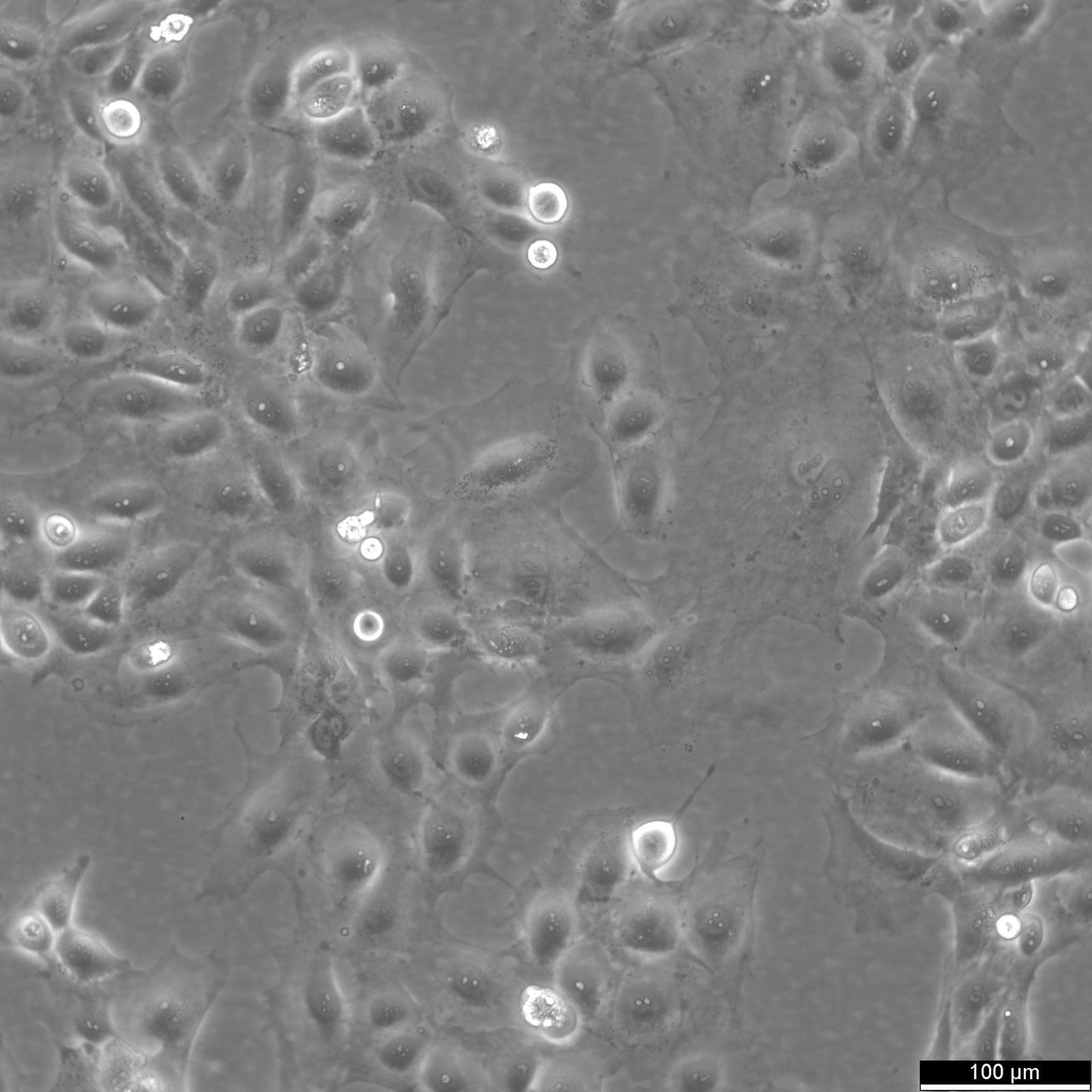
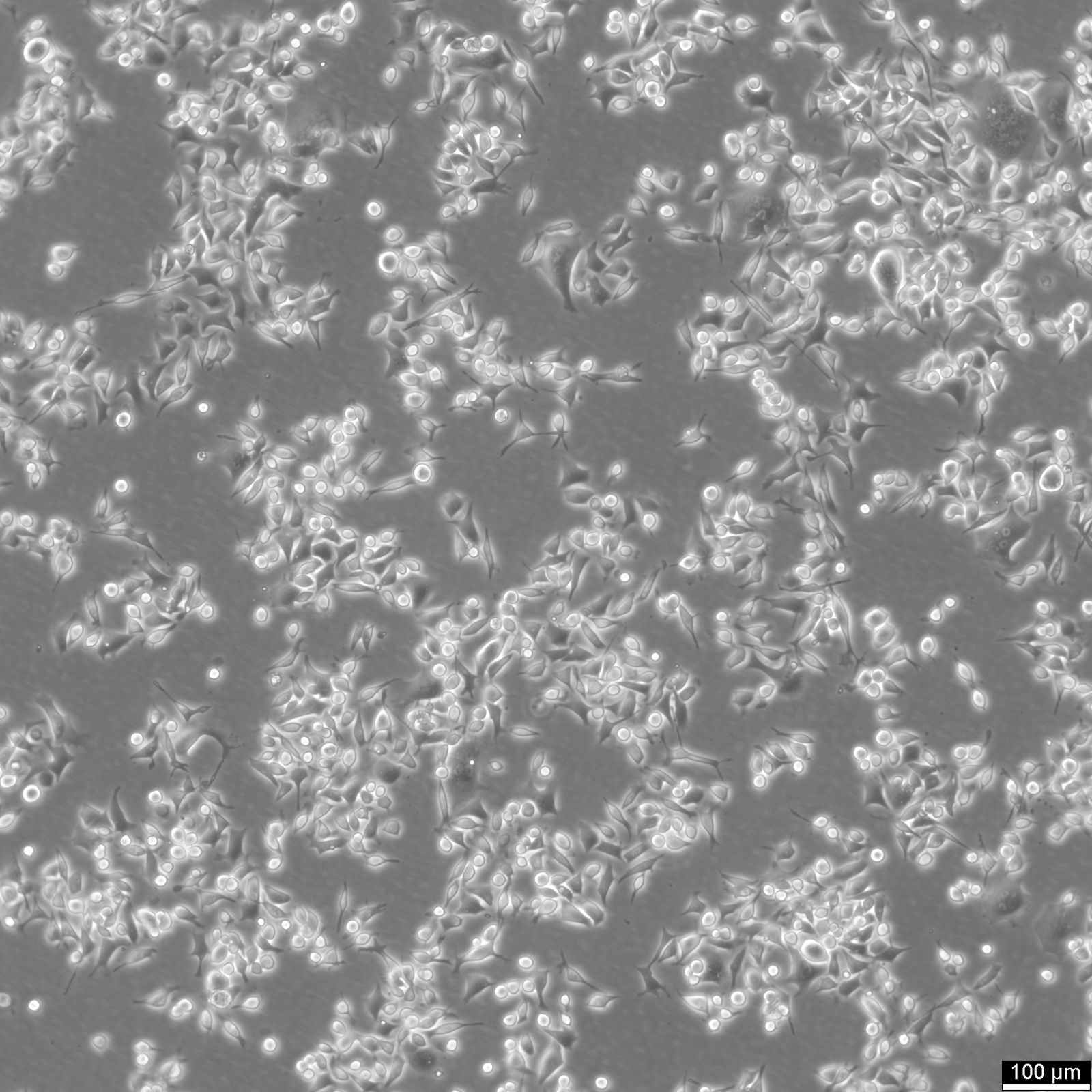
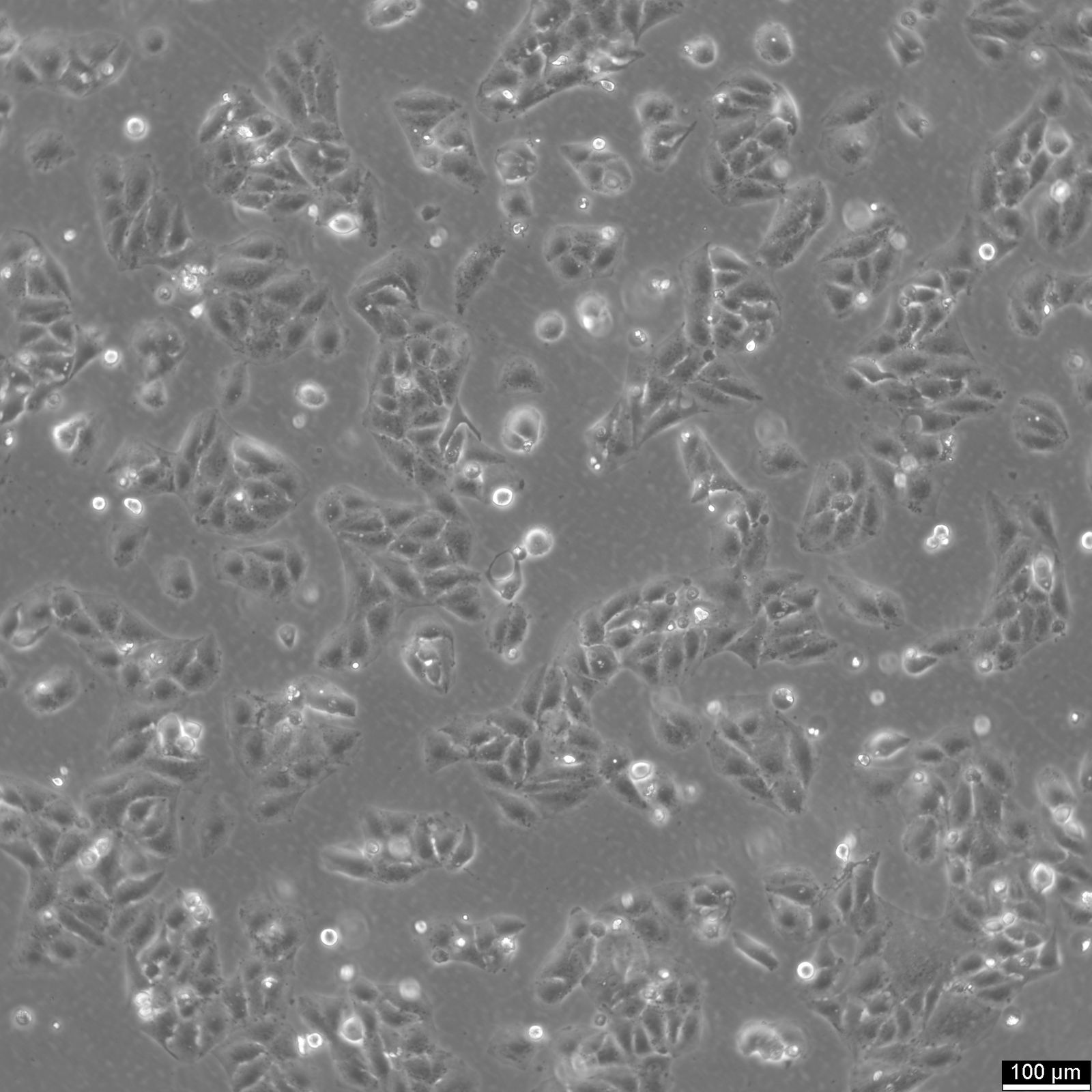
SNU-368 Cells












USD$540.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The SNU-368 cell line is a human hepatocellular carcinoma (HCC) model derived from a primary tumor of a 54-year-old male patient. This cell line is part of a panel of eight HCC cell lines established from Korean patients, designed to reflect the diverse molecular and phenotypic characteristics of liver cancers. SNU-368 cells exhibit a polygonal adherent morphology and display many histological features of the original tumor, including trabecular and acinar arrangements, which are characteristic of Edmondson grade II to IV differentiation. Genetically, SNU-368 cells harbor integrated hepatitis B virus (HBV) DNA and express HBV transcripts, including HBx and preS/S. These features make it a valuable model for studying HBV-related hepatocarcinogenesis. SNU-368 also expresses transferrin and insulin-like growth factor II (IGF-II), but it does not produce alpha-fetoprotein (AFP), either at the RNA or protein level. Such molecular characteristics are important for exploring liver cancer pathways associated with viral infection, growth factor signaling, and metabolic alterations. SNU-368 has been employed in pharmacogenomic studies, particularly in the Liver Cancer Model Repository (LIMORE), to investigate drug responses and identify potential biomarkers for targeted therapies. The cell line’s inclusion in large-scale genomic and transcriptomic analyses underscores its relevance in modeling the heterogeneity of primary HCCs, making it a robust tool for studying the molecular underpinnings of liver cancer and evaluating novel therapeutic agents. |
|---|---|
| Organism | Human |
| Tissue | Liver |
| Disease | hepatocellular carcinoma |
| Synonyms | SNU368 |
Characteristics
| Age | 54 years |
|---|---|
| Gender | Male |
| Ethnicity | Korean |
| Morphology | Polygonal |
| Cell type | Endothelial |
| Growth properties | Adherent |
Regulatory Data
| Citation | SNU-368 (Cytion catalog number 305631) |
|---|---|
| Biosafety level | 2 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_3948 |
Biomolecular Data
| Viruses | HBV |
|---|---|
| Mutational profile | Mutation: ARID1A, Simple, p.Leu1607Profs*41 (c.4817dupT), Unspecified; Mutation: AXIN1, Simple, p.Gln184Ter (c.550C>T), Unspecified; Mutation: TERT, Simple, c.1-124C>T (c.228C>T) (C228T), Unspecifie; Mutation: TP53, Simple, p.Ser106Arg (c.318C>G), Unspecified |
| Karyotype | Has lost chromosome Y. |
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% heat inactivated FBS |
| Dissociation Reagent | Accutase |
| Doubling time | 41 hours |
| Subculturing | Remove medium, add fresh 0.25 % trypsin 0.02 % EDTA solution, stand culture flask at 37''''C for 3 to 5 minutes, add culture medium and collect the cells, transfer the medium into 15ml tube, centrifuge, aspirate the medium, resuspend the pellets with culture medium and dispense into the culture flask |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305631-201125 | Certificate of Analysis | 15. Jan. 2026 | 305631 |
-
Related products
Related products
SNU-601 CellsOrganism Human Tissue Stomach Disease Gastric signet ring cell adenocarcinoma USD$550.00*