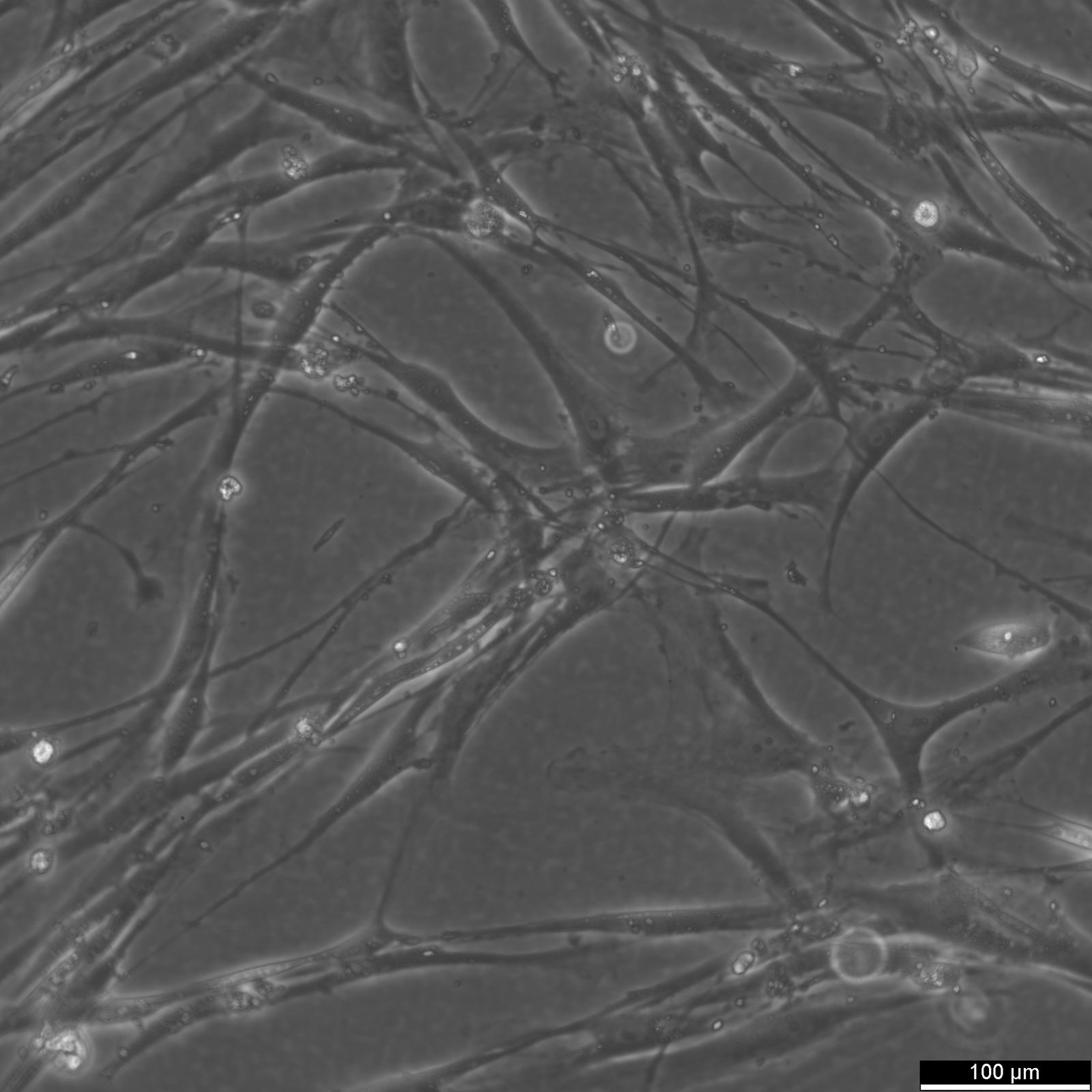
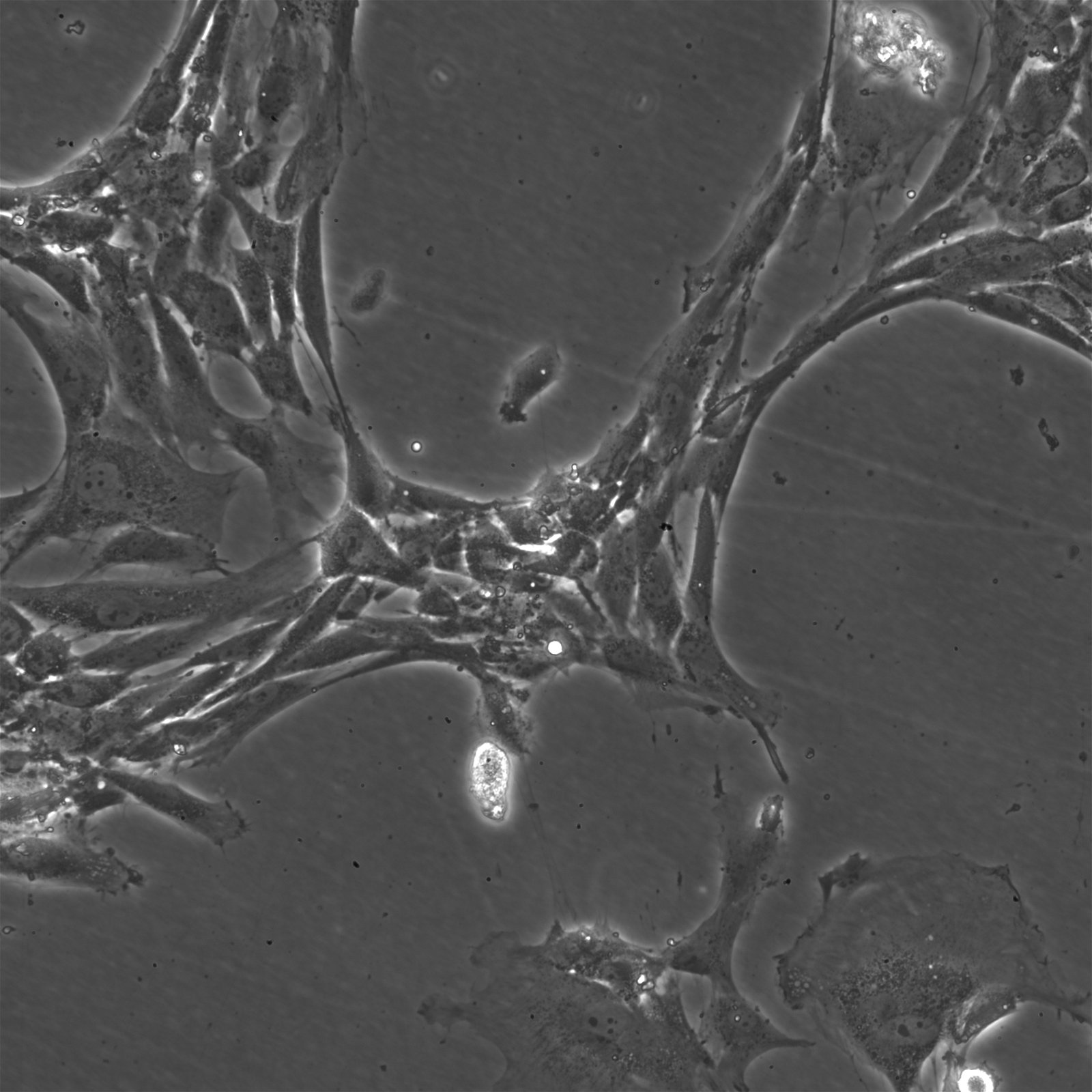
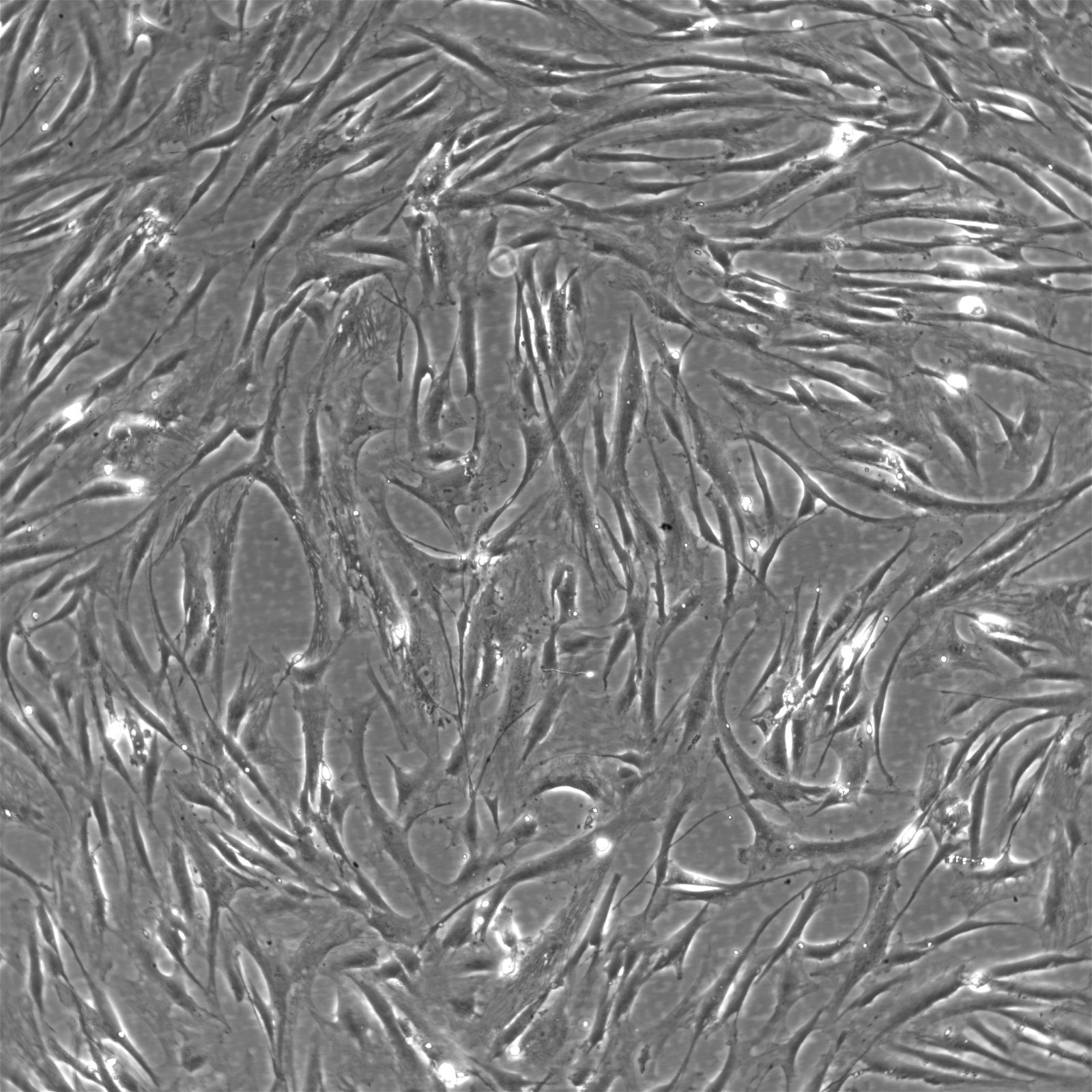
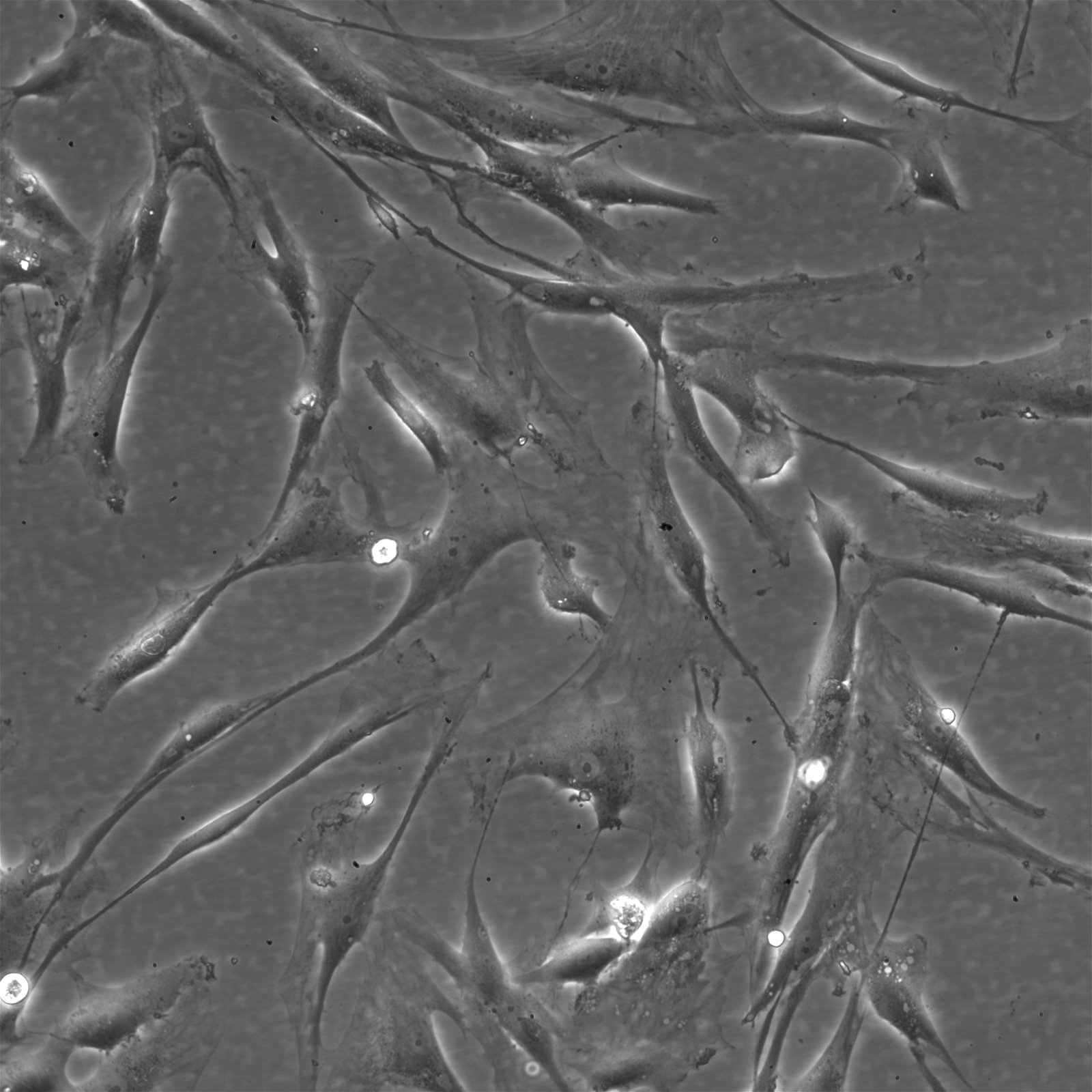
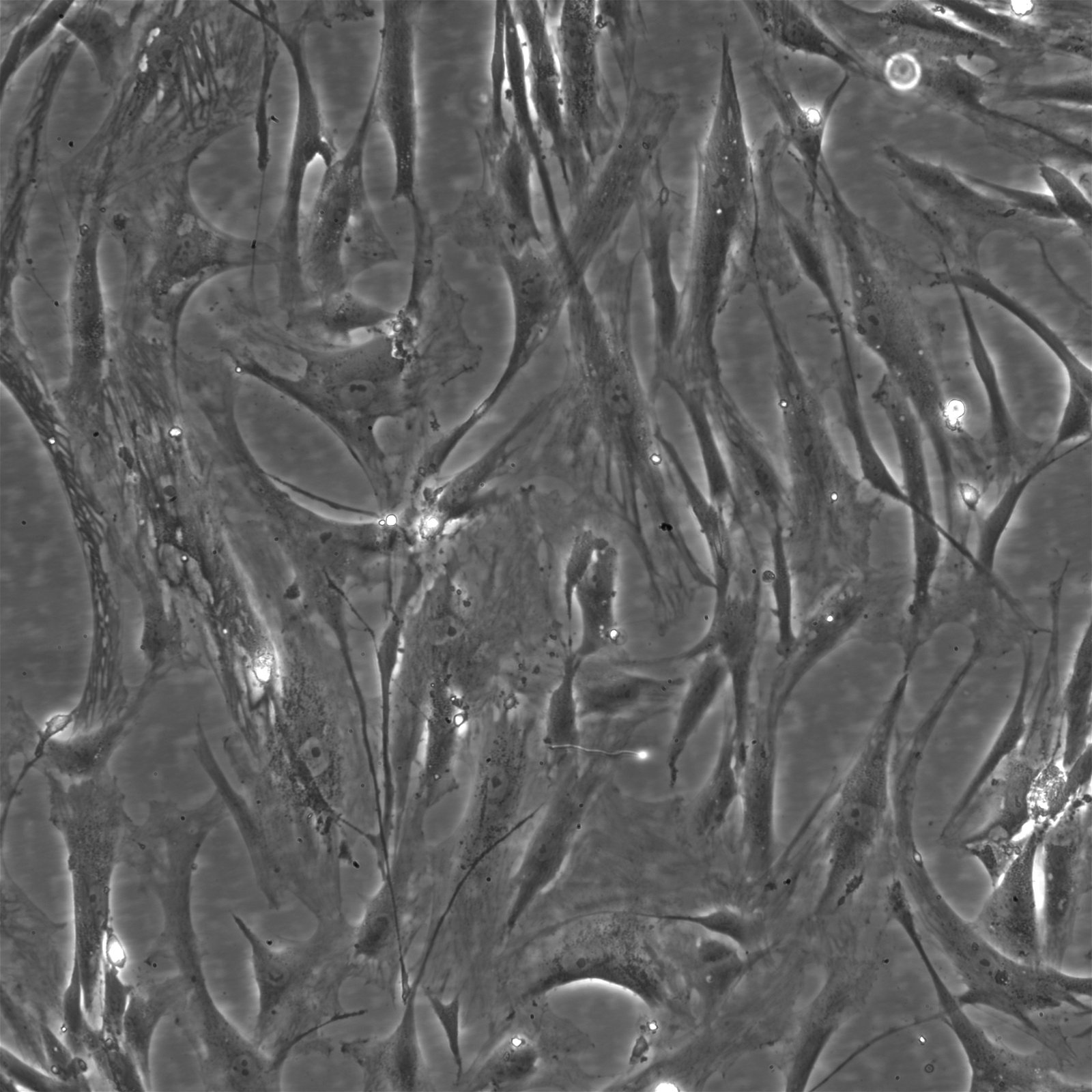
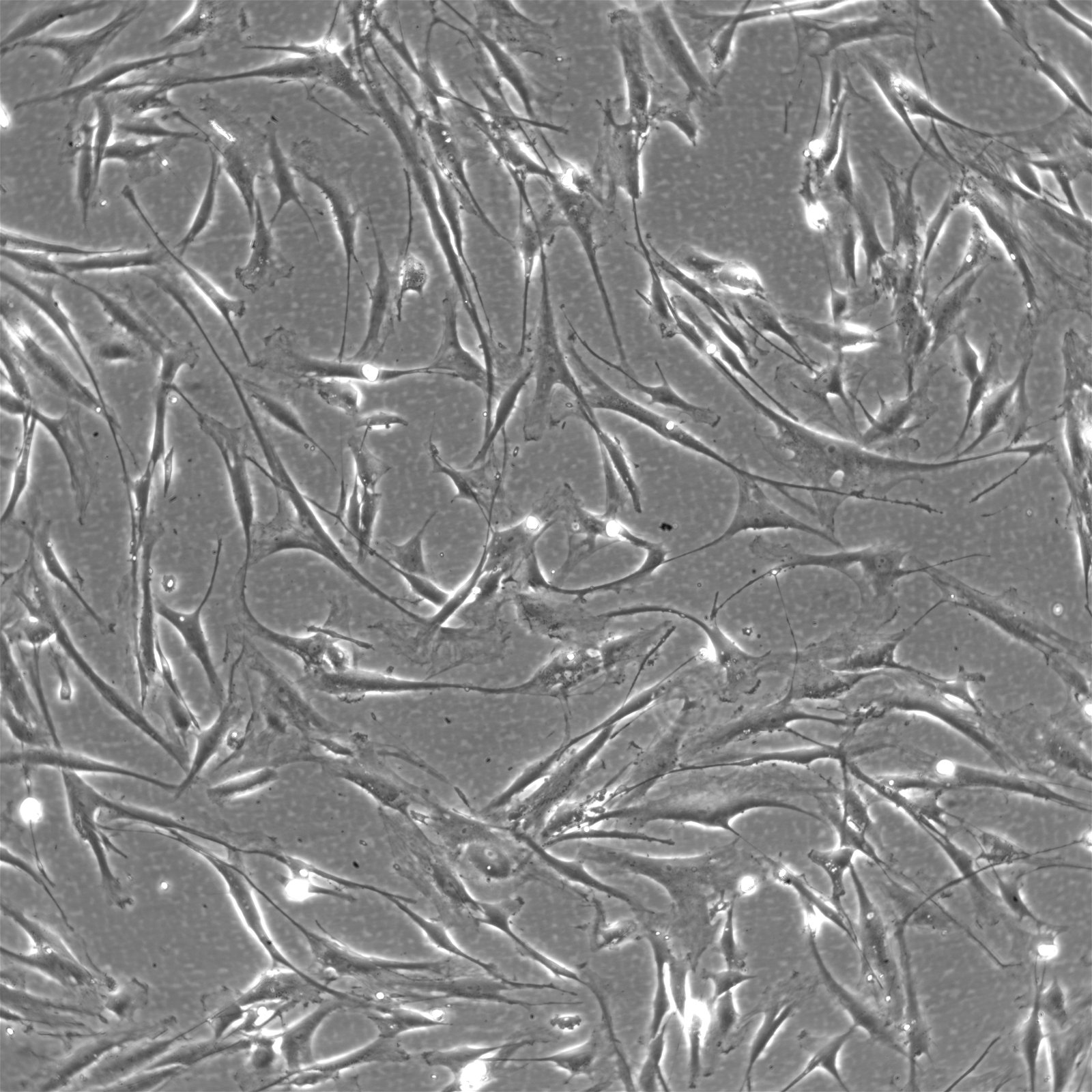
| Description | The Wilms8 cell line was derived from a primary Wilms tumor in a pediatric patient with a germline WT1 mutation. This cell line is characterized by a homozygous nonsense mutation in the WT1 gene (c.1168 C>T, p.R390X), leading to a complete loss of WT1 function. WT1 is crucial for normal kidney development, and its inactivation is a common feature in certain aggressive subtypes of Wilms tumor, particularly those that exhibit mesenchymal differentiation. Wilms8, therefore, provides a valuable model for studying the effects of WT1 loss on tumorigenesis, especially in the context of Wilms tumors that arise with a pronounced stromal component. In addition to the WT1 mutation, Wilms8 cells harbor a mutation in the CTNNB1 gene (p.S45A), which encodes β-Catenin, a key regulator of the Wnt signaling pathway. The mutation at serine 45 disrupts the normal phosphorylation process that leads to β-Catenin degradation, causing its stabilization and accumulation in the nucleus. This results in the constitutive activation of Wnt signaling, which drives cell proliferation and contributes to the oncogenic properties of the Wilms8 cell line. The interplay between WT1 loss and aberrant Wnt signaling in Wilms8 makes it a crucial model for understanding the molecular mechanisms underlying these pathways in Wilms tumor biology. Wilms8 cells display a mesenchymal phenotype, characterized by the expression of vimentin and the absence of epithelial markers such as cytokeratin. This aligns with the stromal differentiation observed in the original tumor. The cells demonstrate a limited ability to undergo further mesenchymal differentiation, such as forming muscle-like cells under specific conditions. Proteomic analyses of Wilms8 have revealed the activation of multiple receptor tyrosine kinases (RTKs), including PDGFRβ and AXL, which are involved in key processes such as cell survival, migration, and proliferation. The activation of downstream signaling pathways, particularly the MAPK and PI3K/AKT pathways, further contributes to the aggressive characteristics of Wilms8 cells. Overall, the Wilms8 cell line serves as an essential tool for investigating the molecular basis of Wilms tumor driven by WT1 loss and aberrant Wnt signaling. Its genetic and phenotypic features make it a robust platform for studying the interaction between these critical pathways and for identifying potential therapeutic targets in Wilms tumors with a stromal component. |
|---|---|
| Organism | Human |
| Tissue | Kidney |
| Disease | Wilms tumor |
| Applications | In vitro cell culture model. Biochemical studies |
Characteristics
| Age | 8 months |
|---|---|
| Gender | Male |
| Ethnicity | Caucasian |
| Morphology | Spindle-shaped |
| Cell type | Wilms cells |
| Growth properties | Adherent |
Regulatory Data
| Citation | Wilms8 (Cytion catalog number 300416) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_A5SJ |
Biomolecular Data
| Mutational profile | WT1 mutation status: homozygous c.1168C>T, p.390x, LOH: , CTNNB1 mutation status: heterozygous TCT>GCT, p.S45A |
|---|
Handling
| Culture Medium | MSCGM kit (from Lonza) |
|---|---|
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
-
Related products
Related products