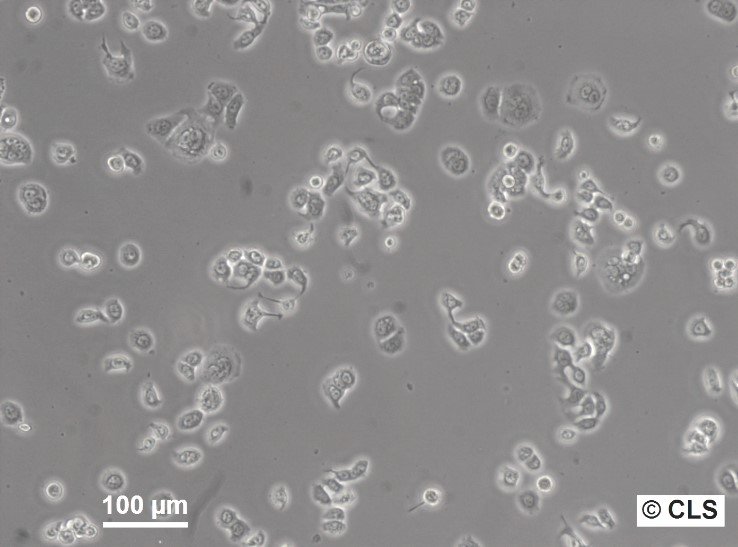
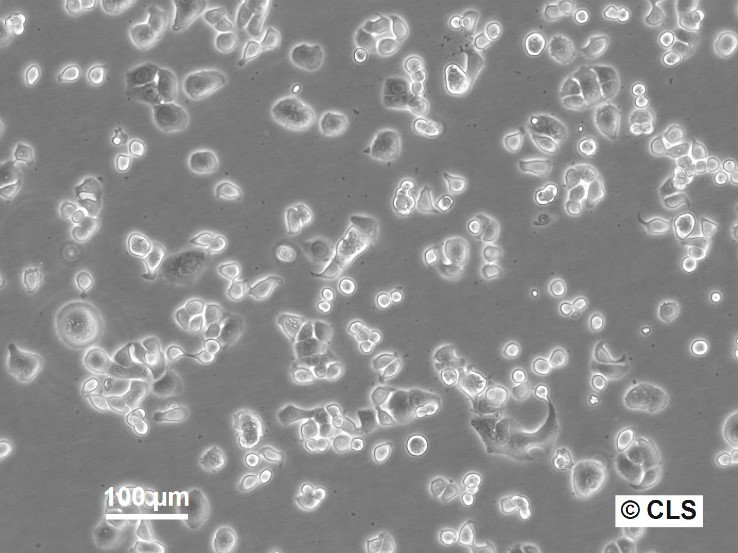
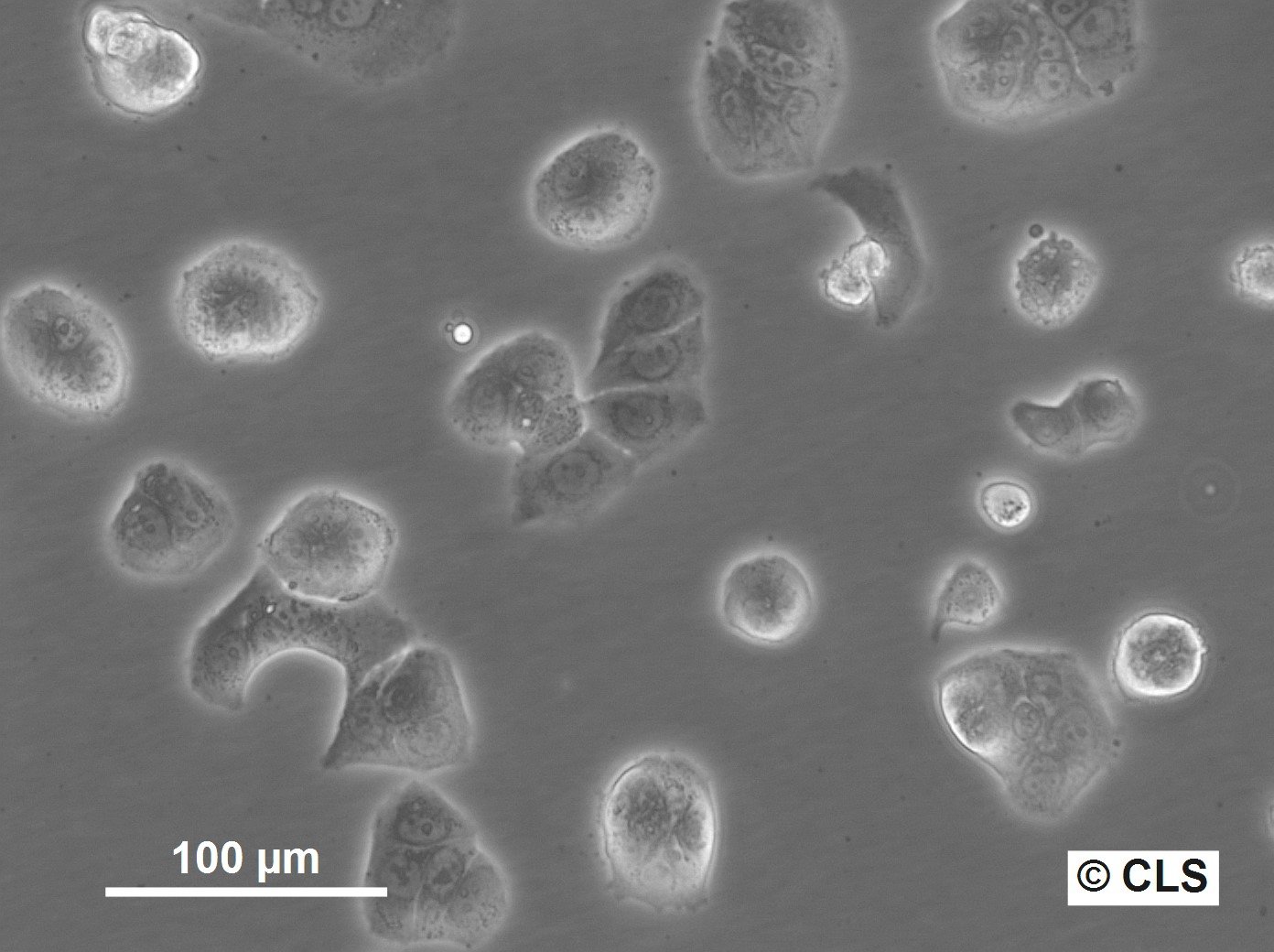
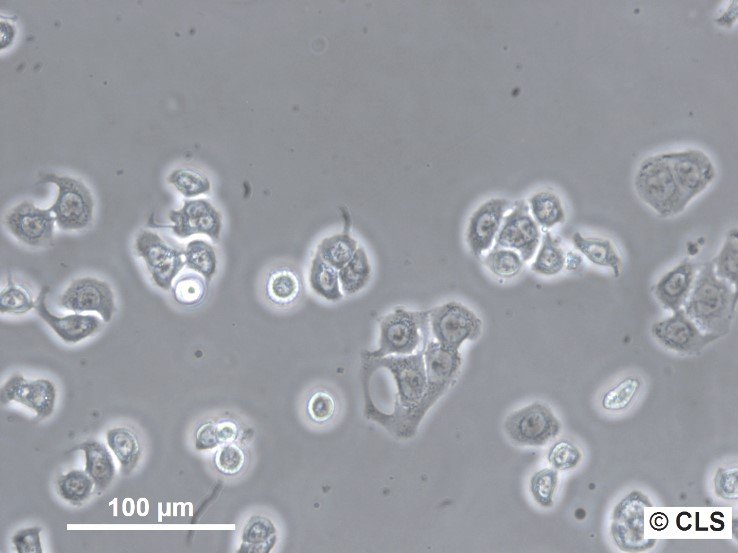
WiDr Cells




















USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | According to Chen TR, 1987, the WiDr cell line is a derivative of the HT-29 cell line. The cells are negative for Colon Antigen 3 expression, but positive for keratin by immunoperoxidase staining. The cells expressed p53 antigen (the p53 produced has a G -> A mutation resulting in Arg -> His at position 273). Growth of WiDr cells is inhibited by tumor necrosis factor alpha (TNF Inhibitors of dihydrofolate reductase are highly cytotoxic to WiDr cells). |
|---|---|
| Organism | Human |
| Tissue | Colon |
| Disease | Adenocarcinoma |
| Synonyms | WiDR, WIDR, WiDr/S, WiDr-TC, WiDrTC, LED-WiDr, Led-WiDr |
Characteristics
| Age | 44 years |
|---|---|
| Gender | Female |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | WiDr (Cytion catalog number 300377) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_2760 |
Biomolecular Data
| Receptors expressed | Epidermal growth factor (EGF) |
|---|---|
| Protein expression | CEA positive |
| Antigen expression | HLA A24, A32, B15, B18 |
| Isoenzymes | PGM1, 1-2, PGM3, 1-2, G6PD, B, ES-D, 1, PEP-D, 1, 6PGD, A |
| Tumorigenic | Yes, in nude mice |
| Products | Carcinoembryonic antigen (CEA) 118 ng/106 cells/10 days, Colon Specific Antigen (CSAp), transforming growth factor beta, keratin |
Handling
| Culture Medium | DMEM:Ham's F12 (1:1), w: 3.1 g/L Glucose, w: 2.5 mM L-Glutamine, w: 15 mM HEPES, w: 0.5 mM Sodium pyruvate, w: 1.2 g/L NaHCO3 (Cytion article number 820400a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 |
| Fluid renewal | 1 to 2 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300377-618acf | Certificate of Analysis | 23. May. 2025 | 300377 |
