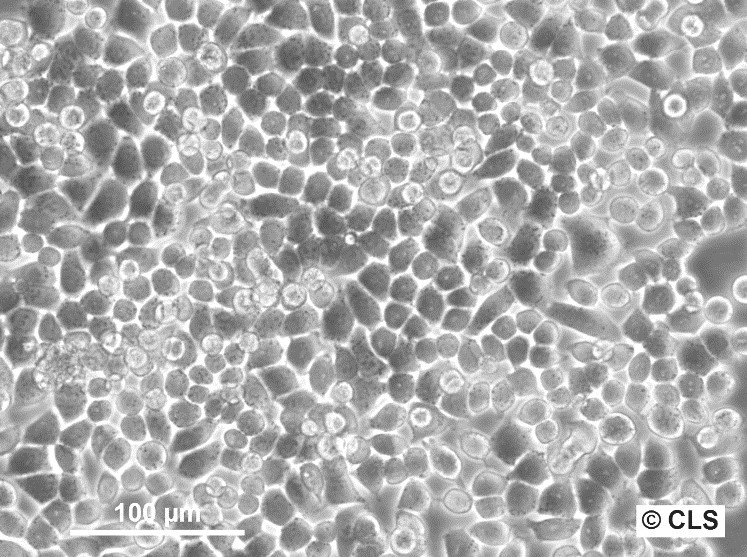
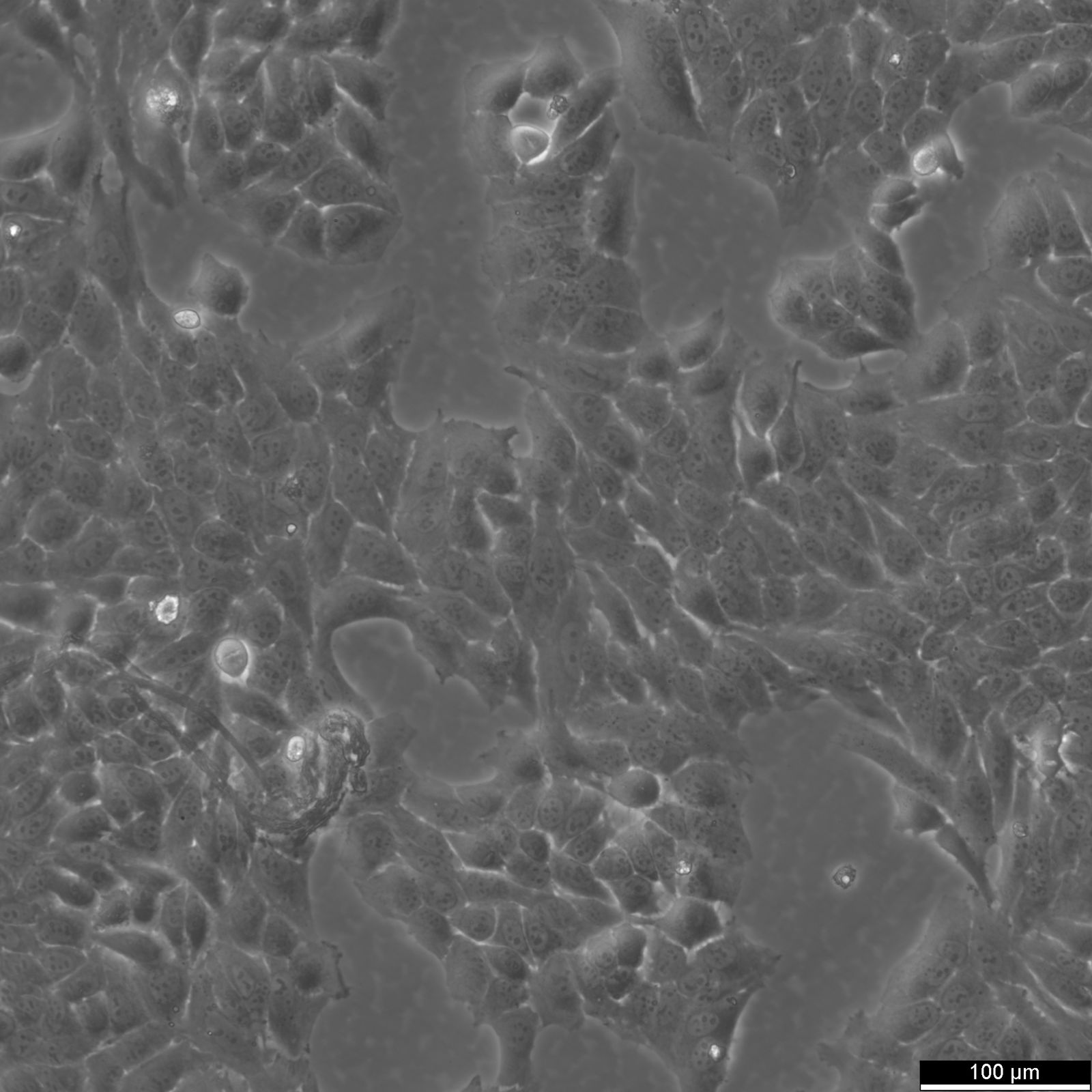
HeLa Cells
























USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
History of Henrietta Lacks' cells
| Description | HeLa cells, derived from the cervical cancer cells of Henrietta Lacks, are an immortal cell line widely employed in biomedical research. The human cell line Hela has significantly contributed to significant research advances and continues to play a pivotal role in laboratories worldwide. In 1951, Henrietta Lacks, a young mother of five, sought medical attention at The Johns Hopkins Hospital for vaginal bleeding, where Dr. Howard Jones identified a significant malignant tumor on her cervix. At that time, the Johns Hopkins Medicine Institute was among the few institutions offering medical care to impoverished African Americans. Henrietta Lacks underwent radium treatment for her cervical cancer, the leading therapy available then. During her treatment, a biopsy was conducted, and a sample of her cancerous cells was sent to Dr. George Otto Gey's lab. Dr. Gey had been attempting to cultivate cells from cervical cancer patients of diverse backgrounds, but without success until Henrietta's cells, which were the first cells to proliferate continuously, a discovery that set them apart from all previous samples. Henrietta Lacks' cervical carcinoma was later found to have been caused by the Human papillomavirus (HPV). HPV is a common virus that can lead to cervical cancer among other diseases. Research on HeLa cells has significantly contributed to understanding the role of HPV in cervical cancer, leading to the development of preventive HPV vaccines, which have had a profound impact on reducing the incidence of HPV-related cancers. These extraordinary cells, termed "HeLa" cells after Henrietta Lacks' initials, have since become instrumental in medical research. They have enabled scientists to investigate cancer cell growth, the impact of various substances, and the workings of viruses, significantly contributing to medical advancements, including the development of vaccines for polio and COVID-19, without the ethical concerns of direct human experimentation. HeLa cells are widely used for gene function studies, recombinant protein production, and gene therapy due to their high transfection efficiency and susceptibility to viral infections. They are pivotal in researching viral behaviors, including replication and pathogenesis, and have played a key role in Hepatitis B research by expressing viral proteins and aiding in the development of diagnostic tests and vaccines, thereby significantly advancing global health measures. HeLa cells continue to be an invaluable resource for ongoing research in medicine and science. The significance of HeLa cells and other immortal cell lines cannot be overstated, as they continue to shape the field of medicine and infectious disease research, and they represent a lasting legacy of Henrietta Lacks and her contributions to scientific advancement. |
|---|---|
| Organism | Human |
| Tissue | Cervix |
| Disease | Adenocarcinoma |
| Applications | Transfection host |
| Synonyms | HELA, Hela, He La, He-La, Henrietta Lacks cells, Helacyton gartleri |
Details
| Age | 30 years |
|---|---|
| Gender | Female |
| Ethnicity | African American |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Documentation
| Citation | HeLa (Cytion catalog number 300194) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0030 |
Genetic profile
| Isoenzymes | G6PD, A |
|---|---|
| Virus susceptibility | Human adenovirus 3, Encephalomyocarditis virus, Human poliovirus 1, Human poliovirus 2, Human poliovirus 3 |
| Reverse transcriptase | Negative |
| Products | Keratin, Lysophosphatidylcholine (lyso-PC) induces AP-1 activity and c-jun N-terminal kinase activity (JNK1) by a protein kinase C-independent pathway |
| Karyotype | The HeLa cell line, with its complex karyotype featuring a high degree of aneuploidy and structural rearrangements, is known for its rapid growth and longevity in culture. HeLa cells typically exhibit 82 chromosomes, although the range can vary from 70 to 164. Notably, 98% of HeLa cells possess a small telocentric chromosome, and 100% exhibit aneuploidy in a substantial number of cells examined. These chromosomal abnormalities underpin their fast growth and immortality, along with their association with cervical cancer and other cancerous cells. |
Handling the Hela cell line
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Doubling time | 28 to 36 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 2 to 3 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 to 48 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality assurance of Hela cells
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300194-1620 | Certificate of Analysis | 26. May. 2025 | 300194 |
| 300194-010425 | Certificate of Analysis | 23. May. 2025 | 300194 |
| 300194-150823 | Certificate of Analysis | 23. May. 2025 | 300194 |
| 300194-220324 | Certificate of Analysis | 23. May. 2025 | 300194 |
| 300194-040725 | Certificate of Analysis | 18. Aug. 2025 | 300194 |
-
Related products
Related products
Hela 229 CellsOrganism Human Tissue Cervix Disease Human papillomavirus-related endocervical adenocarcinoma USD$395.00*HeLa-Luc CellsOrganism Human Tissue Uterus, cervix Disease Human papillomavirus-related endocervical adenocarcinoma USD$800.00*