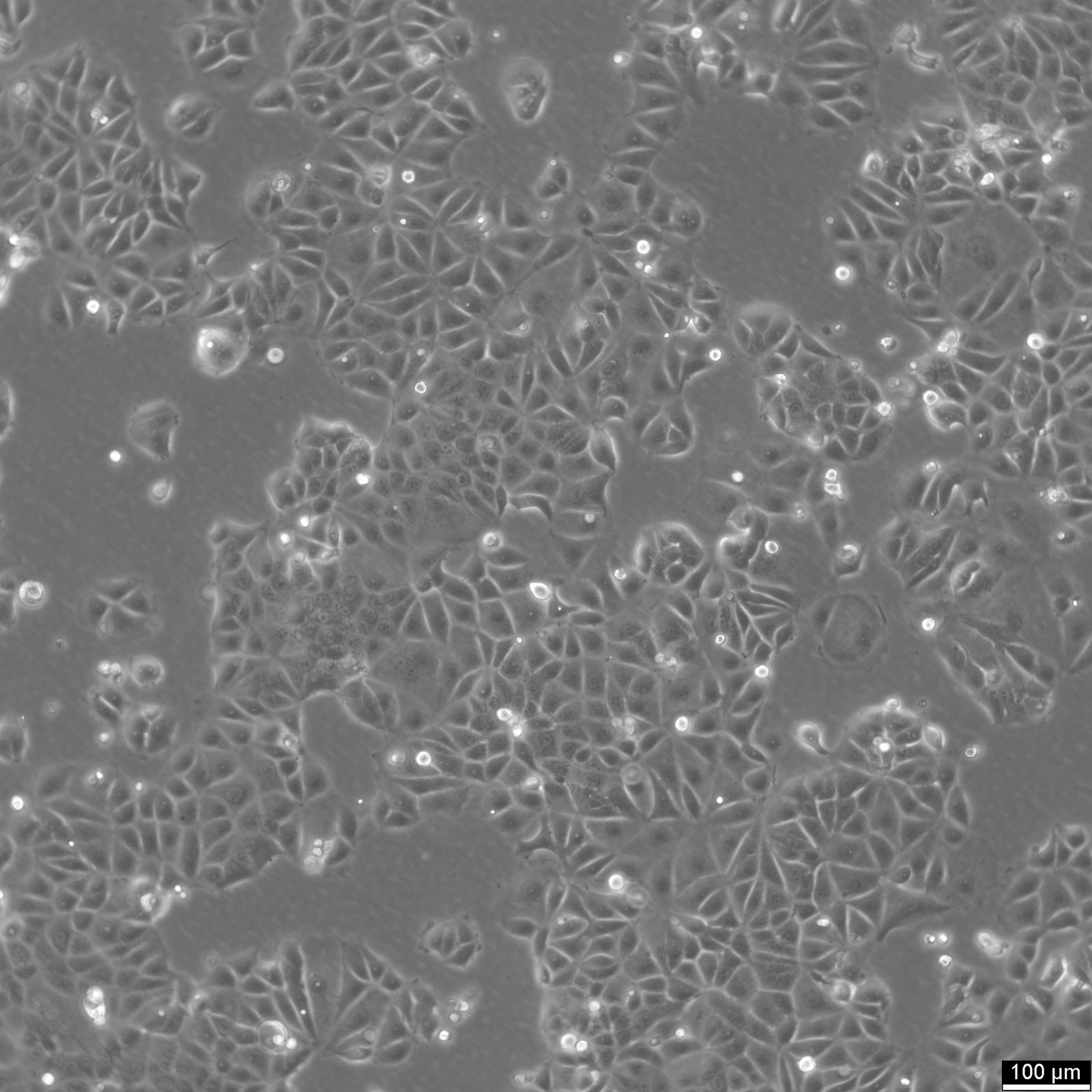
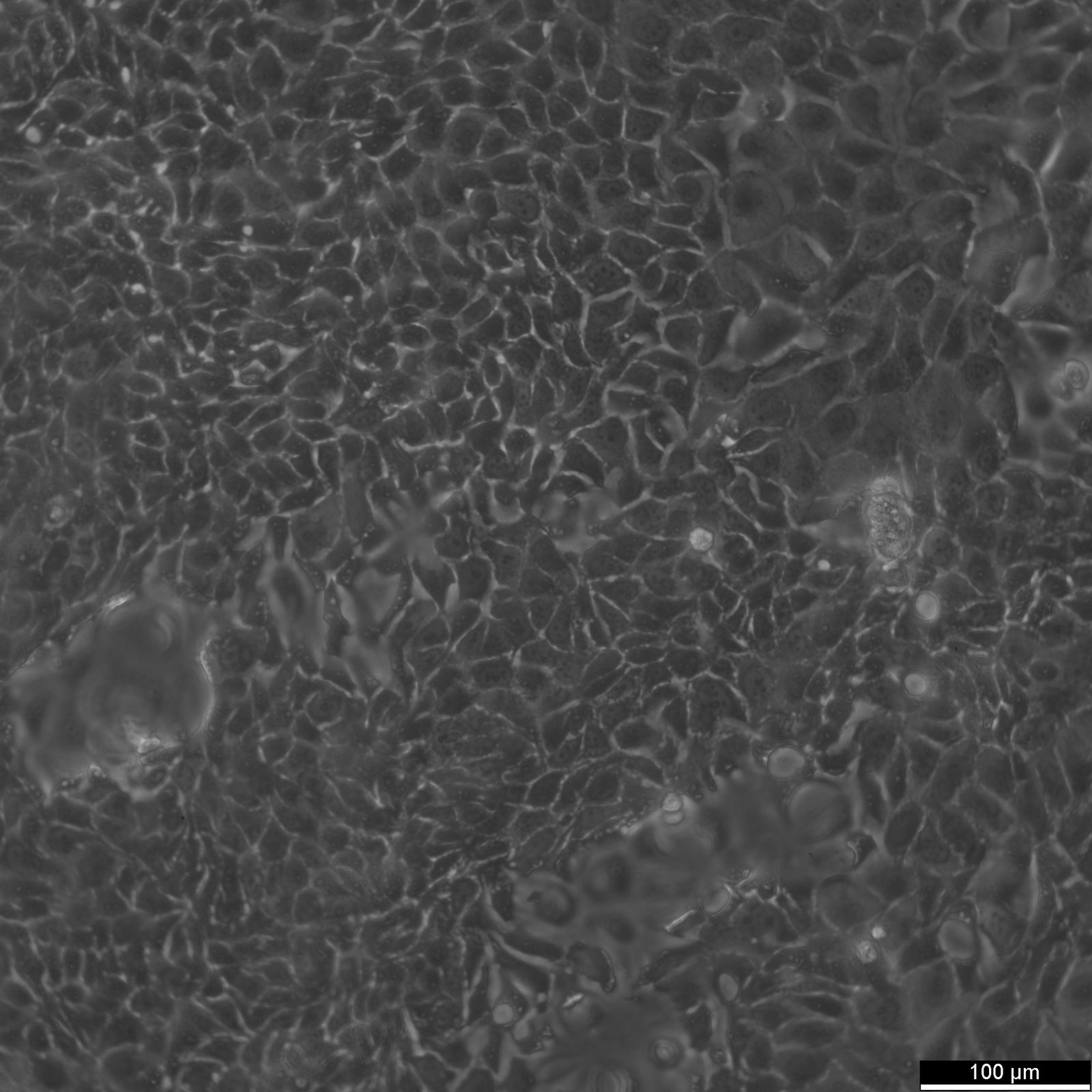
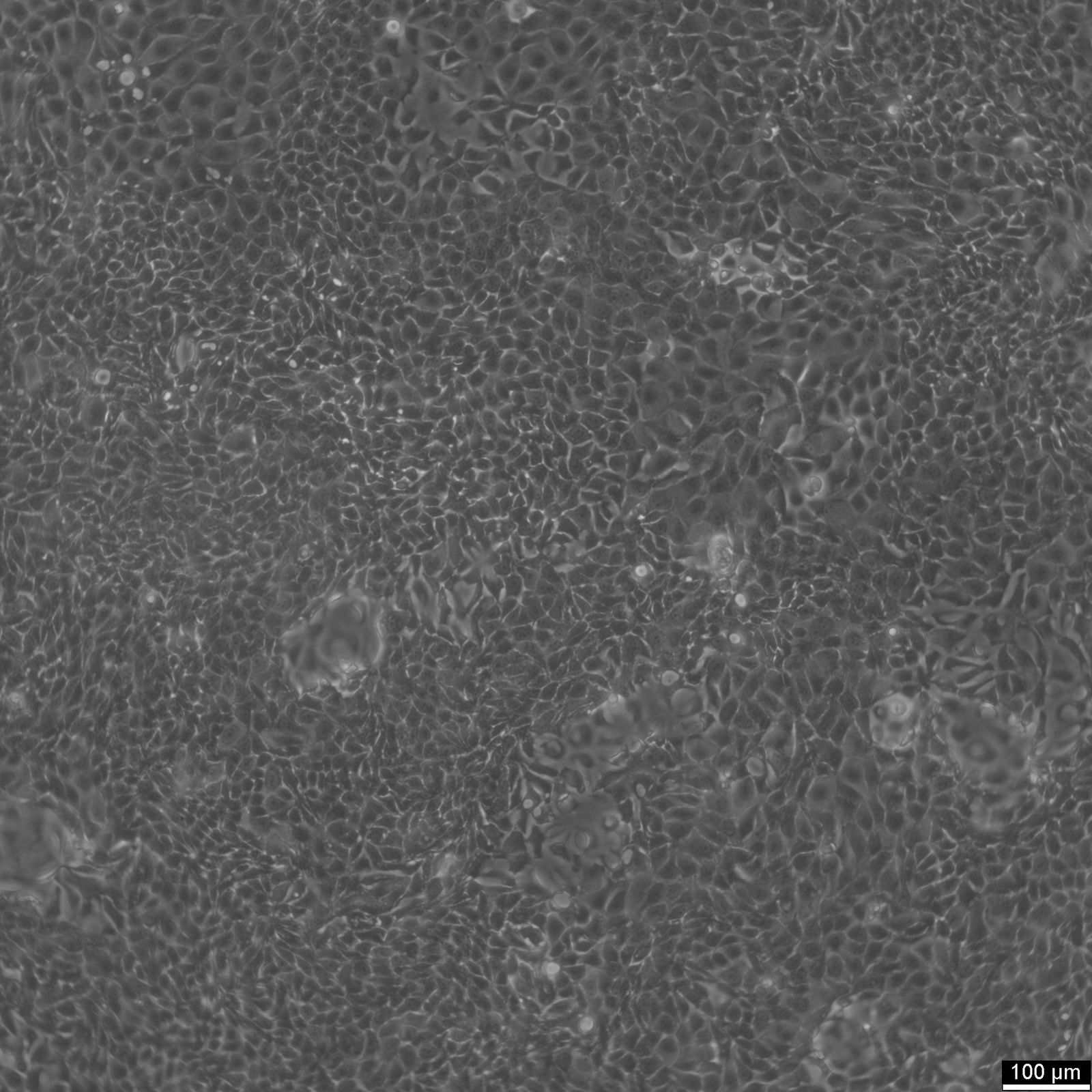
MCF-7 Cells









USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
Essential facts about the MCF7 breast cancer cell line
| Description | MCF7 cells, a widely used research model in human breast cancer research, are utilized extensively as an in vitro model for hormone-dependent breast cancer. Originating from the breast tissue of a 69-year-old white female with metastatic adenocarcinoma, MCF7 cells are a widely used in vitro model for hormone-dependent breast cancer, reflecting the Luminal A subtype. This subtype is characterized by a lower grade and better prognosis compared to more aggressive forms of breast cancer. In the realm of breast cancer research, MCF 7 cells are instrumental in evaluating the efficacy of breast cancer drugs and understanding the dynamics of breast cancer stem cells. They are central to cancer research, serving as a comparative model against more aggressive cell lines like MDA-MB-231. The investigation of therapeutic agents, such as tamoxifen and doxorubicin, is critical in drug discovery efforts targeting hormone-dependent breast cancers and gaining insights into the mechanisms of action and resistance. Similarly, the role of estradiol in modulating the growth and characteristics of these cells is a subject of significant interest, given its relevance to hormone-responsive breast cancers. Research employing the MCF7 breast cancer cell line often delves into the cellular processes of cytotoxicity and apoptosis, especially in response to cancer agents like curcumin, known for its potential in cancer prevention. The study of immune responses, including the action of tumor necrosis factor alpha (TNF alpha) and the impact of bacterial antigens, further enriches our understanding of the tumor microenvironment and potential therapeutic targets. MCF7 cells are meticulously studied in both 2D cell culture and 3D cell culture systems, including spheroid culture, to mimic tumor microenvironments more closely. These methodologies enable a more profound exploration of cell spheroid growth and the behavior of cancer stem cells within microtissues in scaffold-based systems. The MCF7 cell line, with its epithelial cell characteristics and resemblance to human adenocarcinoma cells, is a cornerstone of cancer research. It facilitates not only the exploration of breast cancer drugs and their mechanisms but also the broader implications for cancer treatment, including the potential role of mesenchymal stem cells and the efficacy of targeted therapies in vivo studies. |
|---|---|
| Organism | Human |
| Tissue | Breast |
| Disease | Adenocarcinoma |
| Metastatic site | Pleural effusion |
| Synonyms | MCF 7, MCF.7, MCF7, Michigan Cancer Foundation-7, ssMCF-7, ssMCF7, MCF7/WT, MCF7-CTRL, IBMF-7 |
Features
| Age | 69 years |
|---|---|
| Gender | Female |
| Ethnicity | Caucasian |
| Morphology | Epithelial-like |
| Growth properties | Monolayer, adherent |
Documentation
| Citation | MCF-7 (Cytion catalog number 300273) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0031 |
Genetic profile
| Receptors expressed | The cells express the wildtype and variant estrogen receptors as well as progesterone receptor. |
|---|---|
| Protein expression | P53 negative, pGP9.5 negative, CEA positive |
| Isoenzymes | PGM3, 1, PGM1, 1-2, ES-D, 1-2, AK-1, 1, GLO-1, 1-2, G6PD, B, |
| Oncogenes | Wnt7h +, Tx-4 |
| Tumorigenic | Yes, in nude mice |
| Products | Insulin-like growth factor binding proteins (IGFBP) BP-2, BP-4, BP-5 |
| Mutational profile | TP53 wt |
| Karyotype | The stemline chromosome numbers ranged from hypertriploidy to hypotetraploidy, with the 2S component occurring at 1%. There were 29 to 34 marker chromosomes per S metaphase, 24 to 28 markers occurred in at least 30% of cells, and generally one large submetacentric (M1) and 3 large subtelocentric (M2, M3, and M4) markers were recognizable in over 80% of metaphases. No DM were detected. Chromosome 20 was nullisomic and x was disomic. Phenotype Frequency Product: 0.0154 |
MCF7 cell culturing methods
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Doubling time | 24 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 3 x 104 cells/cm2 |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | Allow the cells to rest for 48 hours past thawing |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality control
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300273-140125 | Certificate of Analysis | 23. May. 2025 | 300273 |
| 300273-200125 | Certificate of Analysis | 23. May. 2025 | 300273 |
| 300273-250823 | Certificate of Analysis | 23. May. 2025 | 300273 |
-
Related products
Related products


