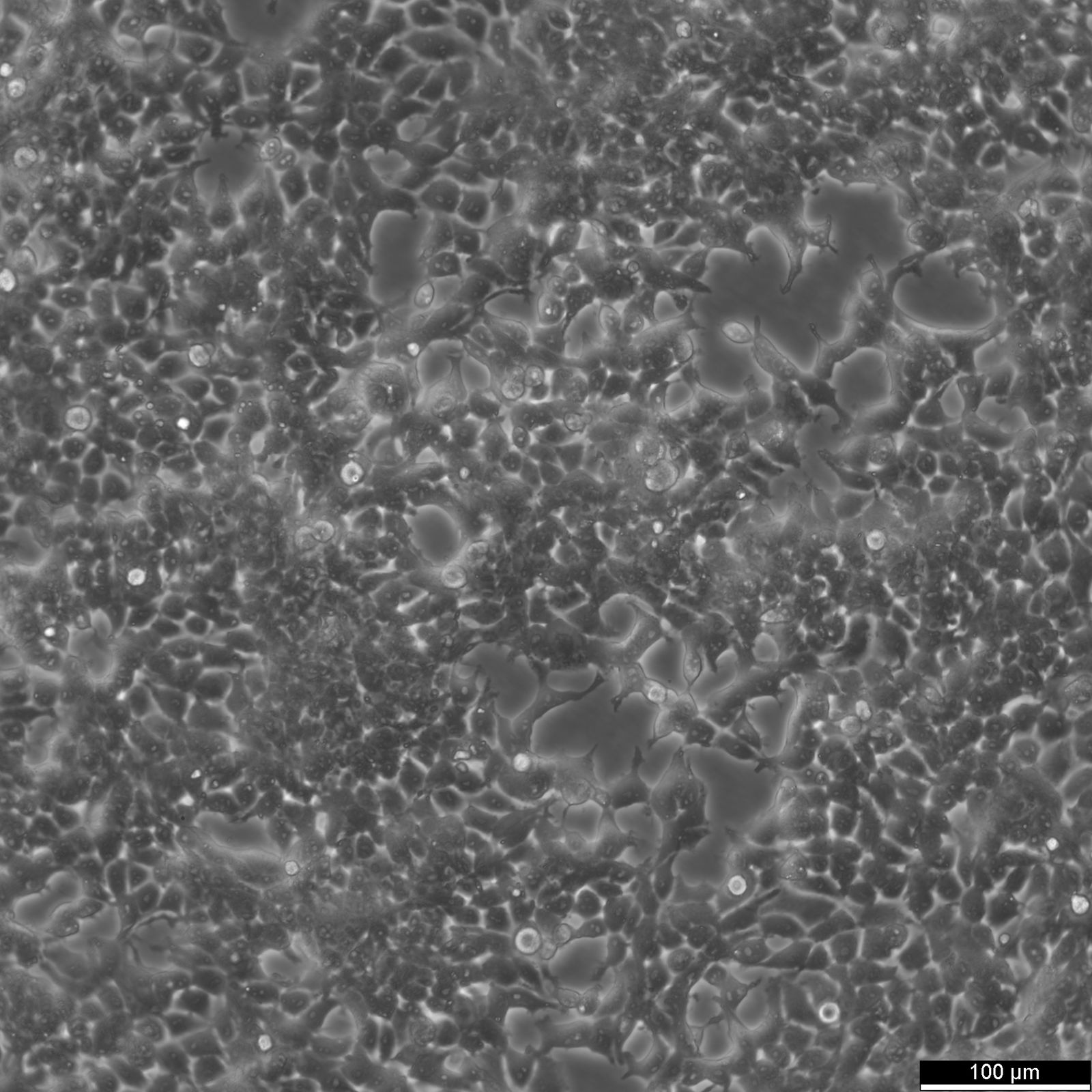
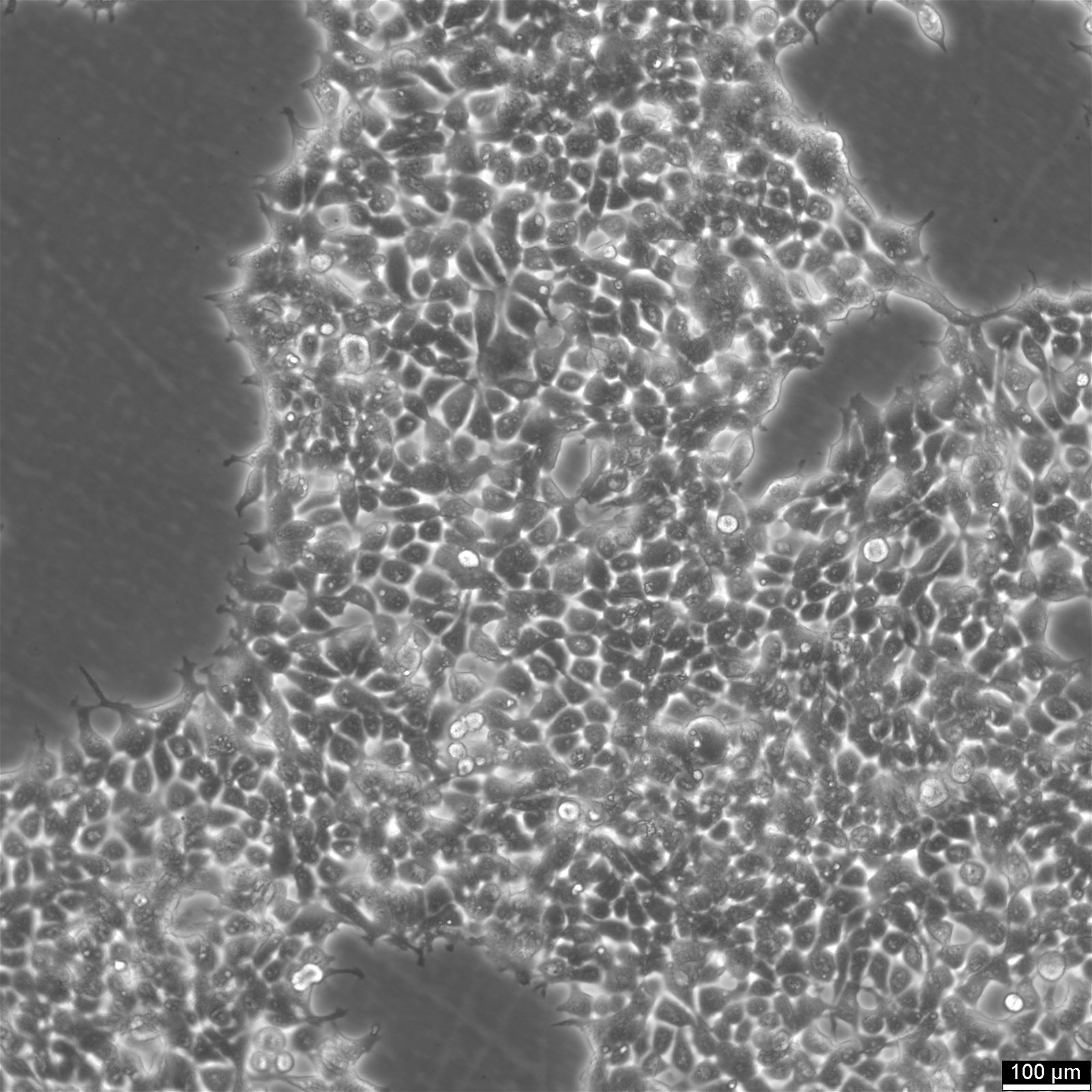
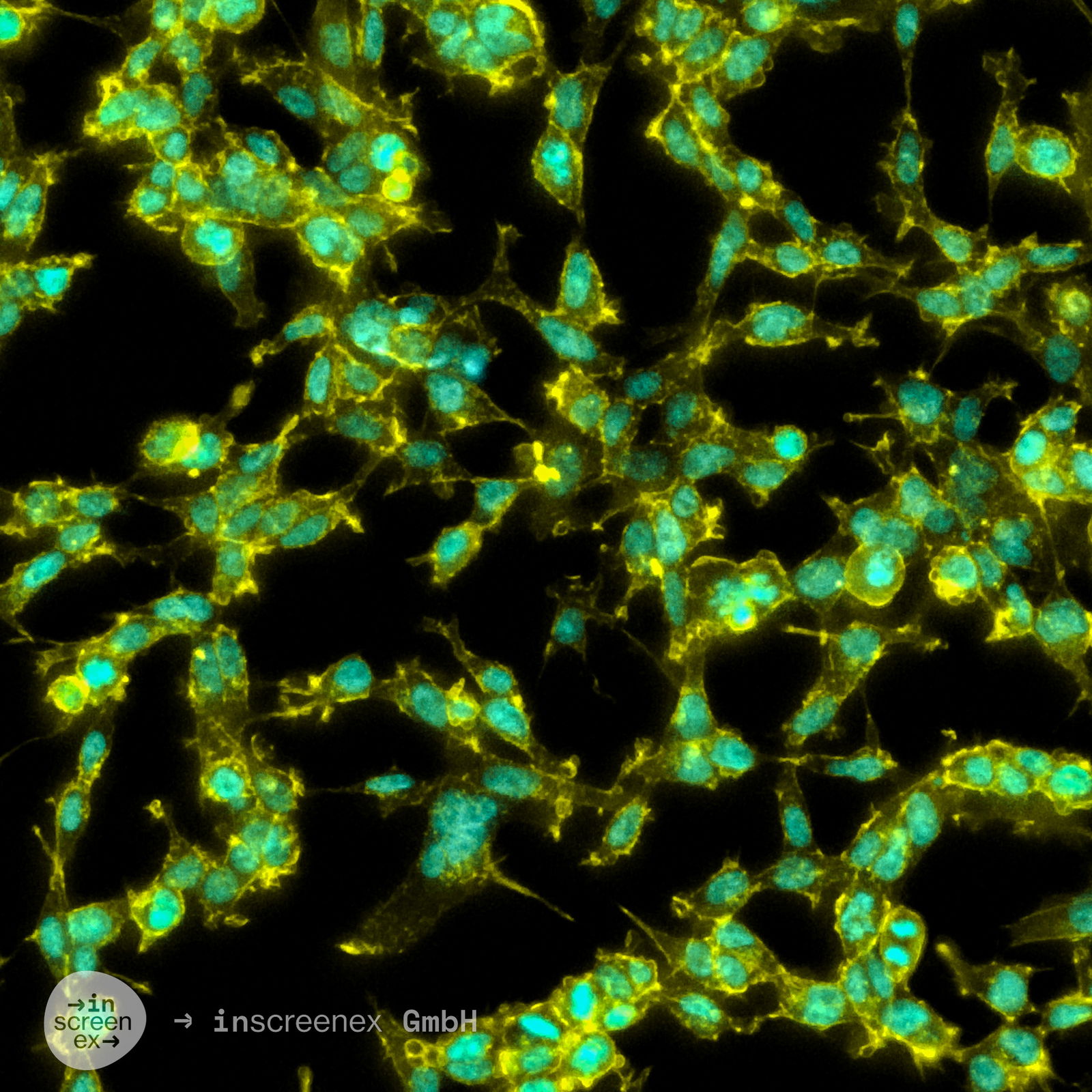
HEK293 Cells
























USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
Essential facts about HEK293 cells
| Description | The HEK293 cell line, an immortalized epithelial cell line derived from human embryonic kidney cells in the 1970s by Alex van der Eb at the University of Utrecht, has become a pivotal experimental model in molecular biology and biotechnological applications due to its remarkable versatility and ease of genetic manipulation. The transformation of the HEK293 cell line involved the integration of a specific segment from Adenovirus 5 DNA, embedding the adenoviral E1A and E1B genes within the cellular genome. The adenoviral DNA modification enabled the cell lines' ability to uptake foreign DNA efficiently, a feature known as high transfection efficiency. The integration of viral DNA into the HEK293 cell genome resulted in cellular immortalization and significantly enhanced the utility of these cells in biotechnological applications by facilitating the stable incorporation and expression of exogenous DNA, a process termed stable transfection. This capability allows for the persistent presence and function of foreign genes within the cells, making HEK293 an invaluable tool for genetic studies and biotechnology. As a result, HEK293 cells have become a fundamental resource in biotechnology for the production of recombinant proteins, including vital therapeutic proteins, and serving as robust host cells for the generation of viral vectors, particularly adenoviral and lentiviral vectors. HEK 293 cells are pivotal in the pharmaceutical industry for high-throughput screening assays, the manufacture of gene therapies targeting specific genes related to single gene disorders, and adenoviral infection studies. In industrial biotechnology, the utility of the human cell line HEK293 extends to recombinant enzyme production, viral vector production, such as adenoviral vectors, protein production and the development of biosensors. Toxicology research benefits from the application of the HEK cell line in assessing the impacts of chemicals on cell biology, including the effects on typical kidney cells and the potential for gene therapies. The ability of the immortal cell line HEK293 to efficiently produce native proteins highlights their essential role in medical research, including cancer research and exploring the foundations of gene therapy. HEK293 cells offer a unique platform for studying cell biology and proteins of interest, surpassing other cell lines in versatility and utility in both research and industrial applications. In comparison, HEK293T cells, a variant of HEK293, are modified to enhance transfection efficiency, HEK293F cells are adapted for suspension culture to facilitate large-scale protein production, and other mammalian cell lines such as Vero cells, derived from monkey kidney tissue, are primarily utilized in vaccine development and viral studies. |
|---|---|
| Organism | Human |
| Tissue | Kidney |
| Applications | Transfection host |
| Synonyms | Hek293, HEK-293, HEK/293, HEK 293, HEK,293, 293, 293 HEK, 293 Ad5, Human Embryonic Kidney 293 |
Specifications about HEK 293
| Age | Fetus |
|---|---|
| Gender | Female |
| Morphology | Epithelial-like |
| Growth properties | Monolayer, adherent |
Documentation
| Citation | HEK293 (Cytion catalog number 300192) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0045 |
| GMO Status | GMO-S1: This HEK293 embryonic kidney-derived cell line contains adenovirus-5 E1A/E1B sequences due to transformation but does not release infectious virus, enabling high proliferative capacity. The modification is stably present in embryonal kidney cells. This classification applies only within Germany and may differ elsewhere. |
Genomics of HEK-293 cells
| Receptors expressed | Vitronectin |
|---|---|
| Protein expression | CEA negative, p53 positive |
| Tumorigenic | In nude mice |
| Virus susceptibility | Transformed with adenovirus 5 DNA adenovirus 5 DNA |
| Ploidy status | 30% of HEK293 cells have hypotriploid karyotypes with 64 modal chromosomes. Higher ploidies were found in 4.2% of cells. |
Culturing guidelines for the human cell line 293
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Doubling time | 30 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 will yield in a confluent layer in about 4 days. |
| Fluid renewal | 2 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality assurance
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300192-021123 | Certificate of Analysis | 23. May. 2025 | 300192 |
| 300192-060922 | Certificate of Analysis | 23. May. 2025 | 300192 |
| 300192-251124 | Certificate of Analysis | 23. May. 2025 | 300192 |
-
Related products
Related products