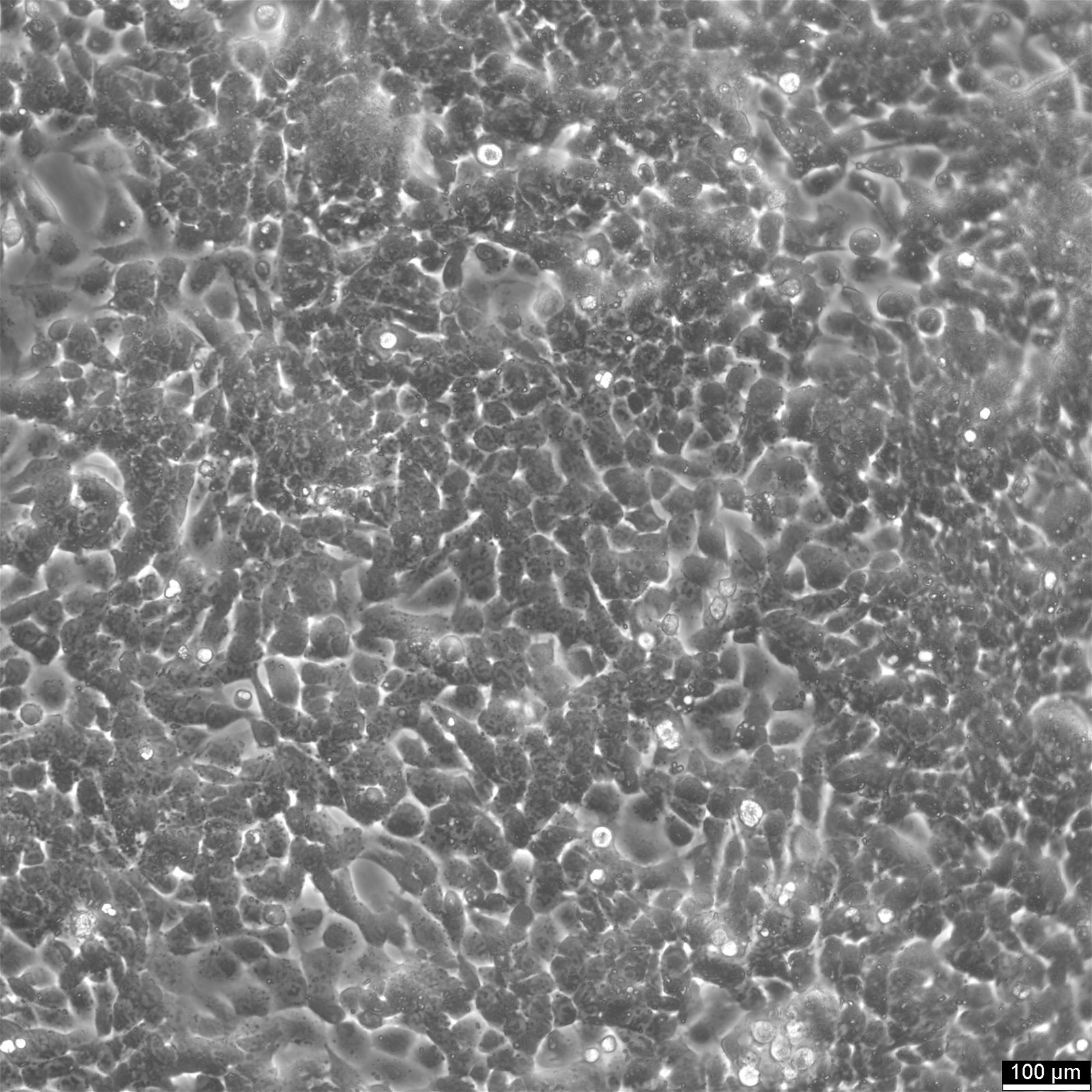
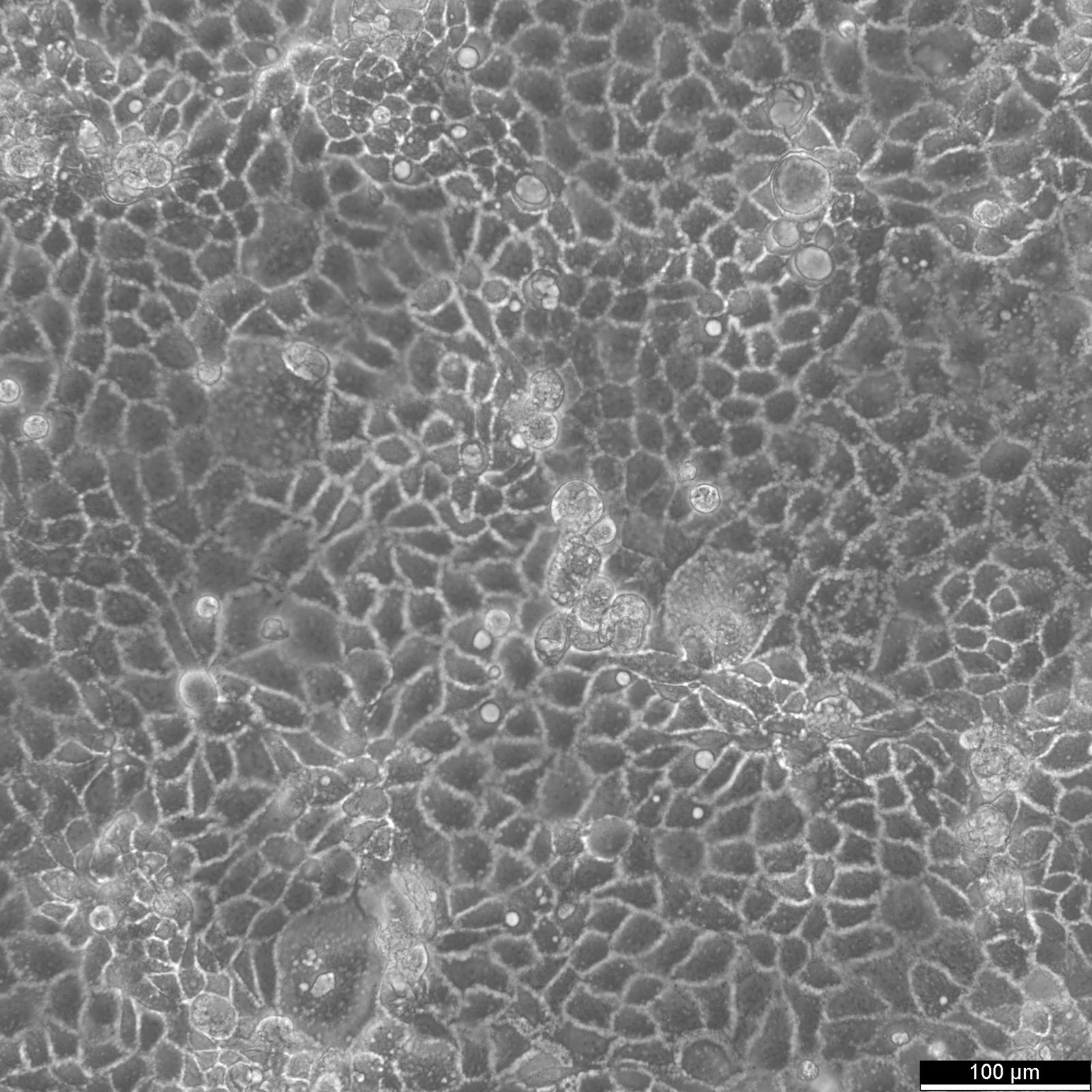
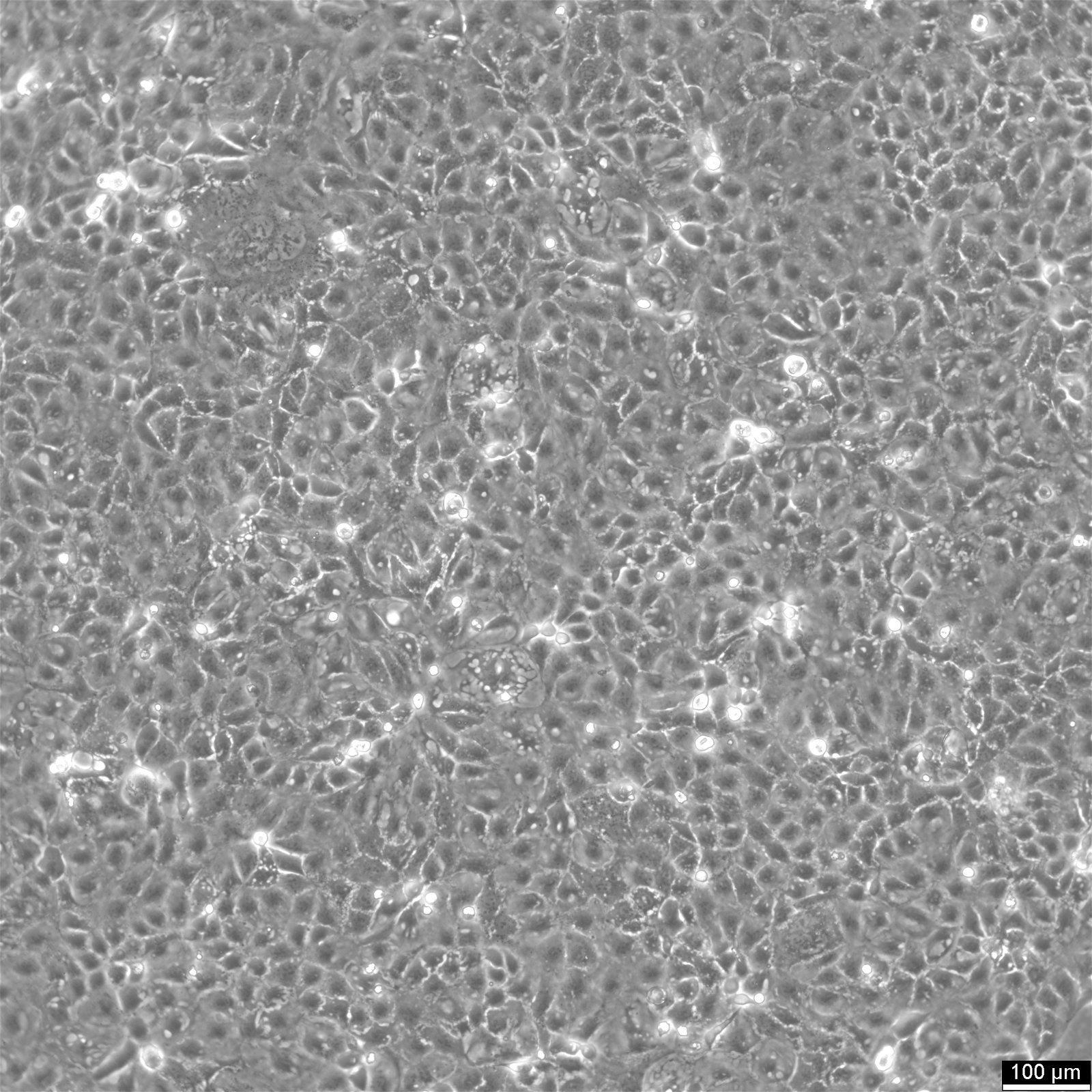
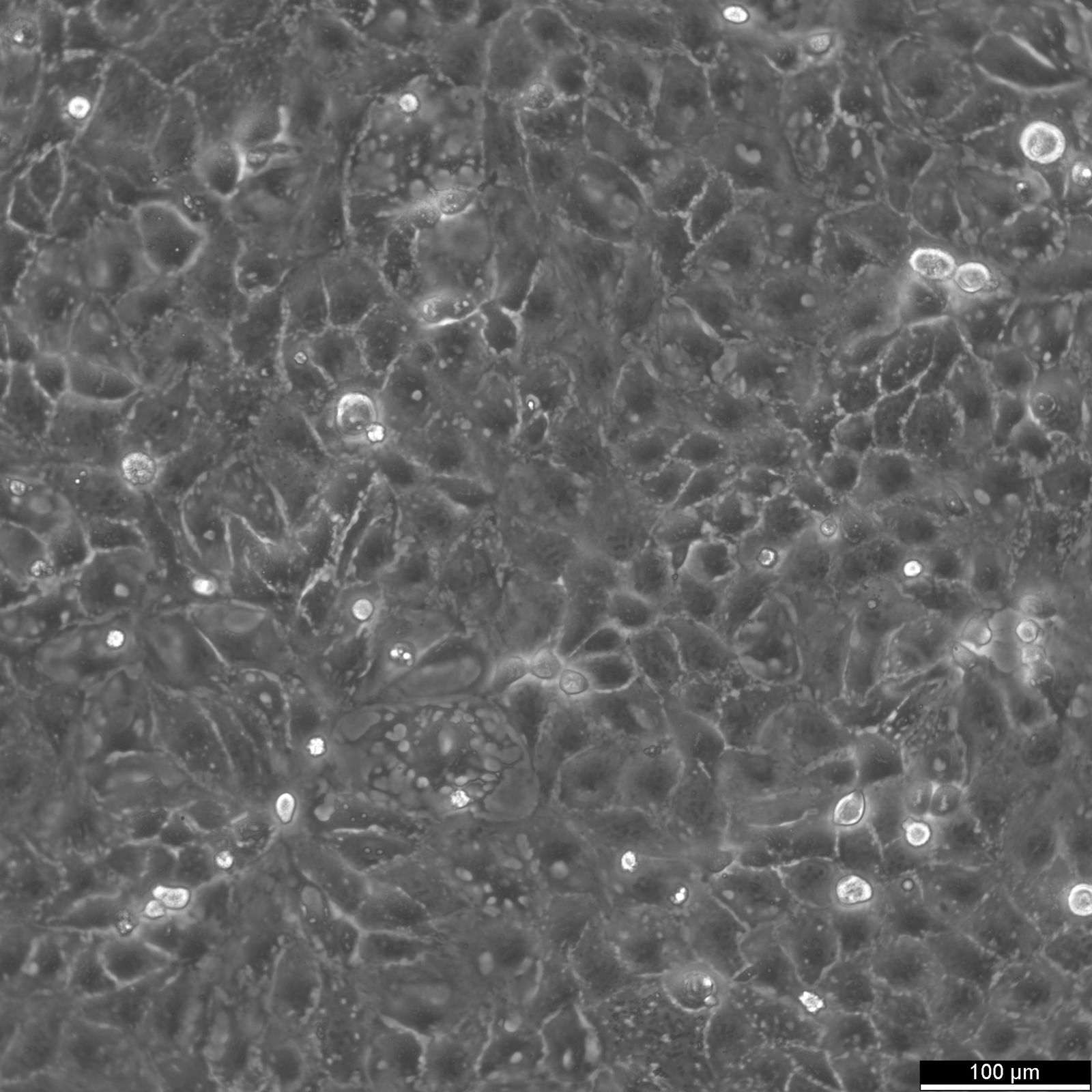
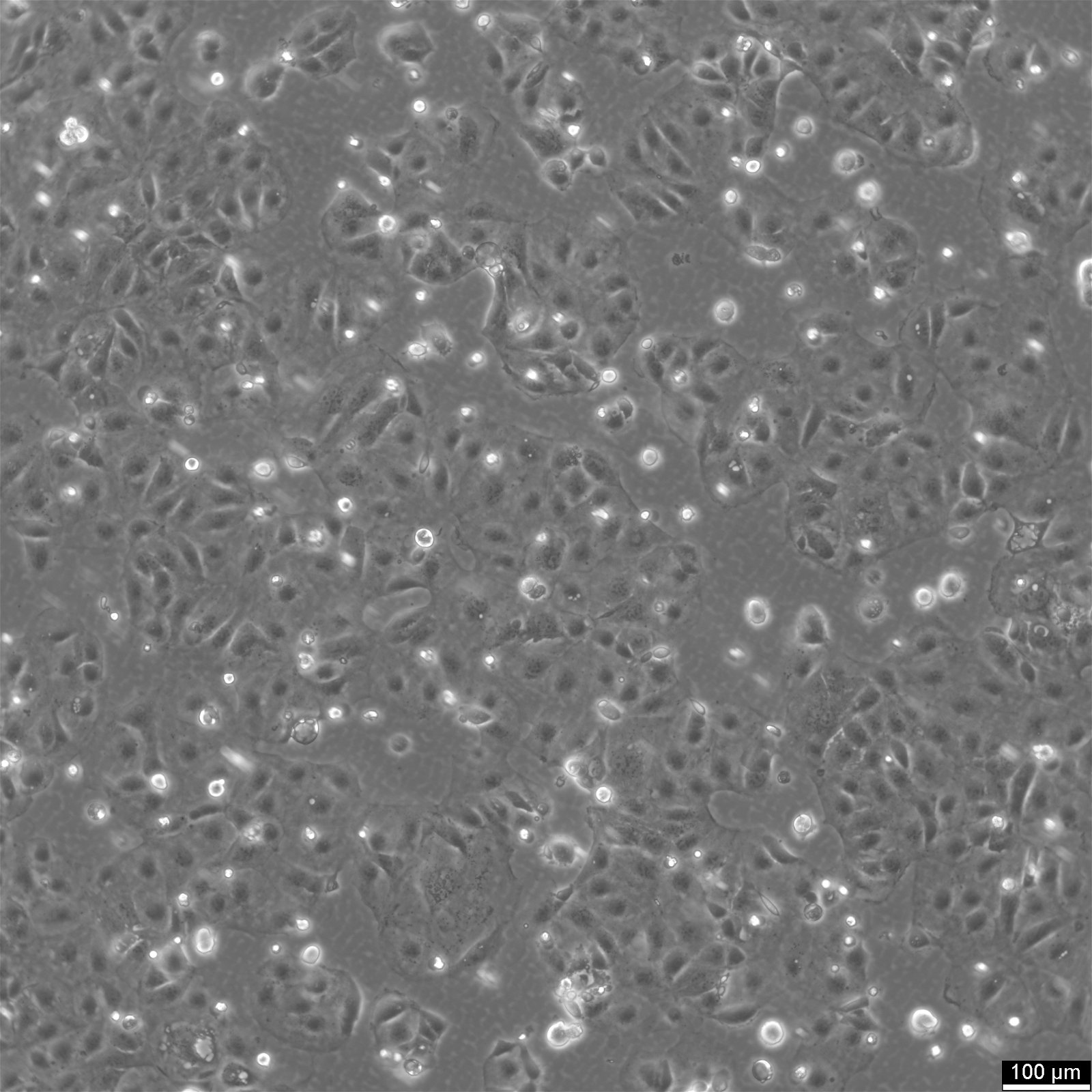
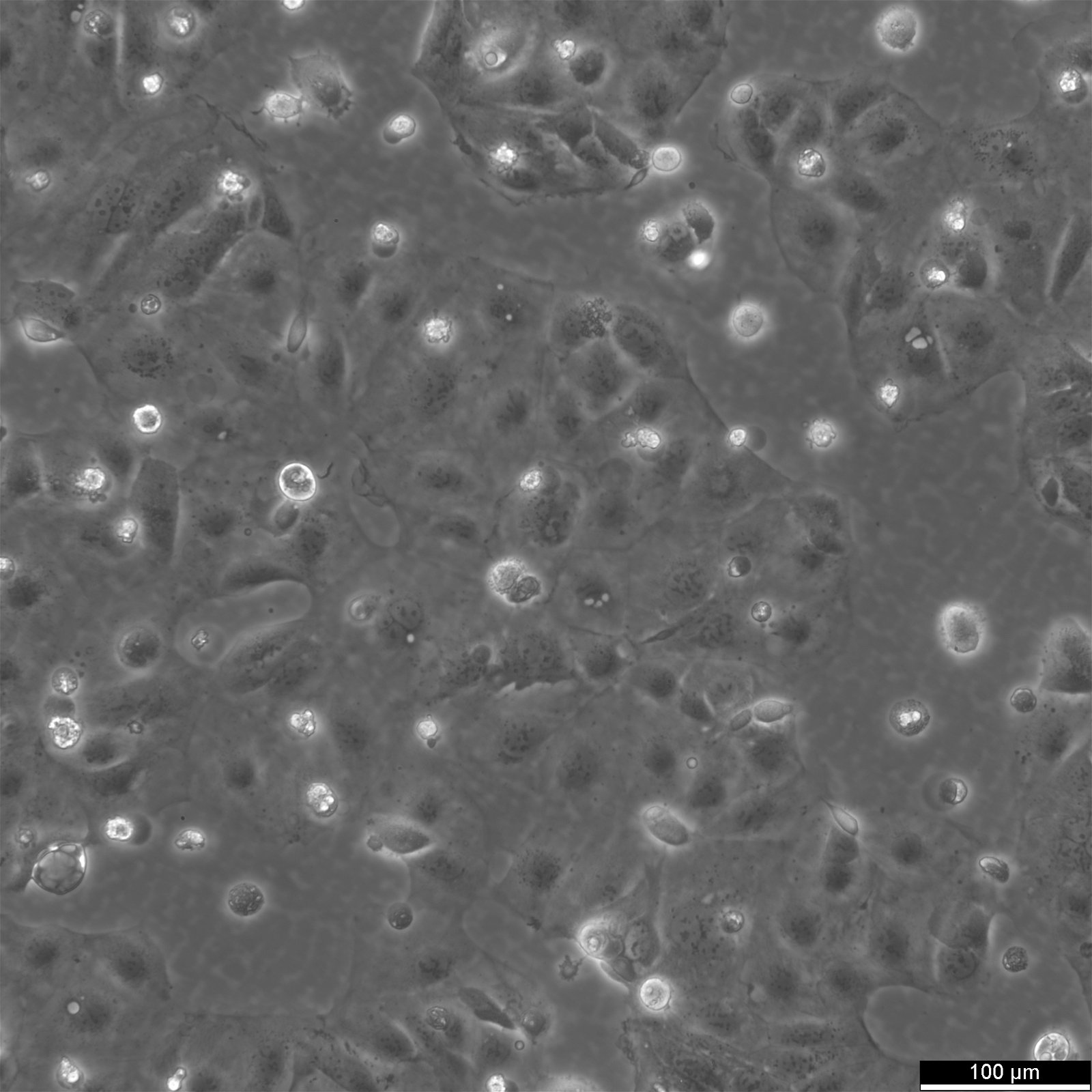
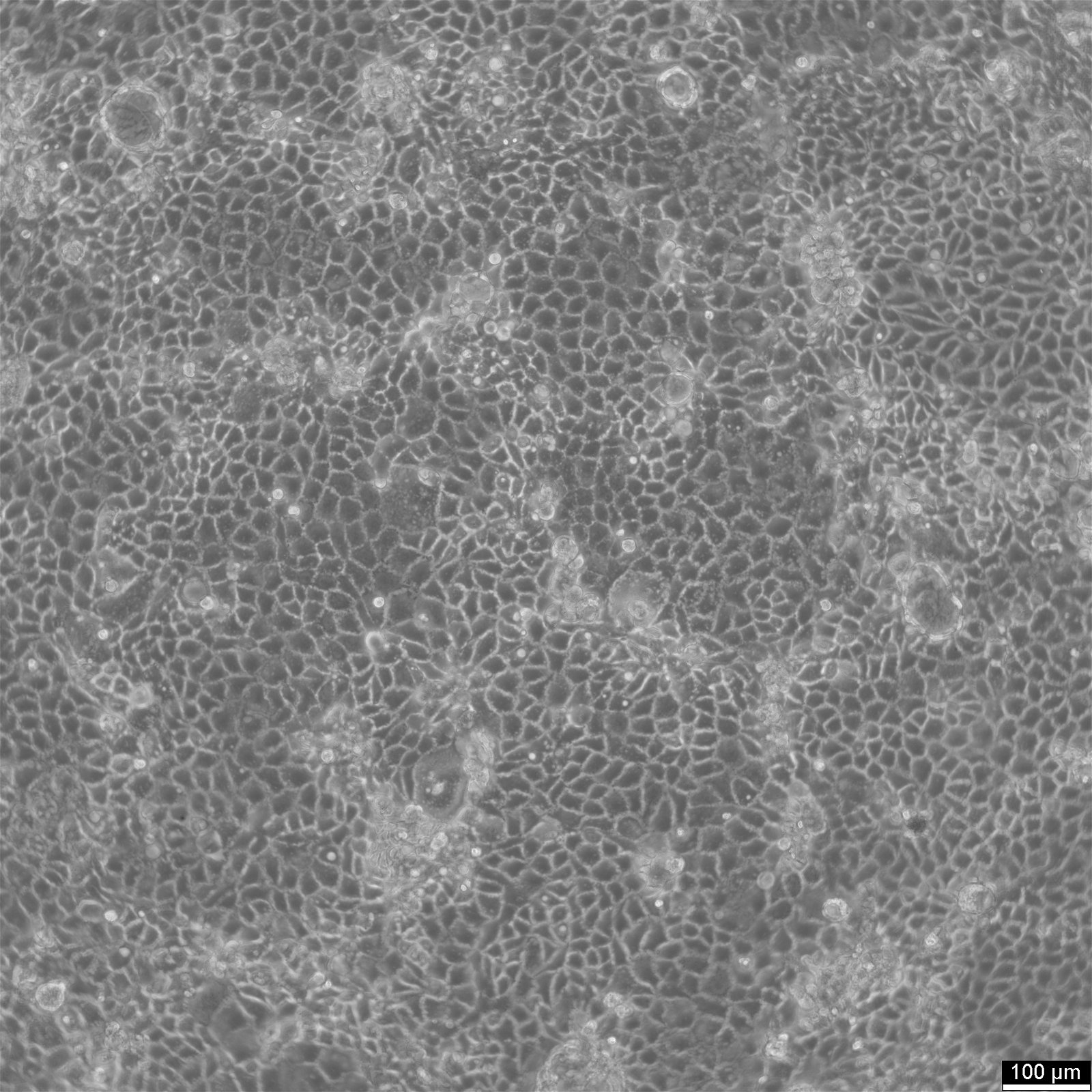
Caco-2 Cells






















USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
Introduction to Caco-2 cells
| Description | Caco-2 cells serve as an advanced in vitro model for the human intestinal barrier, primarily due to their differentiation into a cell monolayer that closely resembles the enterocytes lining the small intestine. When culturing the Caco2 cell line on tissue culture filter inserts with polycarbonate filters, Caco-2 cells undergo spontaneous differentiation. The differentiation of Caco2 cells results in the expression of specialized cell types, complete with microvilli, enzymes, and transporters, paralleling the complex features and mechanisms found in an in vivo situation. In the context of intestinal absorption studies models, Caco-2 cells, which were derived from a human colorectal adenocarcinoma patient, are instrumental due to their ability to develop high TEER values, signifying intact tight junctions and epithelial barrier function. These properties are crucial for assays like the cholesterol efflux assay and investigations into cellular transport, including the movement of lipid nanoparticles and the detection of protein interactions. Caco-2 cells are pivotal for intestinal absorption studies, providing a reliable in vitro approximation of the intestinal epithelium. Mimicking intestinal enterocytes, these cells facilitate analyses of oral drug absorption by simulating the intestinal barrier. Researchers utilize Caco-2 cells to predict how substances traverse the intestinal mucosa, which is essential for the pharmacokinetic profiling of oral medications. Furthermore, they are a key tool in investigating intestinal cholesterol uptake, homeostasis and transport, which are vital processes for understanding lipid metabolism and associated diseases. Caco-2 cells remain a cornerstone in colon carcinoma and toxicology research, not only for their relevance to human gastrointestinal studies but also for their role in providing detailed insights into the biliary pathway, the metabolism of xenobiotics within the colon, cancer and toxicology research. |
|---|---|
| Organism | Human |
| Tissue | Colon |
| Disease | Adenocarcinoma |
| Applications | Model of the GI (gastrointestinal) tract, measurement of the Trans-Epithelial/Endothelial Eletrical Resistance (TEER). Caco-2 cells develop high TEER values of up to 2000 cm2 (as measured by CLS using the CellZscope, nanoAnalytics, Münster, Germany). |
| Synonyms | CaCo-2, CACO-2, Caco 2, CACO 2, CACO2, CaCo2, CaCO2, Caco2, Caco-II |
Details
| Age | 72 years |
|---|---|
| Gender | Male |
| Ethnicity | Caucasian |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Documentation
| Citation | CaCo-2 (Cytion catalog number 300137) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0025 |
Genetic makeup of the Caco-2 cell line
| Receptors expressed | Heat stable enterotoxin (Sta, E. coli), epidermal growth factor (EGF), retinoic acid binding protein I and retinol binding protein II, keratin positive. |
|---|---|
| Antigen expression | Blood Type O, Rh+, HLA class II negative |
| Isoenzymes | Me-2, 1, PGM3, 1, PGM1, 1, ES-D, 1, AK-1, 1, GLO-1, 1, G6PD, B. |
| Tumorigenic | Yes, in nude mice. Form moderately well differentiated adenocarcinomas consistent with colonic primary (grade II) |
| Virus resistance | Human immunodeficiency virus (HIV, LAV) |
| Ploidy status | (P14), hypertetraploid |
| MSI-status | Stable (MSS) |
Handling
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Doubling time | 60 to 70 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 will result in a 90% confluent monolayer in about 4 days |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality control
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300137-220424 | Certificate of Analysis | 23. May. 2025 | 300137 |
| 300137-170625 | Certificate of Analysis | 18. Aug. 2025 | 300137 |
-
Related products
Related products