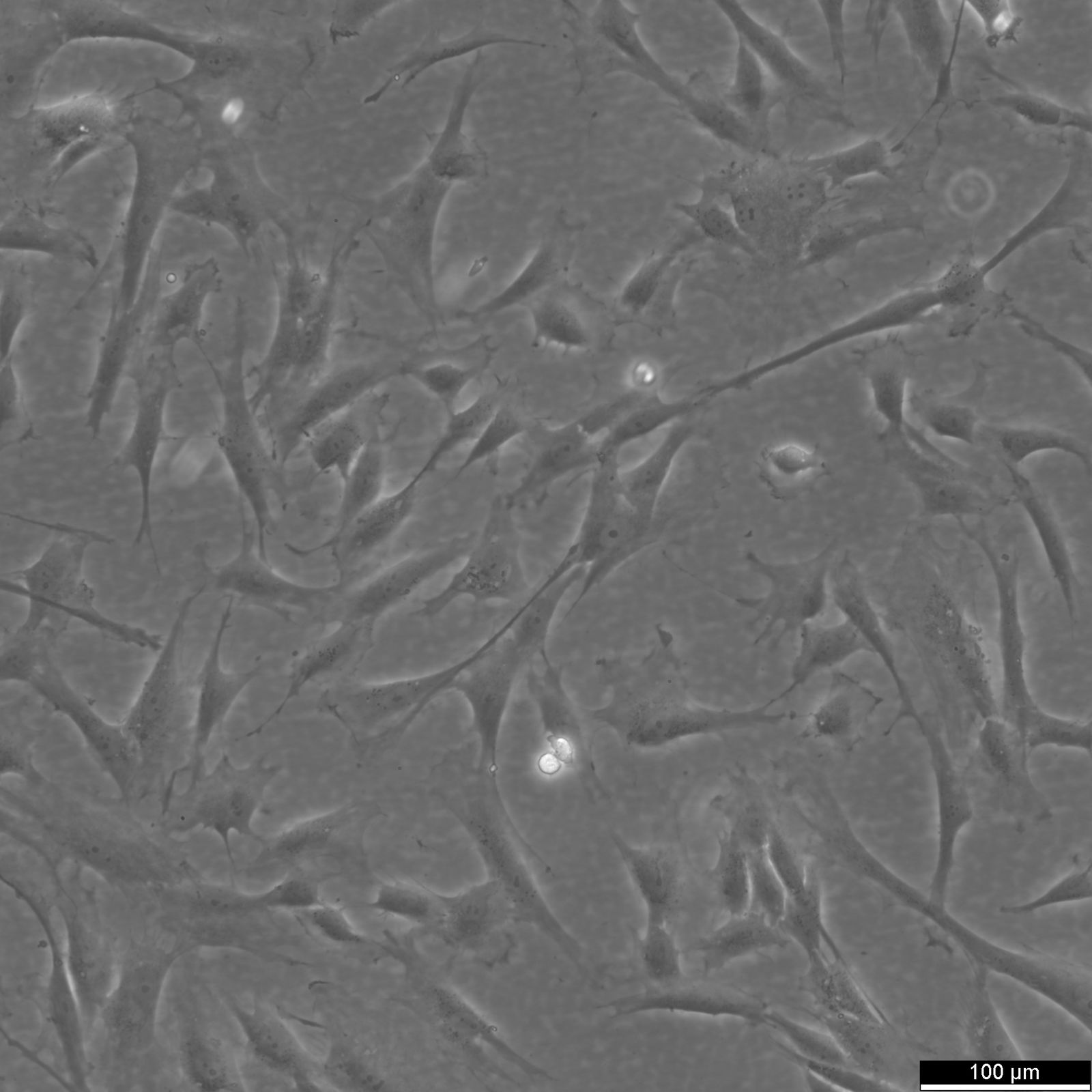
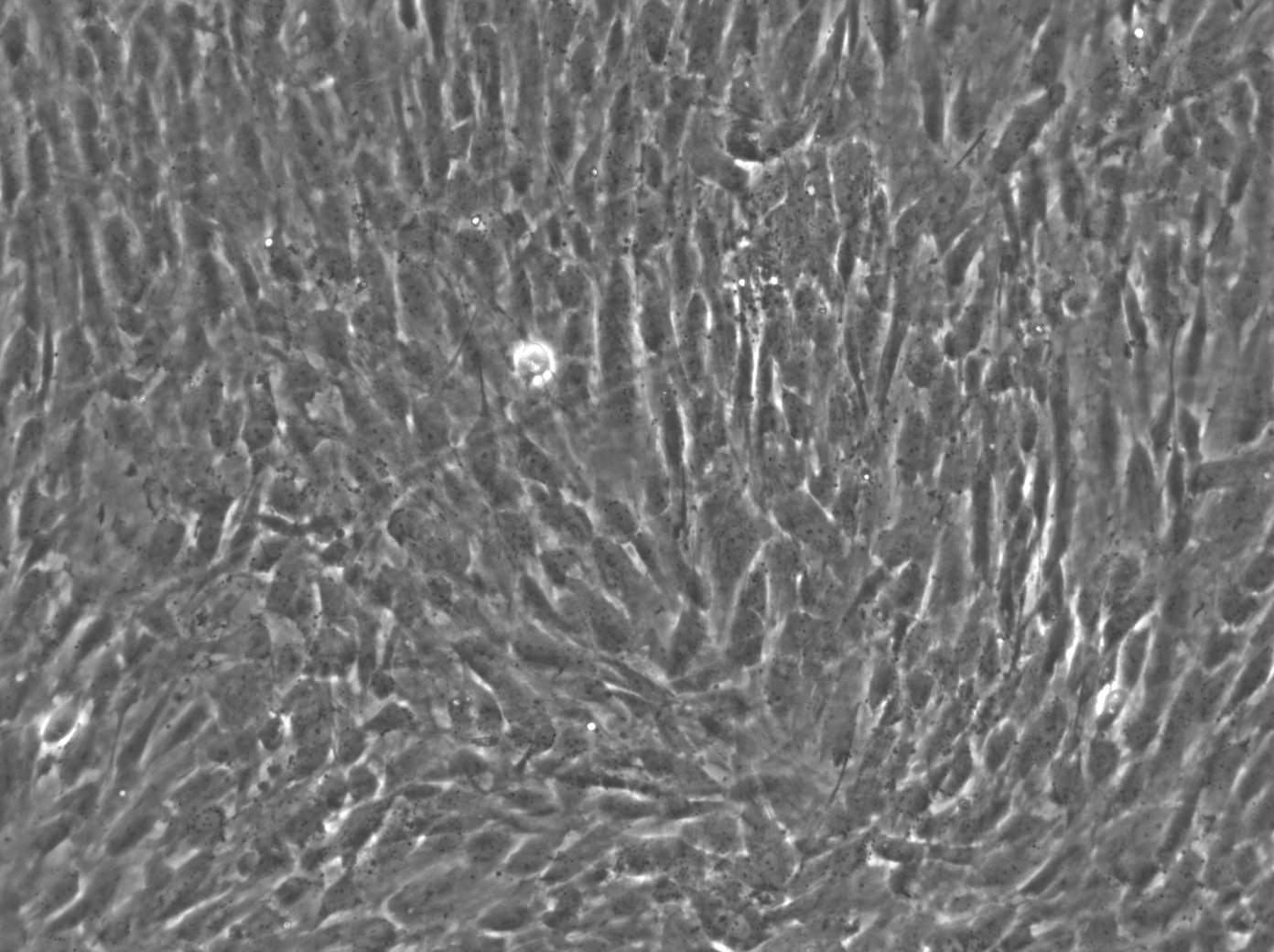
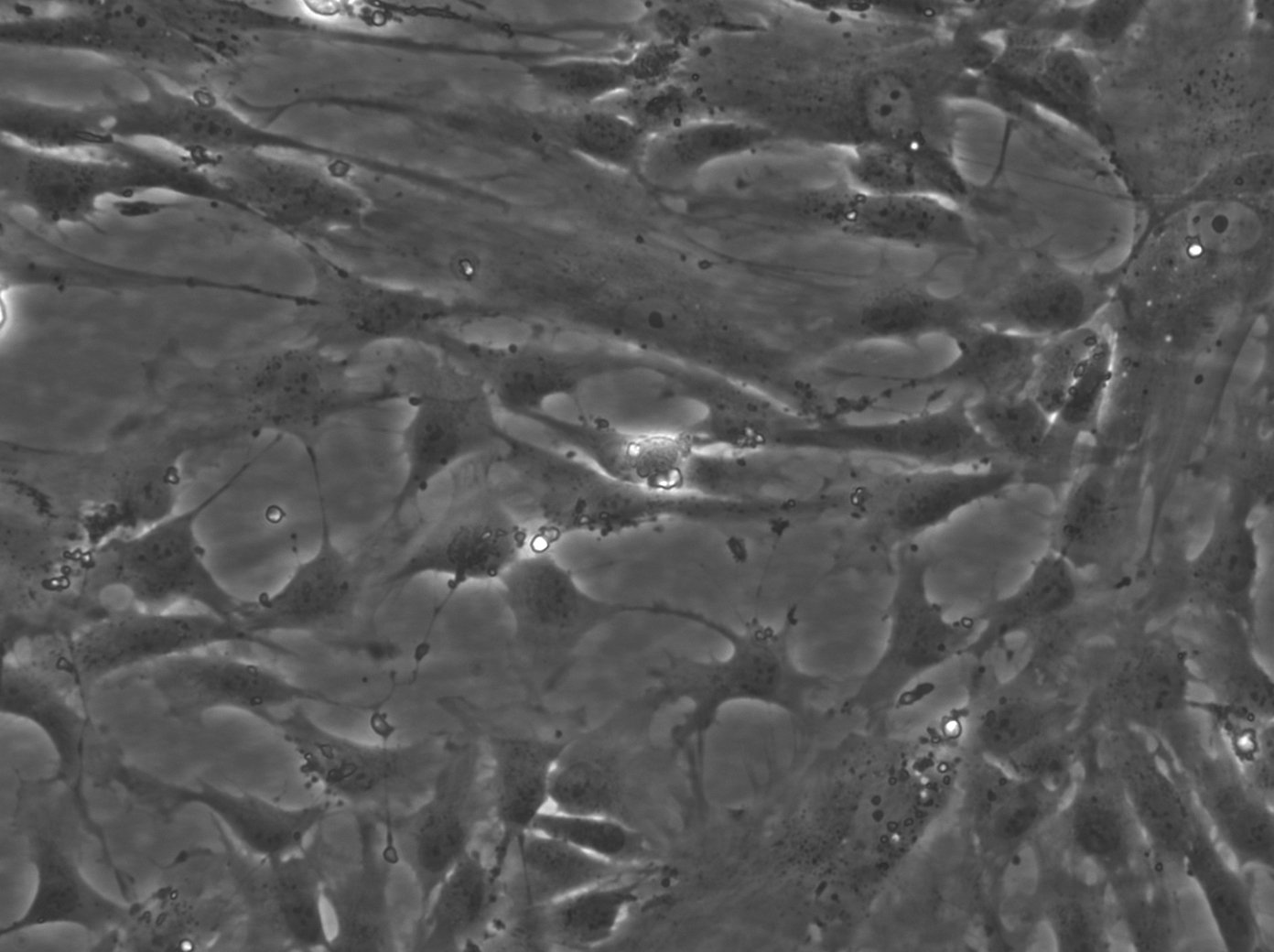
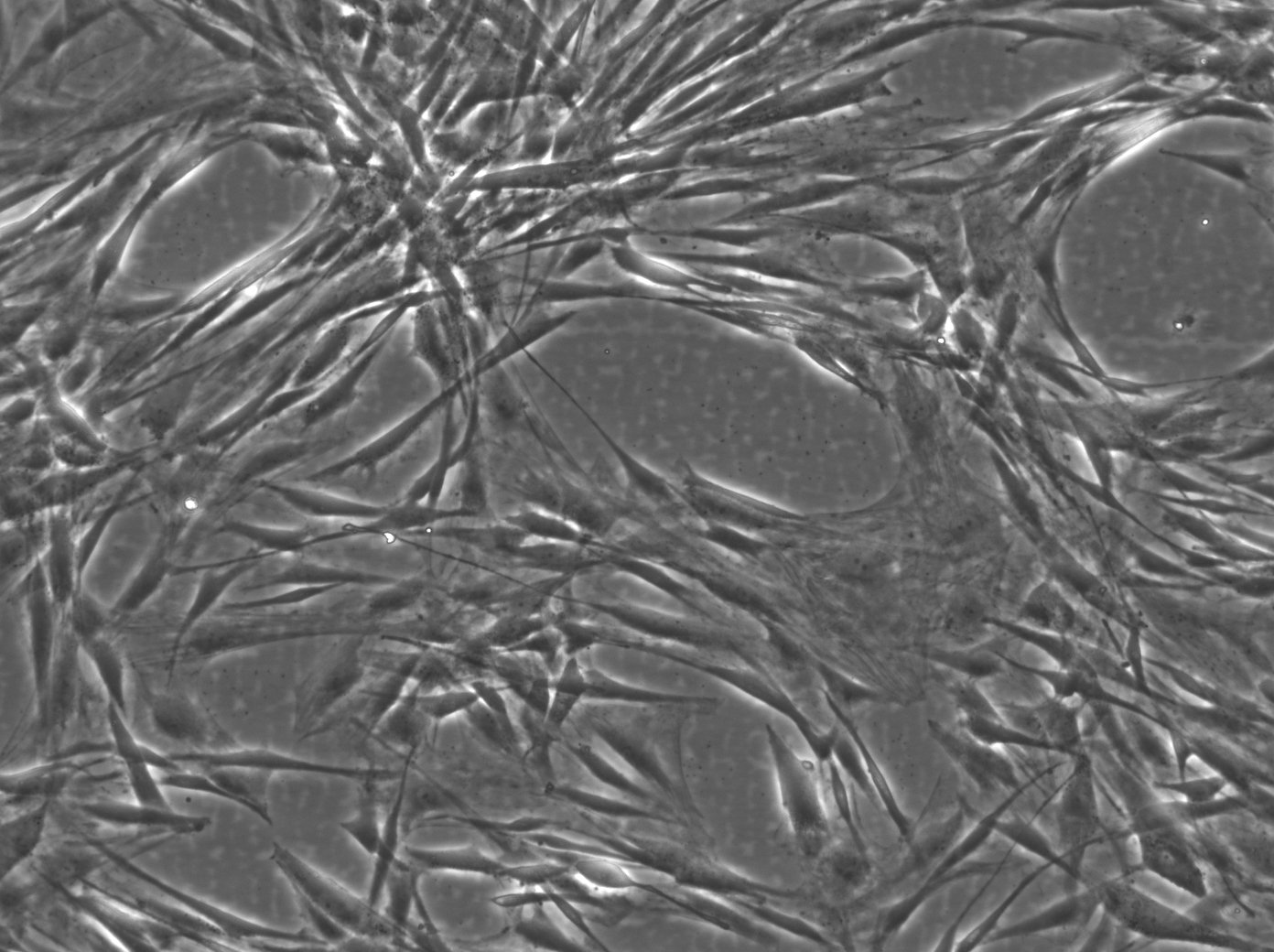
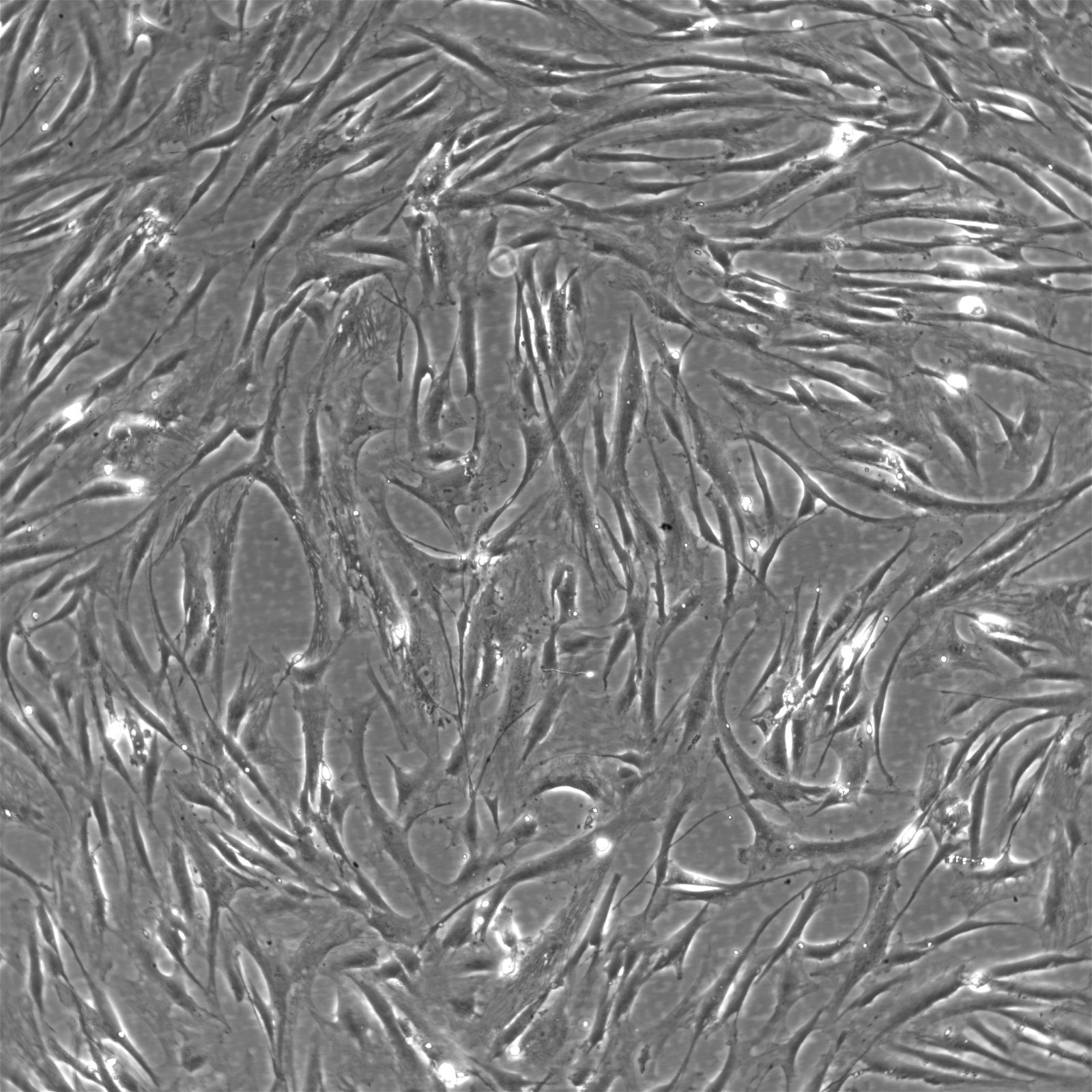
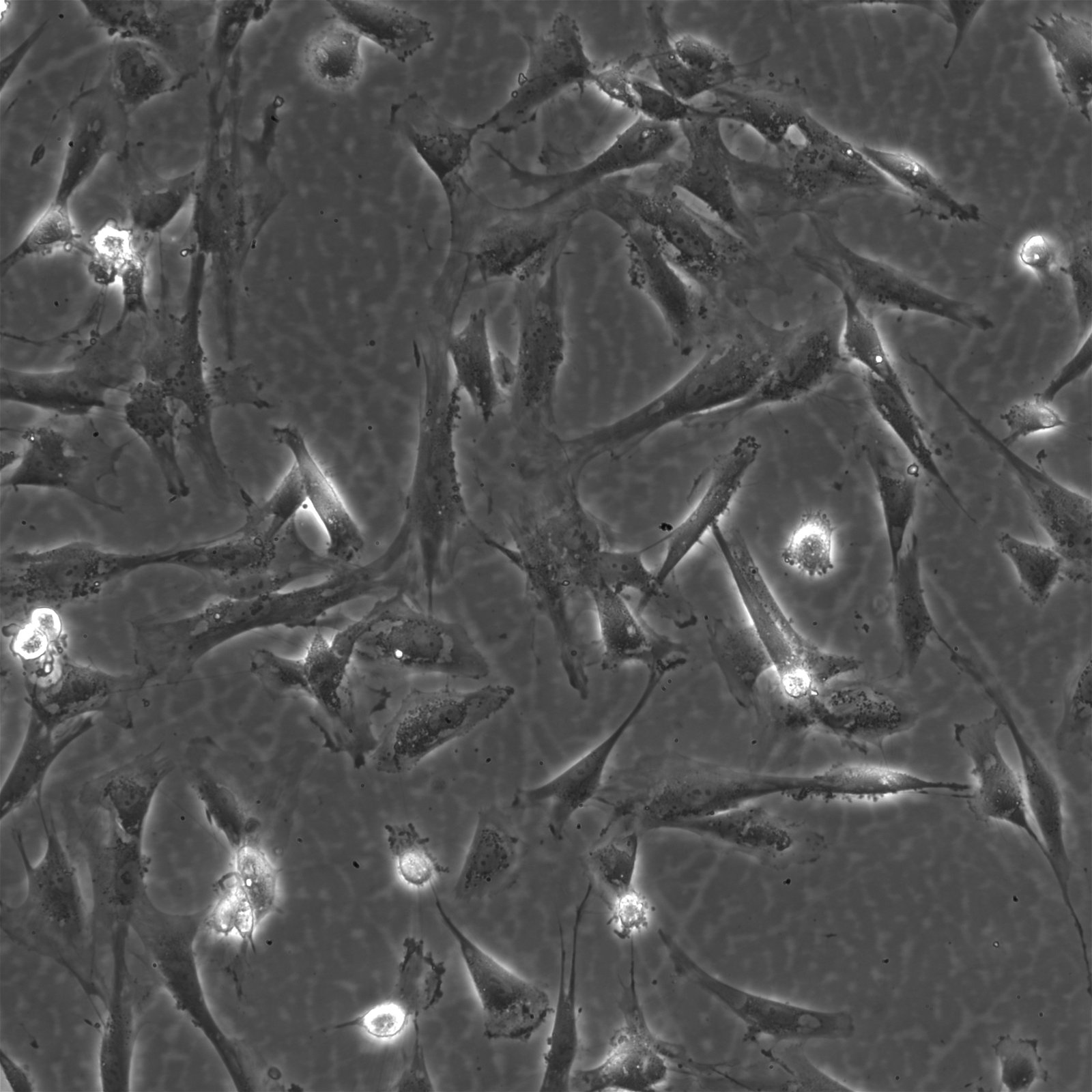
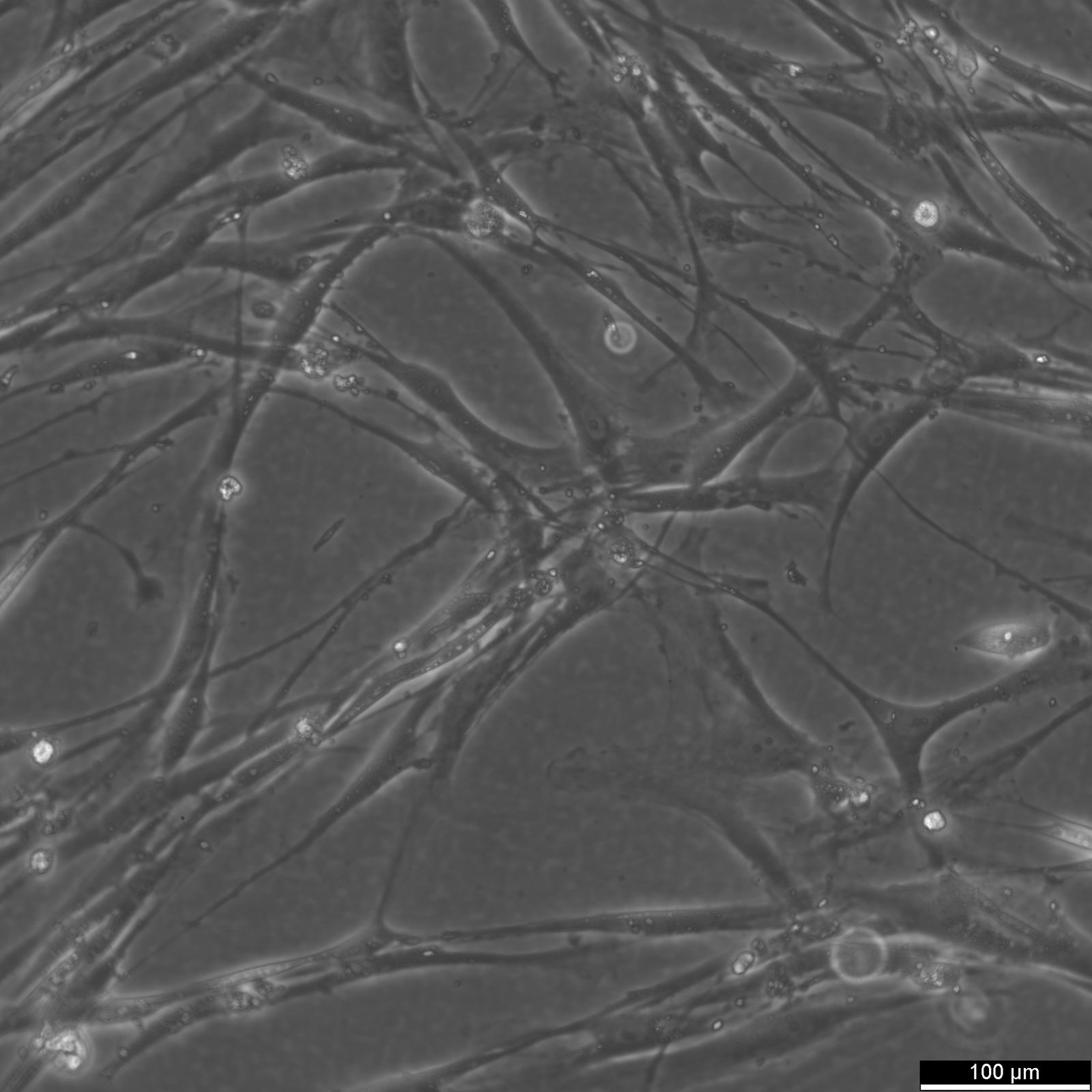
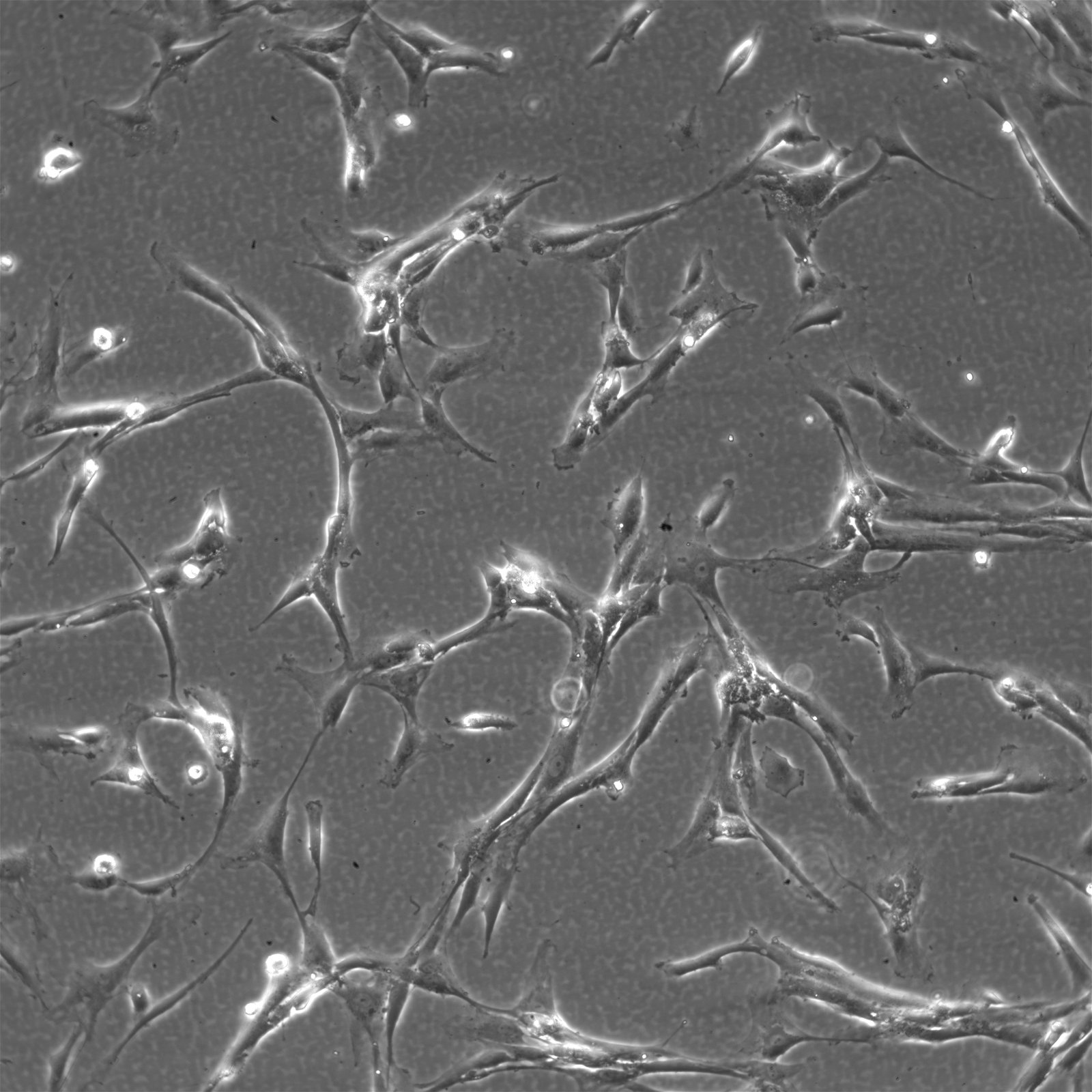
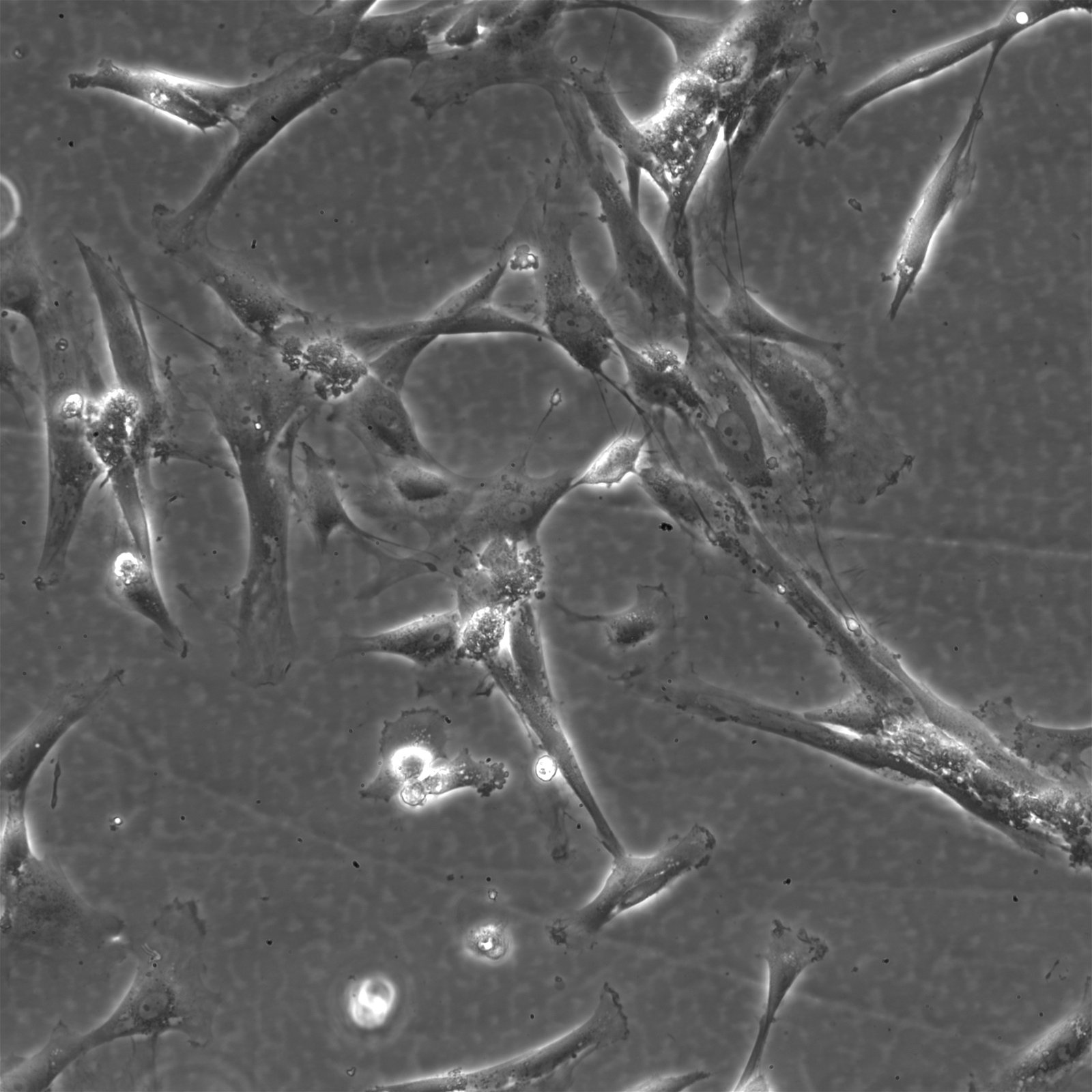
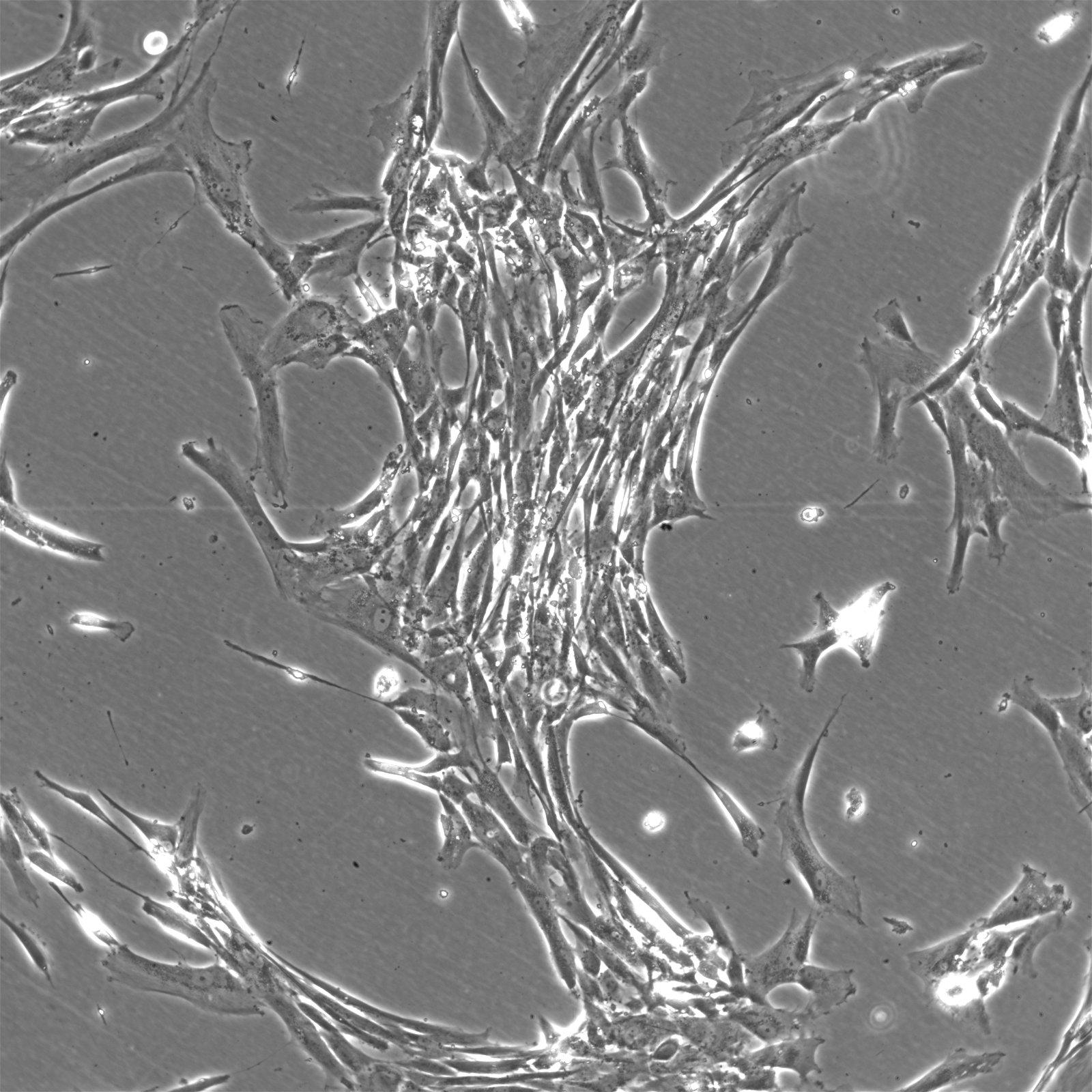
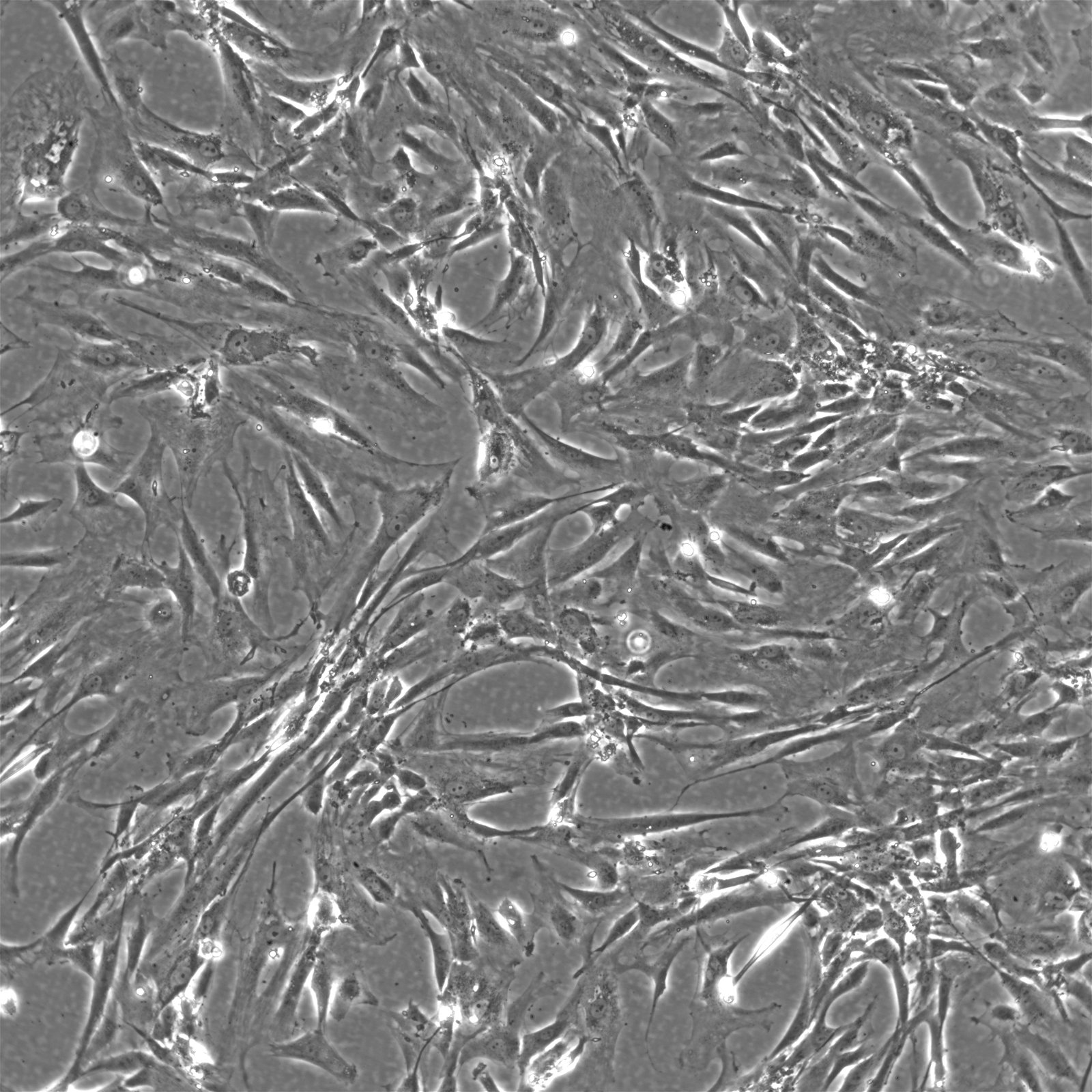
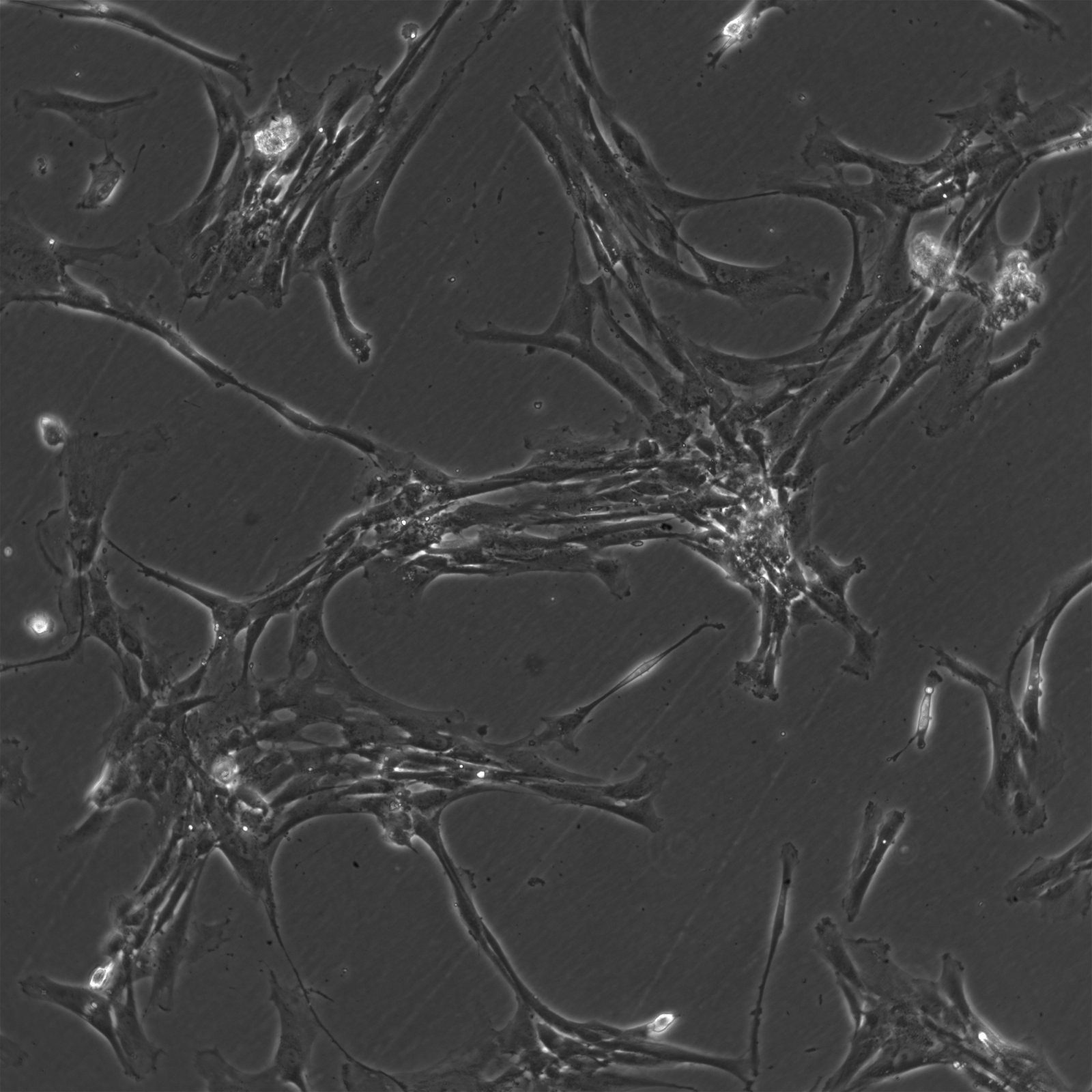
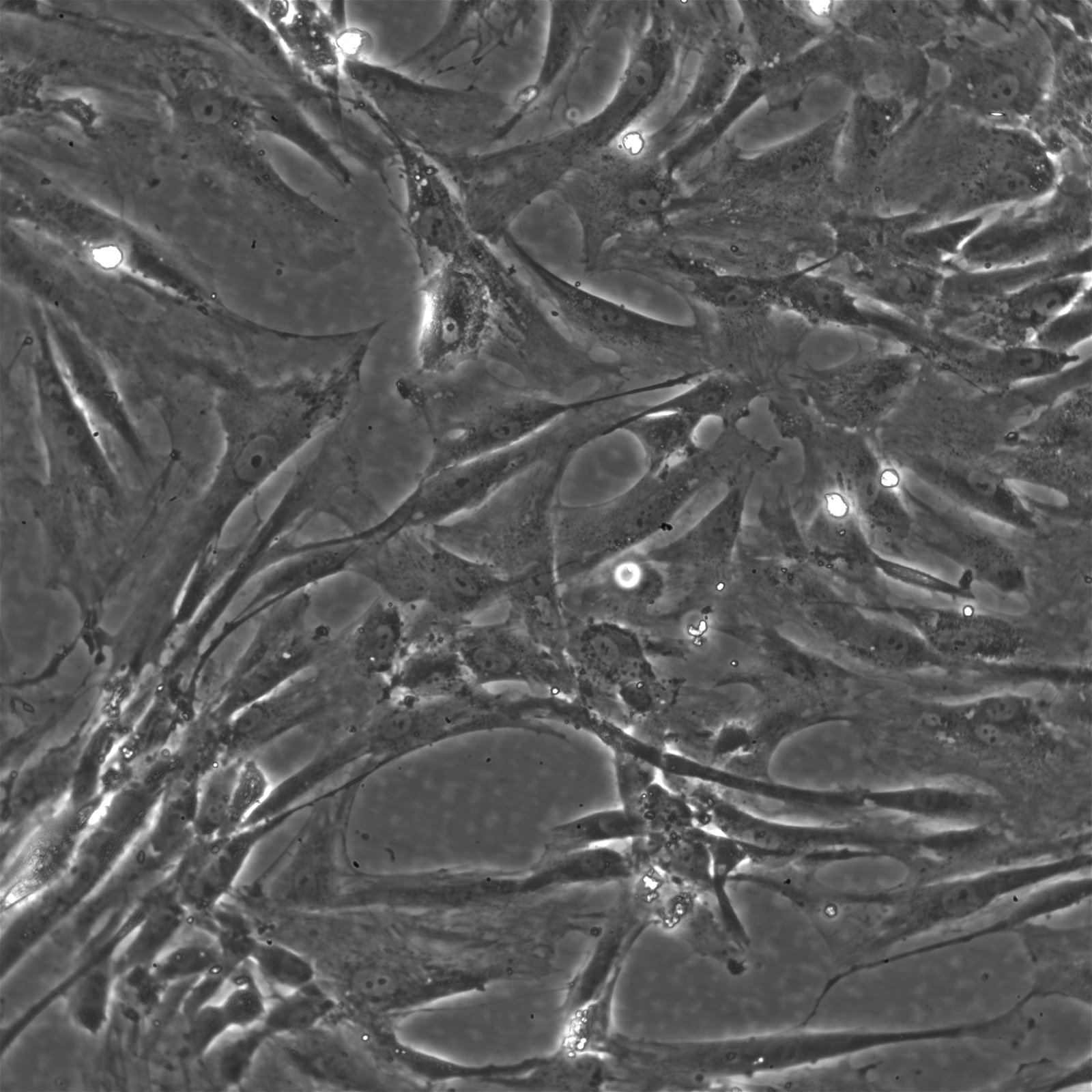
Wilms11 Cells





















USD$800.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The Wilms11 cell line was derived from a primary Wilms tumor (nephroblastoma) in a pediatric patient. Unlike many other Wilms tumor cell lines, Wilms11 is characterized by the presence of wild-type WT1, meaning it does not harbor mutations in the WT1 gene, which is typically associated with Wilms tumors exhibiting more aggressive or stromal phenotypes. However, the Wilms11 tumor exhibited significant stromal differentiation, with large areas of rhabdomyomatous differentiation, indicative of mesenchymal elements within the tumor. The presence of wild-type WT1, coupled with the tumor’s stromal differentiation, provides a unique model for understanding Wilms tumor biology in cases where WT1 mutations are absent. Genetic studies of Wilms11 have shown that this cell line carries a tumor-specific mutation in CTNNB1, the gene encoding β-Catenin, which plays a crucial role in the Wnt signaling pathway. In Wilms11, this mutation affects serine 45, a key phosphorylation site involved in β-Catenin degradation. The CTNNB1 mutation results in the stabilization of β-Catenin, leading to its accumulation and constitutive activation of the Wnt signaling pathway, a driver of cell proliferation and tumorigenesis. This makes Wilms11 an important model for studying the interplay between Wnt signaling and Wilms tumor development, particularly in cases where WT1 remains intact. Proteomic analyses of Wilms11 have revealed activation of several receptor tyrosine kinases (RTKs), including PDGFRβ and AXL, which are involved in driving tumor cell growth and survival. Downstream signaling pathways, such as the MAPK and PI3K/AKT pathways, are also activated in Wilms11 cells, contributing to their tumorigenic behavior. The ability of Wilms11 cells to undergo mesenchymal differentiation, particularly rhabdomyomatous differentiation, highlights their potential as a model for studying the mesenchymal components of Wilms tumor. Overall, Wilms11 serves as a valuable tool for investigating the molecular mechanisms that drive Wilms tumorigenesis in the absence of WT1 mutations but in the context of Wnt pathway activation. |
|---|---|
| Organism | Human |
| Tissue | Kidney |
| Disease | Wilms tumor |
| Applications | In vitro cell culture model. Biochemical studies |
Characteristics
| Age | 22 months |
|---|---|
| Gender | Male |
| Ethnicity | Caucasian |
| Morphology | Spindle-shaped |
| Cell type | Wilms cells |
| Growth properties | Adherent |
Regulatory Data
| Citation | Wilms11 (Cytion catalog number 300420) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_A5SM |
Biomolecular Data
| Mutational profile | WT1 mutation status: homozygous WT1 c.901c>T, p.R301x. LOH: . CTNNB1 mutation status: wild type |
|---|
Handling
| Culture Medium | MSCGM kit (from Lonza) |
|---|---|
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300420-221 | Certificate of Analysis | 23. May. 2025 | 300420 |
| 300420-240226 | Certificate of Analysis | 25. Mar. 2026 | 300420 |
-
Related products
Related products