HEK293-FOLR1 Cells

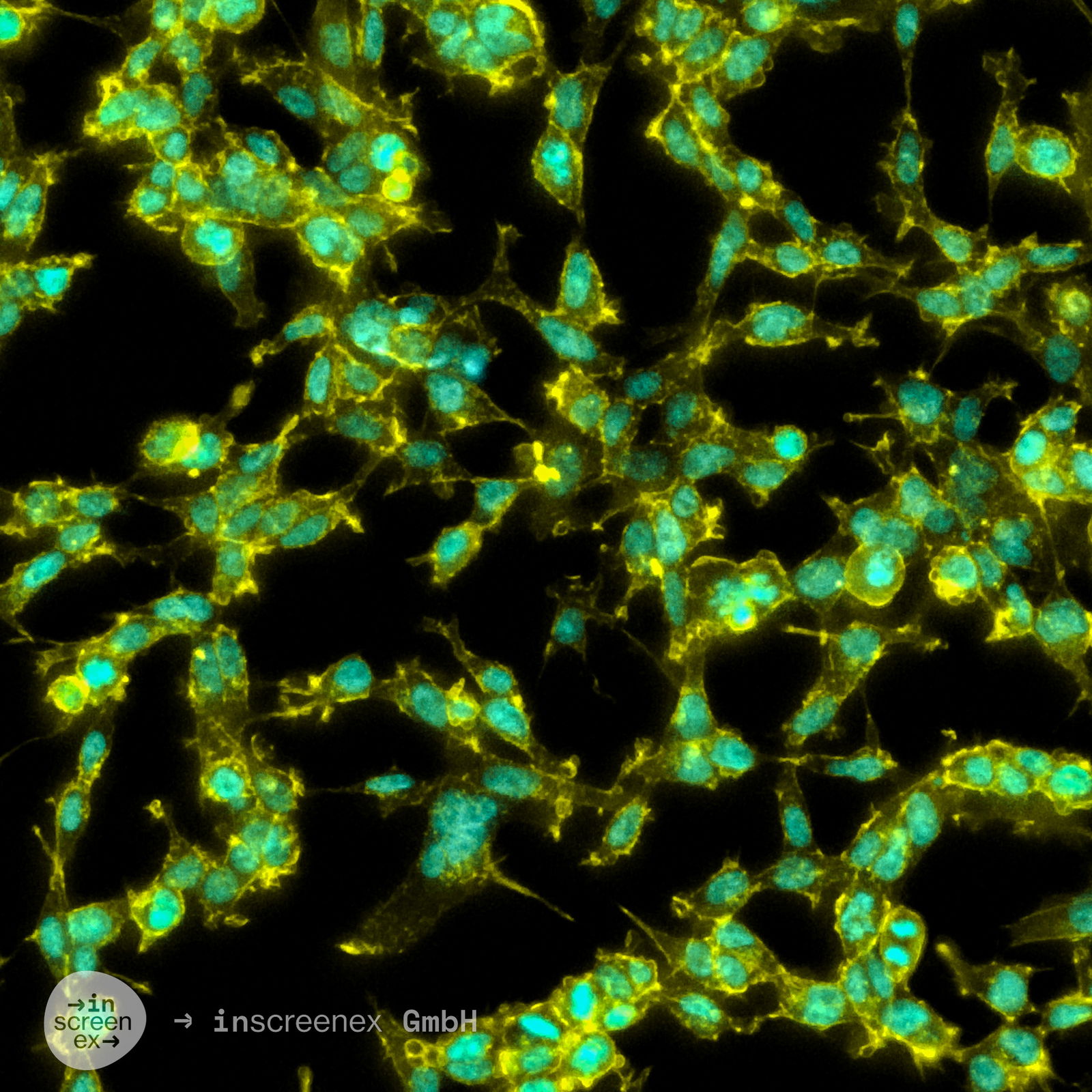









USD$2,200.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | Disclaimer: The prices displayed for cell lines are exclusively for academic/not-for-profit customers. For commercial entitites the price is approximately €6,250. The HEK293-FOLR1 cell line is a stable recombinant HEK293 cell line engineered to express the FOLR1 receptor at a medium-high level, approximately 15,000 molecules per cell. This cell line was developed using inscreenex's landing pad technology, ensuring precise and reproducible integration of the FOLR1 gene at a specific, pre-validated genomic locus. FOLR1, also known as Folate Receptor Alpha (FRα) or FBP, is a GPI-anchored membrane protein with a strong affinity for folate, facilitating its transport into cells. FOLR1 is markedly overexpressed in certain cancers, such as ovarian, breast, and non-small cell lung cancers, making it a significant target for immuno-oncology therapies, including CAR T cell therapies and bispecific antibodies. The expression of FOLR1 in this cell line was confirmed using flow cytometry with a target-specific antibody, ensuring reliable and consistent receptor density across the cell population. |
|---|---|
| Organism | Human |
| Tissue | Fetal Kidney |
Characteristics
| Age | Fetus |
|---|---|
| Gender | Female |
| Morphology | Epithelial-like |
| Growth properties | Monolayer, adherent |
Regulatory Data
| Citation | HEK293-FOLR1 (Cytion catalog number 305425) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| GMO Status | GMO-S1: This HEK293 cell line contains a FOLR1 expression cassette for folate receptor research. This classification applies only within Germany and may differ elsewhere. |
Biomolecular Data
| Receptors expressed | FOLR1 (Folate Receptor Alpha (FRα) or FBP) |
|---|
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS, 1 mM sodium pyruvate, 10 mM HEPES, 1% NEAA. Add Geneticin (G418-Sulfat) to achieve a final concentration of 1 mg/mL. |
| Dissociation Reagent | Trypsin-EDTA |
| Subculturing | For routine adherent cell culture: Aspirate the old culture medium from the adherent cells, and wash them with PBS to remove any remaining medium. After aspirating the PBS, add the appropriate volume of Trypsin/EDTA solution based on the culture vessel size (e.g., 1 ml for a T25 flask, 3 ml for a T75 flask) and incubate at room temperature or 37°C until the cells detach (5-10 minutes). Monitor detachment under a microscope, and gently tap the vessel if necessary to release the cells. Once detached, add complete medium to inactivate the Trypsin/EDTA, gently resuspend the cells, and transfer an aliquot of the cell suspension into a new culture vessel containing fresh medium. Place the vessel in an incubator set to 37°C with 5% CO2, and change the medium every 2-3 days. |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, split the cells at a ratio of 1:2 to 1:3 in T25 flasks and allow the cells to recover from the freezing process and to adhere for at least 24 hours. For best attachment and viability after thawing the cells, we recommend using Collagen-coated flasks or plates for the initial seeding after cryo-recovery. Collagen coating is not required for subsequent routine culture of the cells. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|

-
Related products
Related products