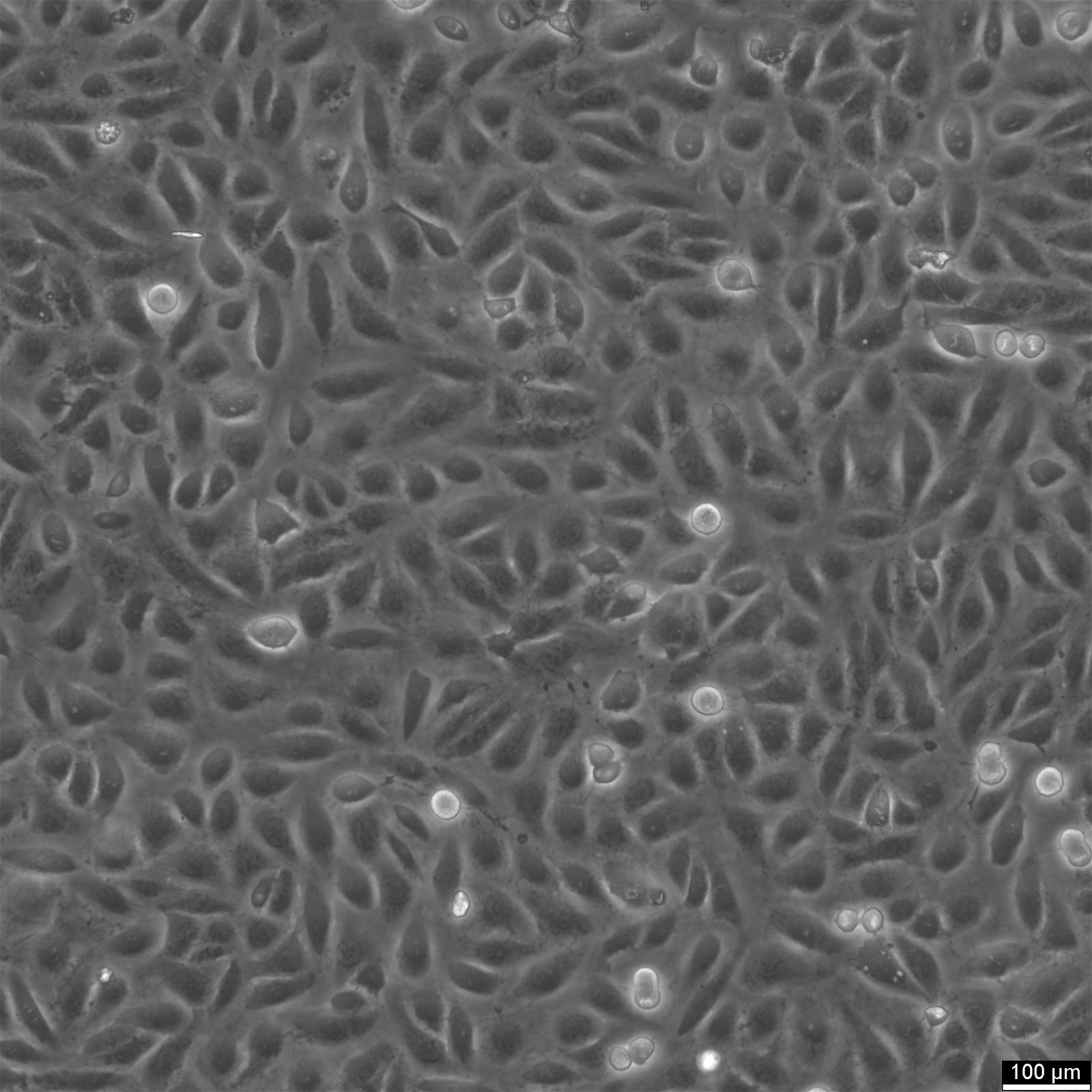
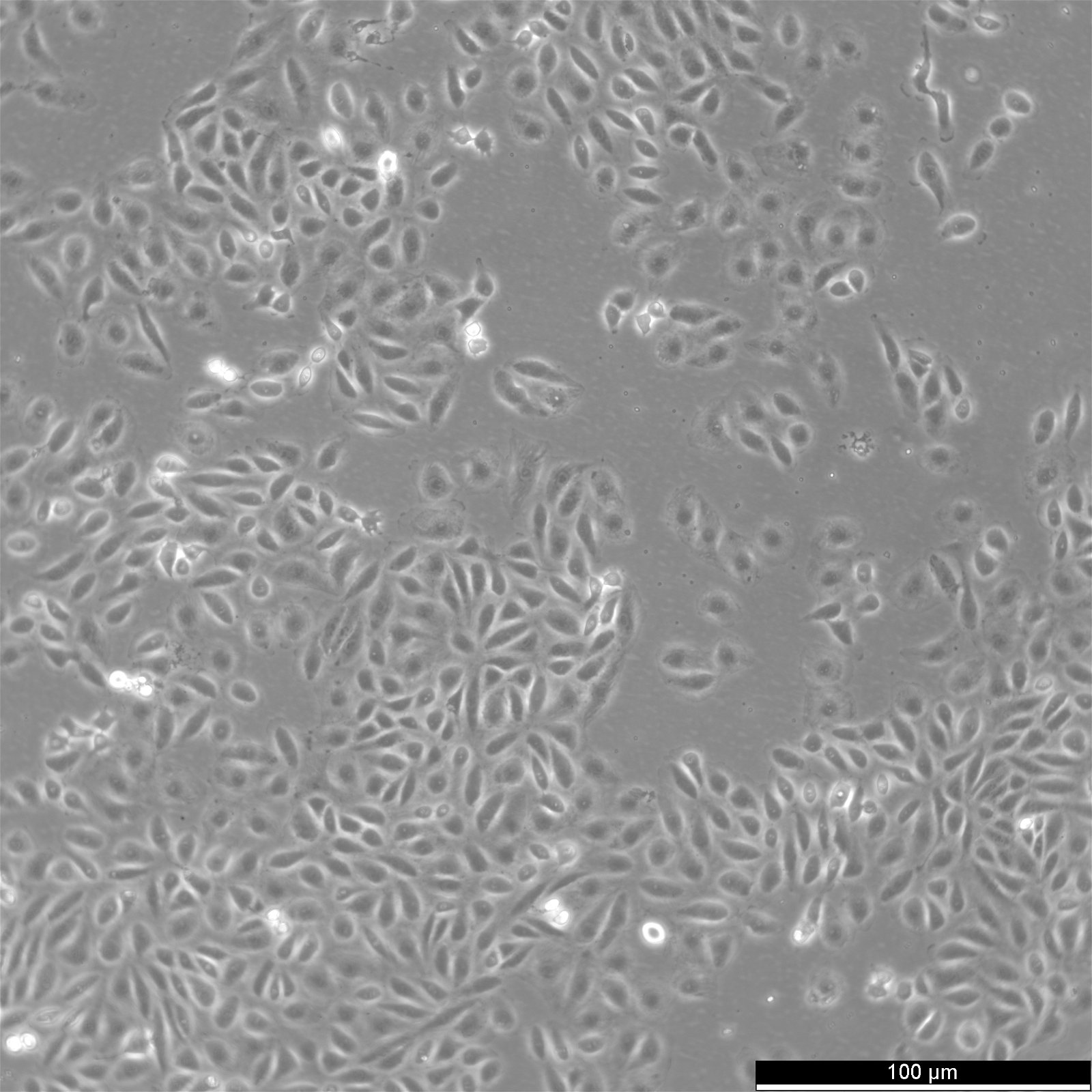
TPC-1 Cells










USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The TPC-1 cell line originates from a papillary thyroid carcinoma (PTC) and is widely utilized as a model for studying the molecular mechanisms of thyroid cancer. This cell line is notable for harboring the RET/PTC1 rearrangement, a hallmark genetic alteration in PTC. The RET/PTC1 fusion results in constitutive activation of RET tyrosine kinase signaling, driving oncogenic processes such as increased cellular proliferation, survival, and differentiation. This genetic feature has made TPC-1 a valuable tool in understanding thyroid oncogenesis and in evaluating targeted therapies. Derived from a well-differentiated thyroid tumor, TPC-1 retains epithelial characteristics and exhibits features associated with thyroid differentiation, including thyroglobulin production. TPC-1 has been extensively studied for its signaling pathways, particularly the MAPK and PI3K/AKT pathways, which are activated downstream of RET/PTC1. These pathways are critical to thyroid tumor progression and represent targets for therapeutic intervention. In addition to its genetic and cellular characteristics, TPC-1 has been employed in in vitro and in vivo models to investigate the effectiveness of RET inhibitors and other targeted therapies. Its well-characterized genetic background and responsiveness to pharmacological agents make it a crucial model for translational research in thyroid cancer. Studies comparing TPC-1 with other thyroid cancer cell lines have also highlighted its role in identifying common and distinct molecular features of thyroid cancer subtypes, aiding in the development of personalized treatment strategies. |
|---|---|
| Organism | Human |
| Tissue | Thyroid |
| Disease | Thyroid gland papillary carcinoma |
| Synonyms | TPC1 |
Characteristics
| Age | Adult |
|---|---|
| Gender | Female |
| Morphology | Epithelial |
| Growth properties | Adherent |
Regulatory Data
| Citation | TPC-1 (Cytion catalog number 305054) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_6298 |
Biomolecular Data
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS, 4.5 g/L Glucose |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305054-181124 | Certificate of Analysis | 23. May. 2025 | 305054 |
