F9 Cells







USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
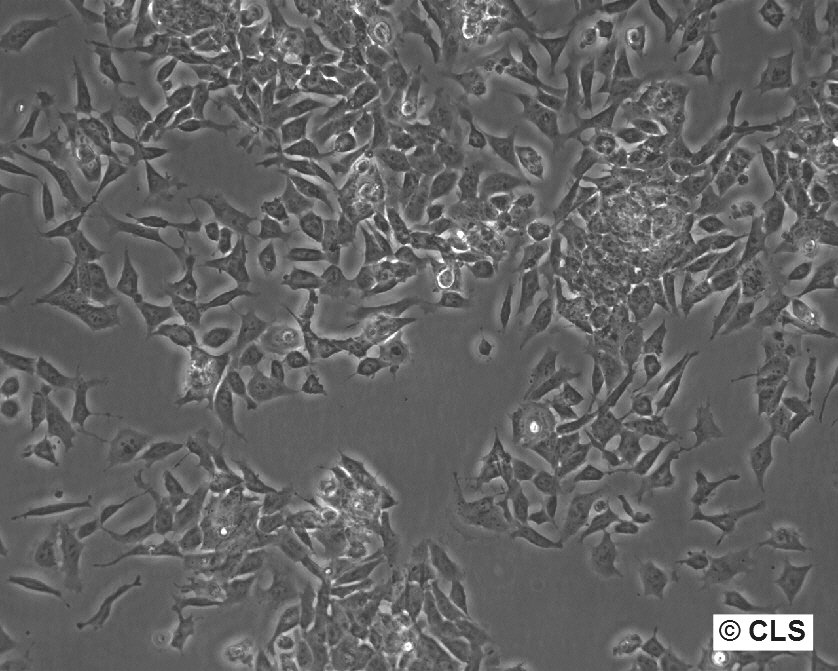
| Description | The F9 cell line, a murine embryonal carcinoma model derived from a testicular teratoma of the C57BL/6 mice, serves as an important tool in developmental biology and embryology. F9 cells are capable of differentiation into parietal endoderm when exposed to retinoic acid and dibutyryl cyclic AMP (cAMP). This differentiation is marked by significant changes in cellular behavior and protein expression, including the synthesis of plasminogen activator, laminin, and type IV collagen. These proteins are crucial for understanding the processes of tissue development and matrix formation in early embryonic stages. It is noted that cAMP's effectiveness in inducing differentiation in F9 cells is conditional upon prior treatment with retinoic acid, indicating a complex interplay between these signaling molecules in triggering developmental pathways. Additionally, F9 cells are characterized by having three copies of the beta 1 integrin gene, which may influence cell adhesion and mobility, further underscoring their utility in studying cell interactions and extracellular matrix composition. Safety profiling of these cells includes testing for ectromelia virus (mousepox), for which they have been found negative, ensuring their suitability for a broad range of experimental applications without the risk of viral contamination. |
|---|---|
| Organism | Mouse |
| Tissue | Testis |
| Disease | Teratocarcinoma |
Characteristics
| Breed/Subspecies | 129/Sv |
|---|---|
| Age | Embryo |
| Gender | Male |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | F9 (Cytion catalog number 400174) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 10090 |
| CellosaurusAccession | CVCL_0259 |
Biomolecular Data
| Viruses | MAP-test negative: Sendai, Ektromelie, Polyoma, K-Virus, Kilham, Reo 3, PVM, LCM, M.pulmonis, MVM, Theiler's GD VII, Toolan's H-1, MHV, LDV, RCV/SDA, M-Adenovirus, B.piliformis. |
|---|---|
| Products | Plasminogen activator, laminin, type IV collagen |
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 3.7 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | Coat cell culture flasks with Gelatine. 1 x 104 cells/cm2 will yield in a confluent layer in about 4 days. |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 400174-412 | Certificate of Analysis | 23. May. 2025 | 400174 |
