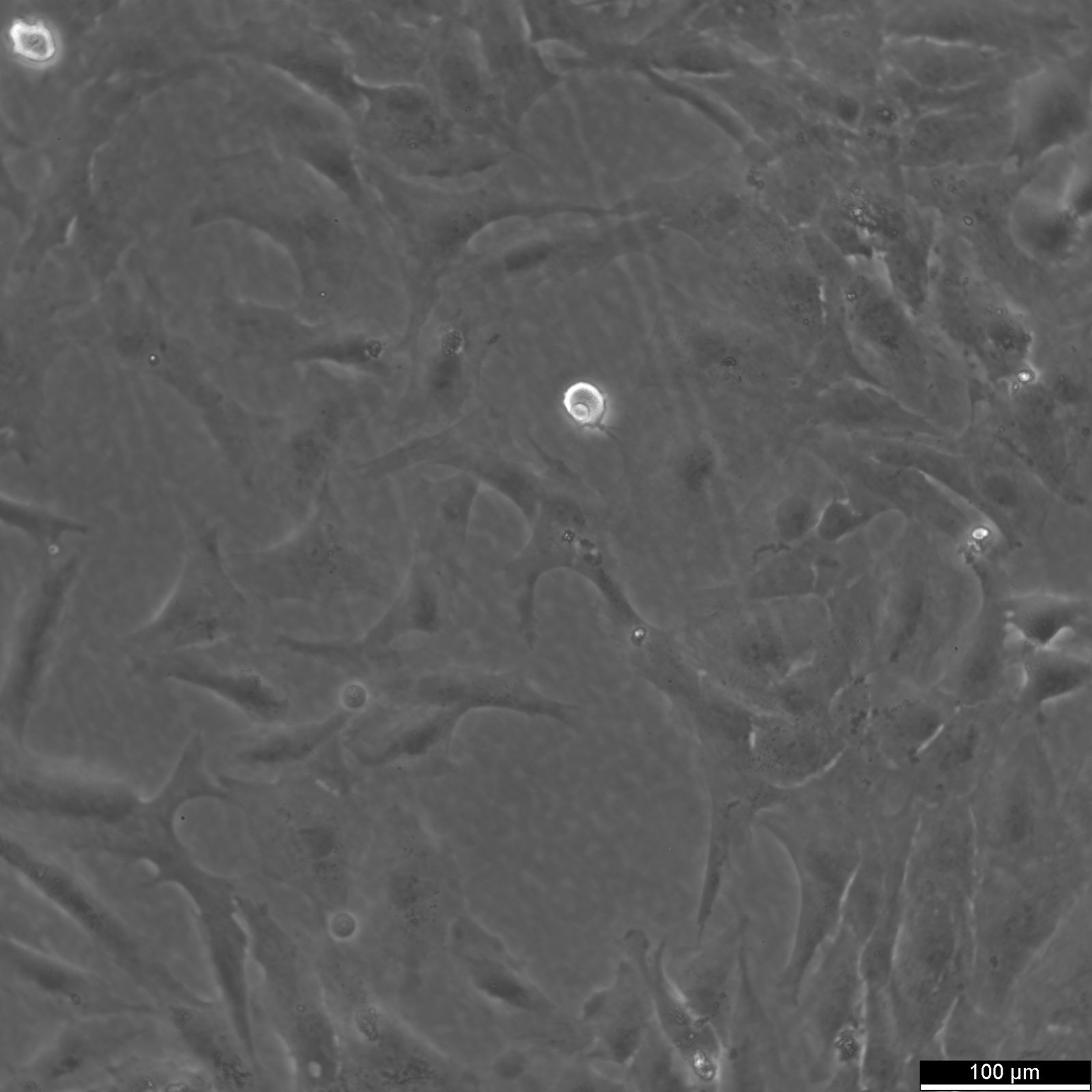
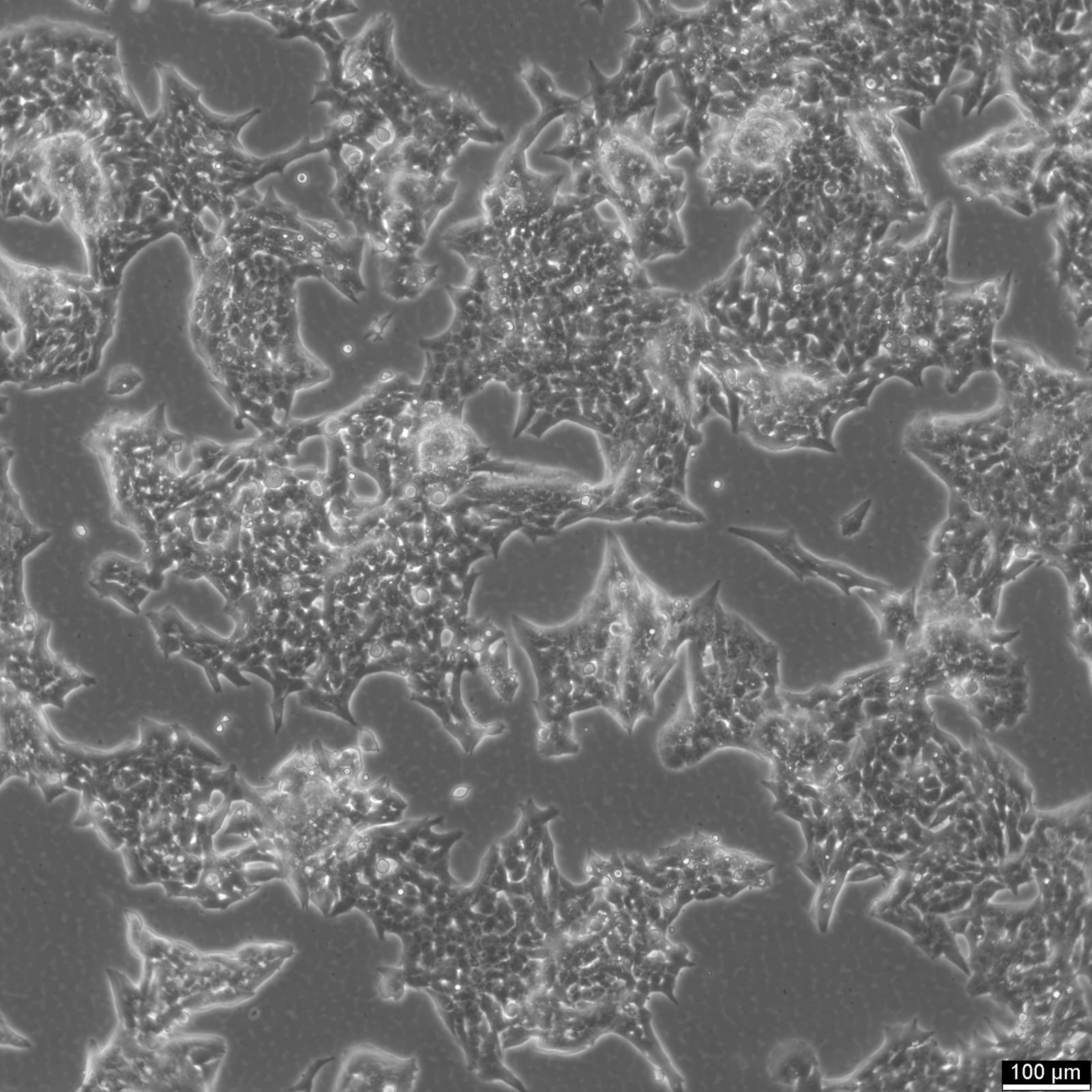
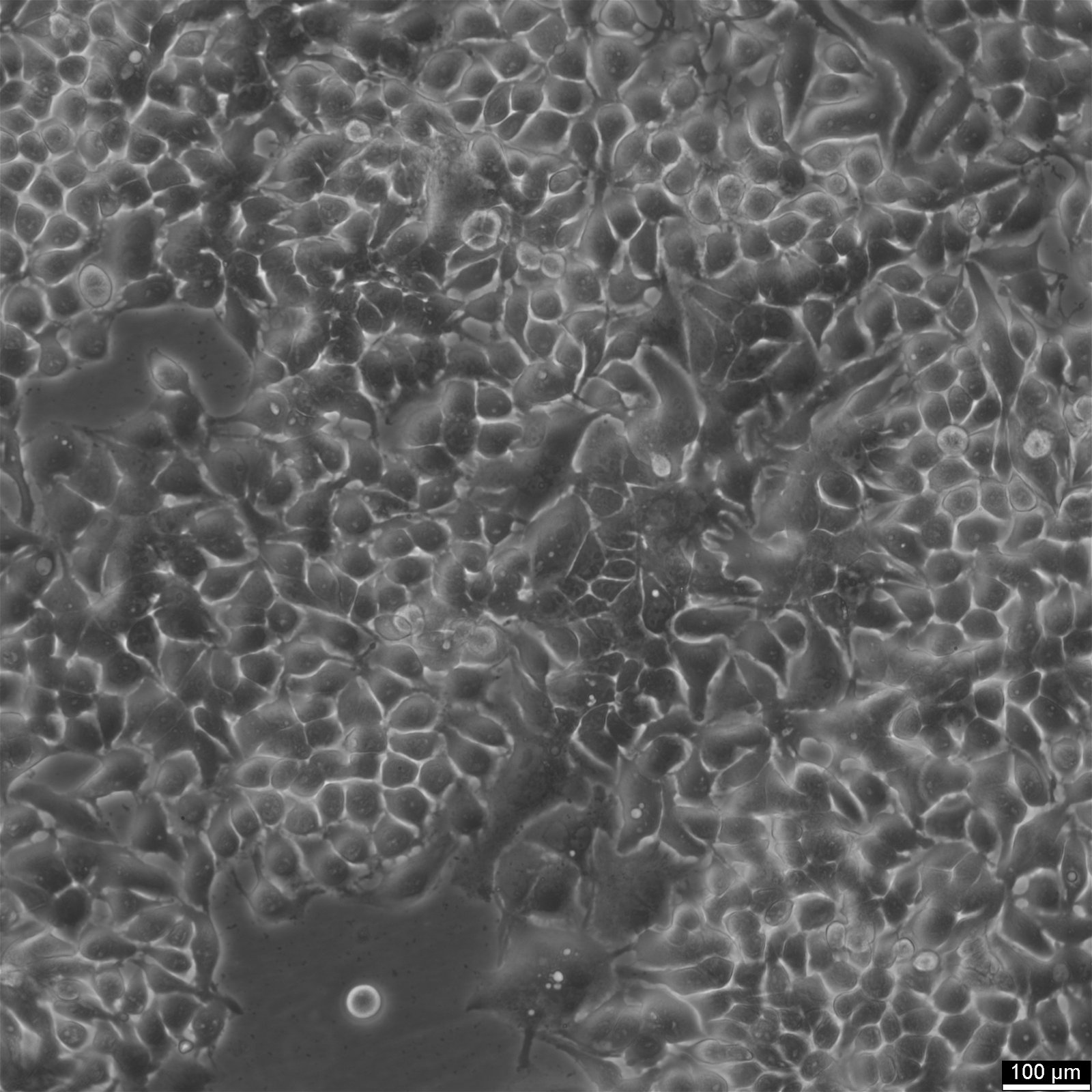
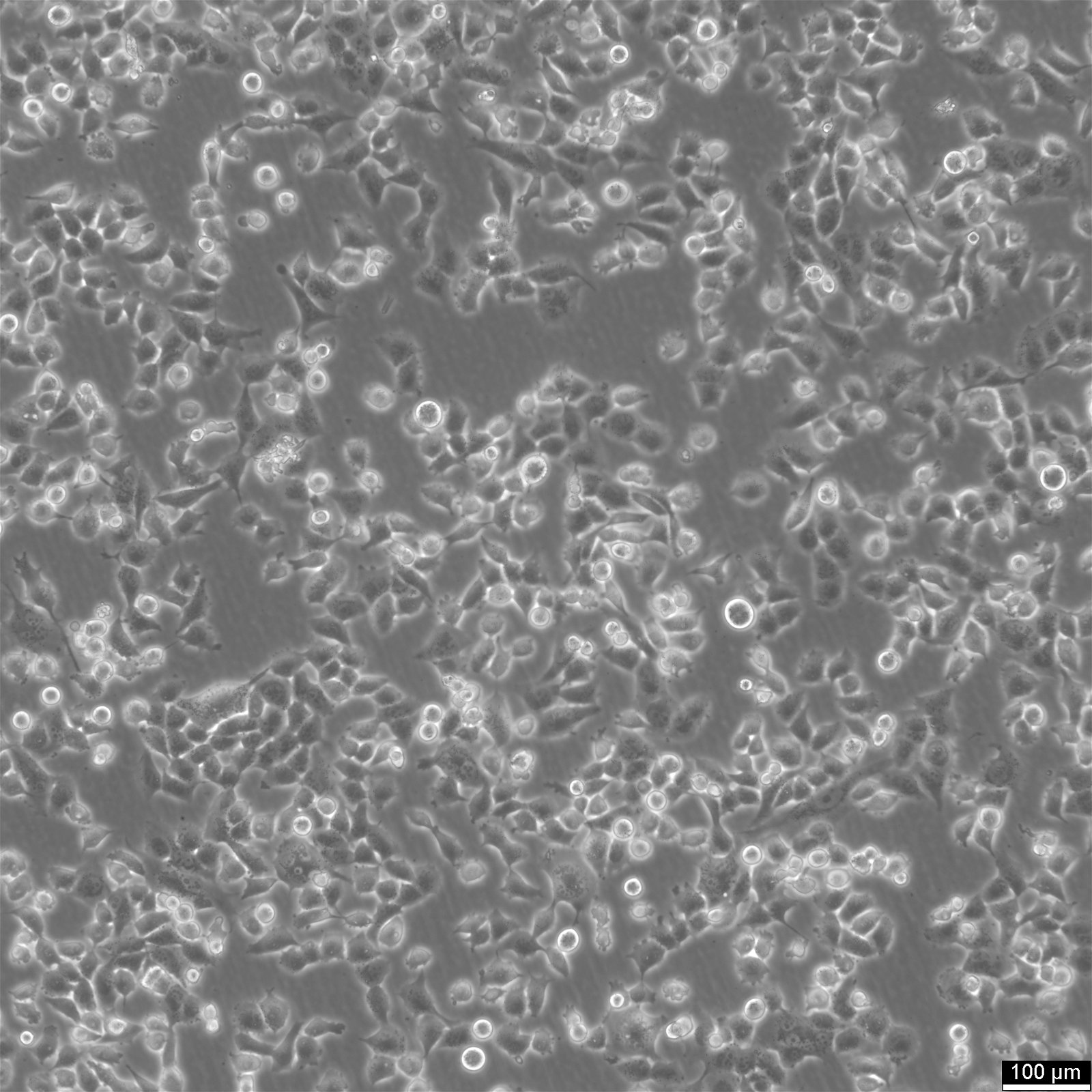
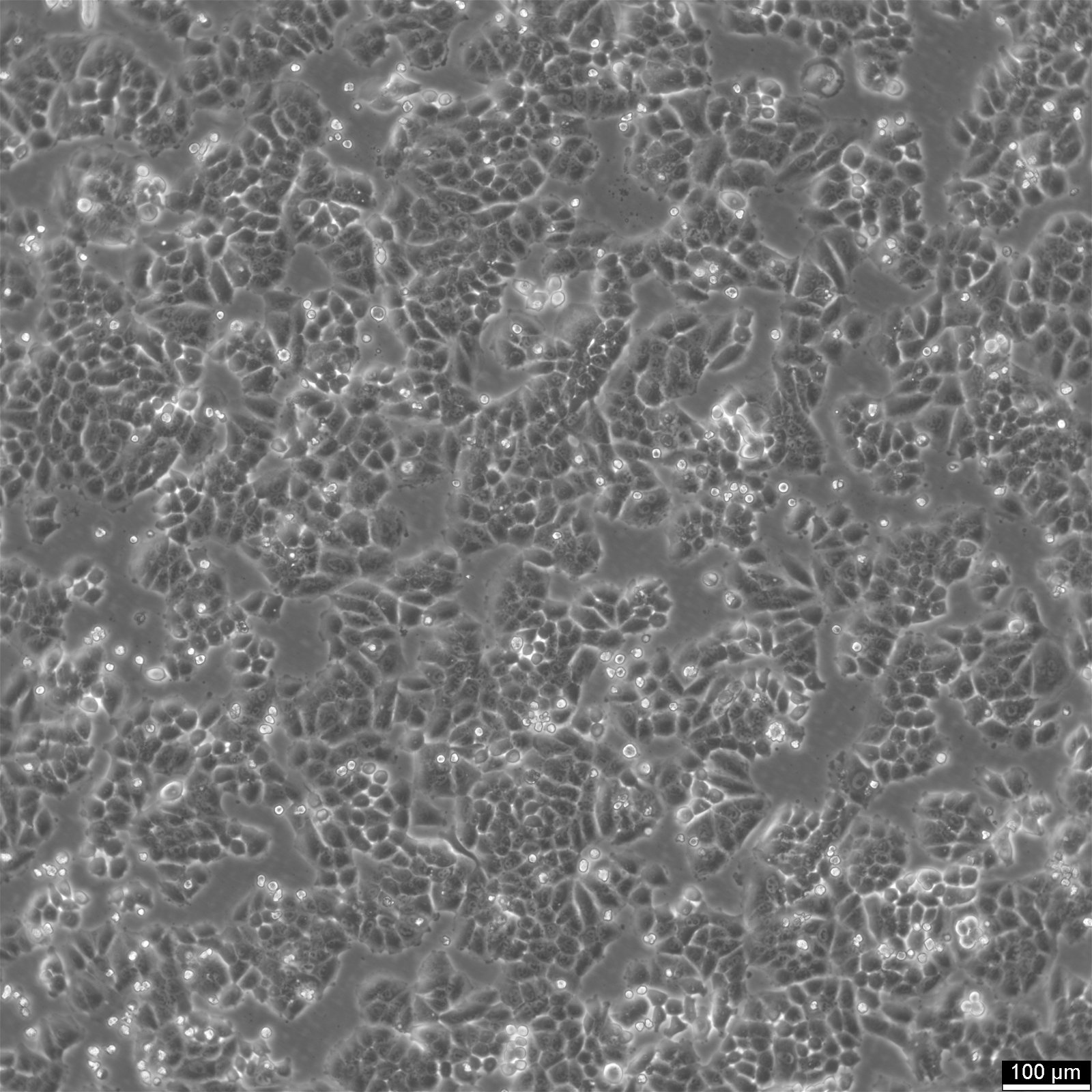
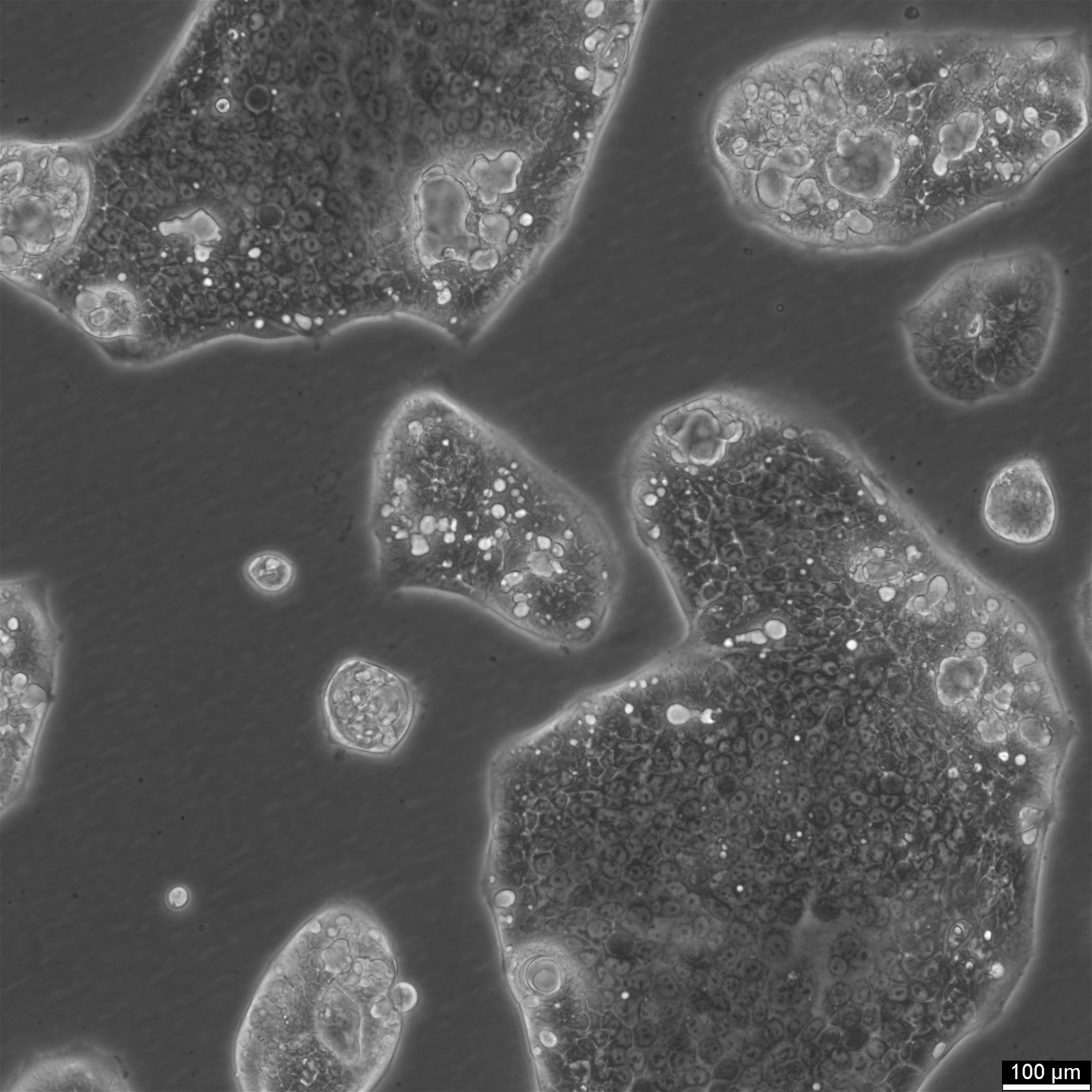
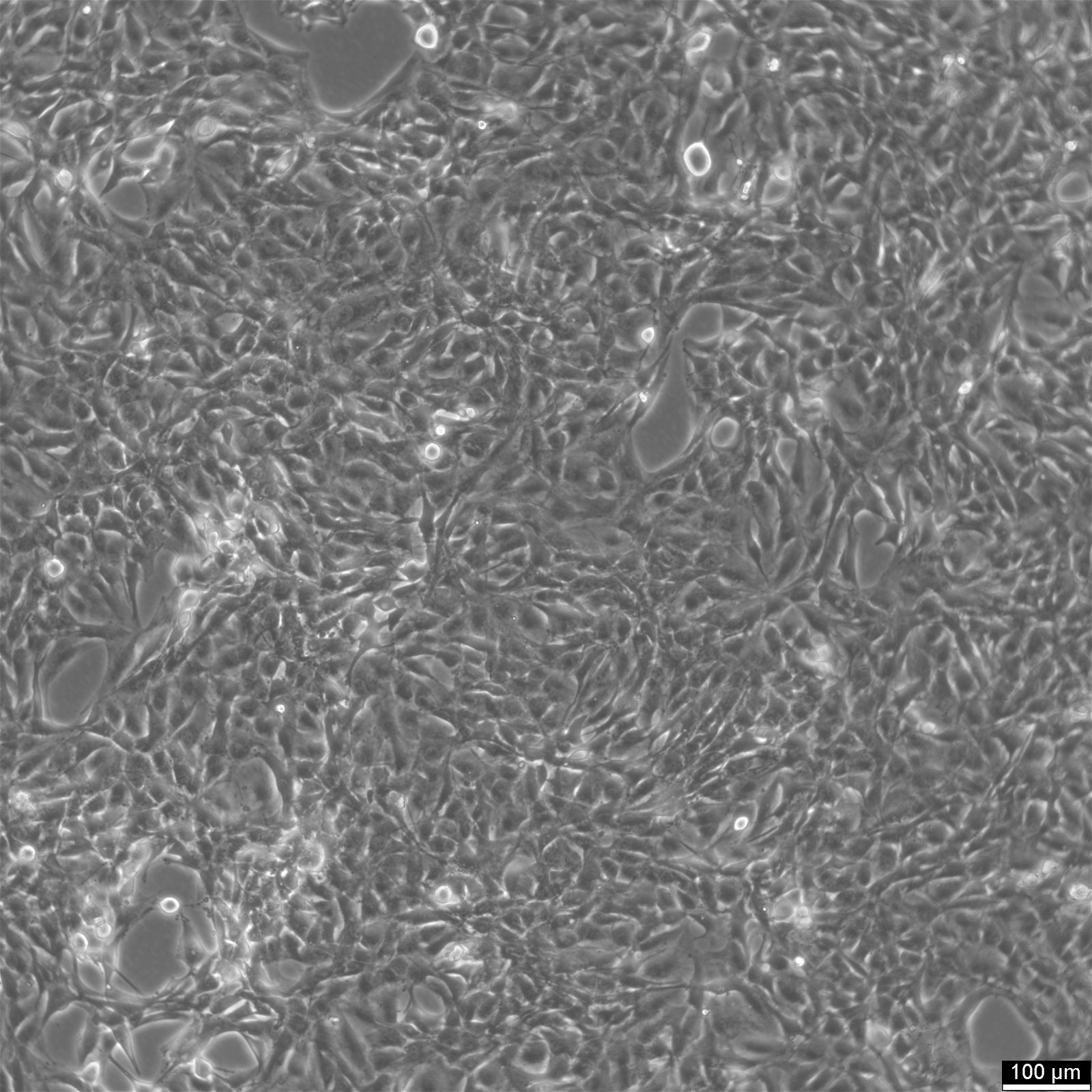
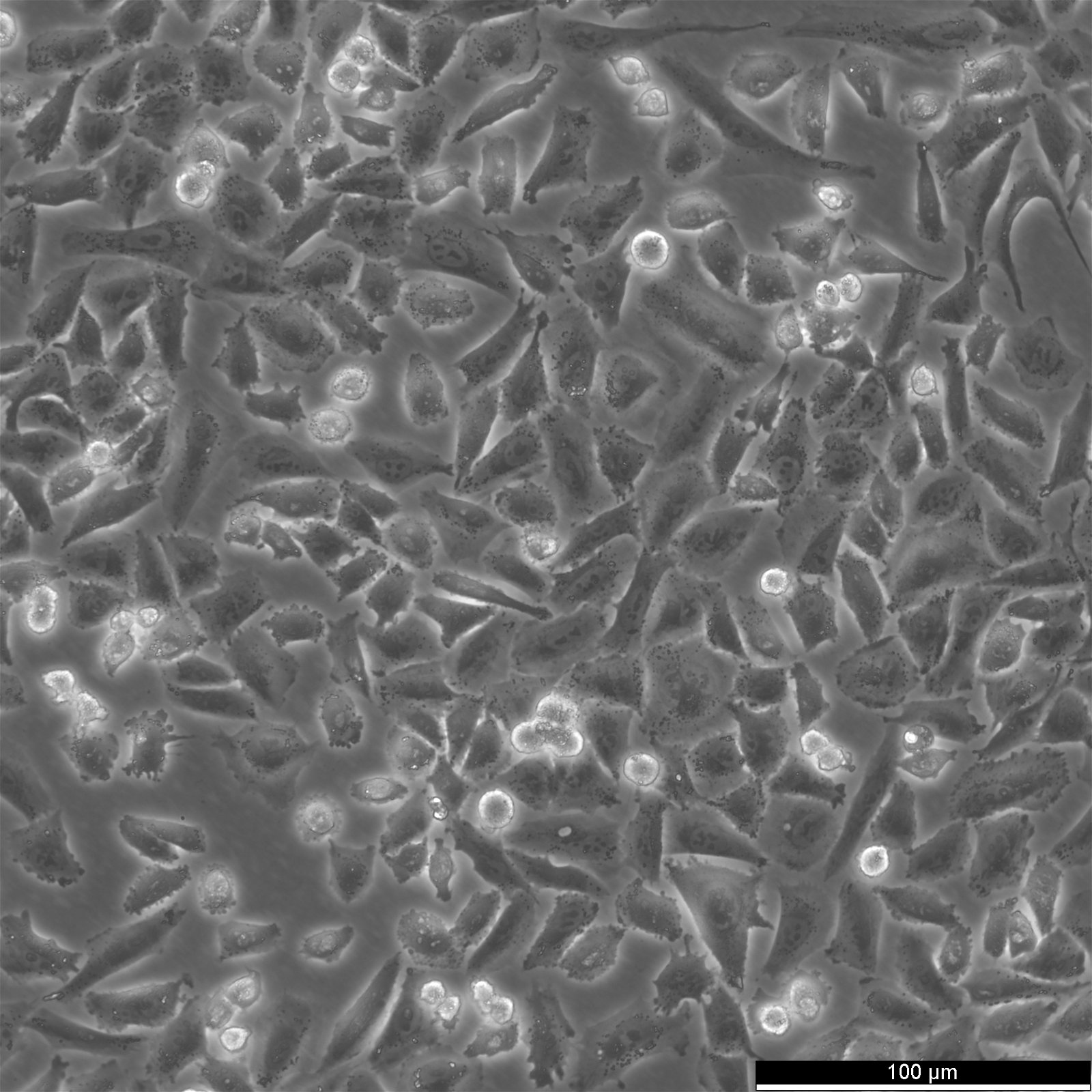
NCI-H2052 Cells




































CAD$593.40*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | NCI-H2052 is a human mesothelioma cell line derived from a pleural biopsy specimen of an adult patient diagnosed with malignant mesothelioma. As part of the NCI-Navy Medical Oncology Branch cell line panel, it has been widely utilized in mesothelioma research due to its reproducible growth characteristics and defined histological origin. The cell line was established under IRB-approved protocols aimed at generating clinically annotated cancer models, making it particularly valuable for translational studies linking in vitro behavior with patient disease characteristics. Phenotypically, NCI-H2052 displays epithelial morphology, a feature consistent with the epithelioid subtype of mesothelioma. The cells grow as adherent monolayers in vitro and are maintained in RPMI-1640 medium supplemented with 10% fetal bovine serum. Genomic profiling has identified alterations characteristic of mesothelioma, including dysregulation of pathways involving CDKN2A and NF2, though NCI-H2052 specifically retains wild-type BAP1 and displays relatively low mutation burden compared to other mesothelioma models. These molecular traits position NCI-H2052 as a reference model for studying mesothelioma pathogenesis and therapeutic response, especially in contexts excluding BAP1-driven phenotypes. This cell line has been incorporated into comprehensive pharmacogenomic and transcriptomic datasets, where it contributes to the comparative analysis of mesothelioma subtypes and therapeutic sensitivities. It has shown moderate responsiveness to agents targeting the PI3K/mTOR axis and has been used in high-throughput screening platforms to identify potential synthetic lethal interactions and novel treatment approaches. Due to its molecular profile and origin, NCI-H2052 remains a cornerstone in mesothelioma drug development and molecular characterization studies. |
|---|---|
| Organism | Human |
| Tissue | Pleural effusion |
| Disease | Pleural sarcomatoid mesothelioma |
| Synonyms | H2052, H-2052, H2052_MM, NCIH2052 |
Characteristics
| Age | 65 years |
|---|---|
| Gender | Male |
| Ethnicity | Caucasian |
| Morphology | Epithelial |
| Cell type | Epithelial like |
| Growth properties | Adherent |
Regulatory Data
| Citation | NCI-H2052 (Cytion catalog number 305836) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_1518 |
Biomolecular Data
| Mutational profile | Mutation: Gene deletion, CDKN2A, Homozygous. Gene deletion, LATS2, Homozygous. Mutation, NF2, Simple, p.Arg341Ter (c.1021C>T), Homozygous, RASSF2, Simple, p.Glu294Ter (c.880G>T), Heterozygous, TERT, Simple, c.1-124C>T (c.228C>T) (C228T), Unspecified, Note=In promoter (PubMed=31068700) |
|---|
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Doubling time | 48 hours |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305836-051125 | Certificate of Analysis | 11. Dec. 2025 | 305836 |
-
Related products
Related products