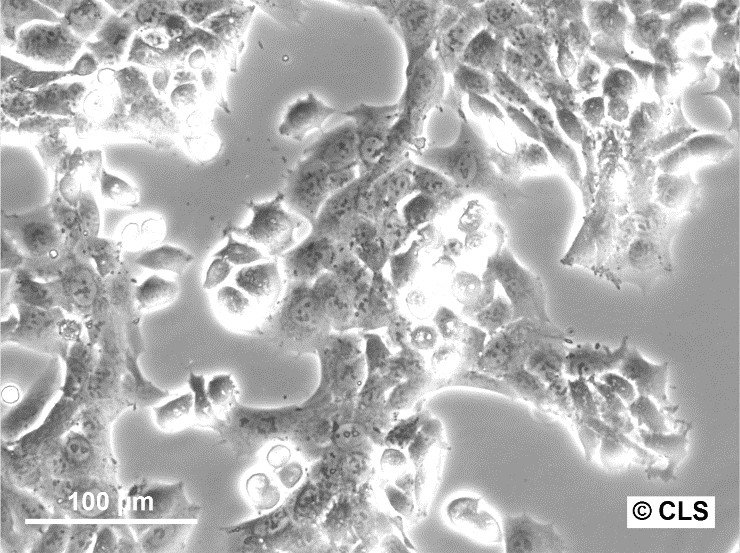
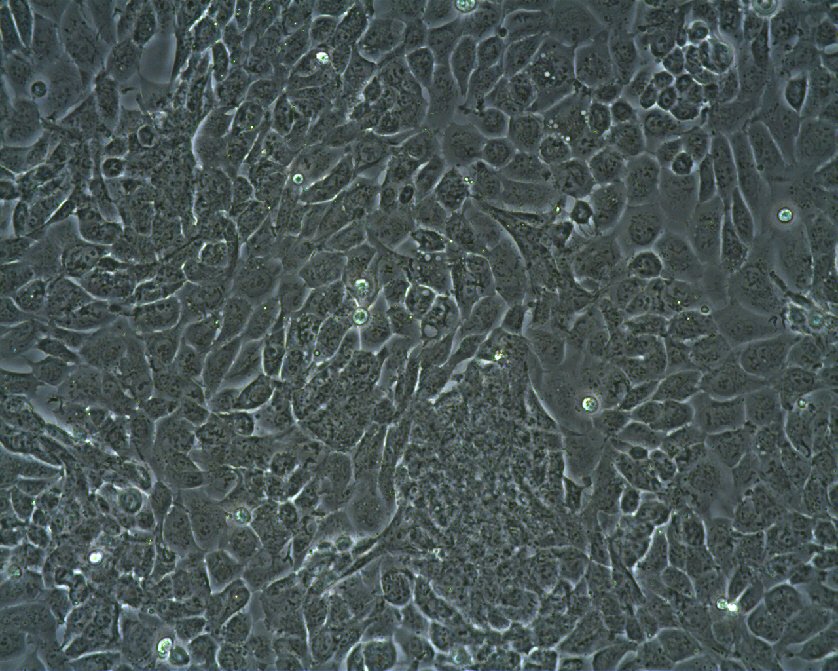
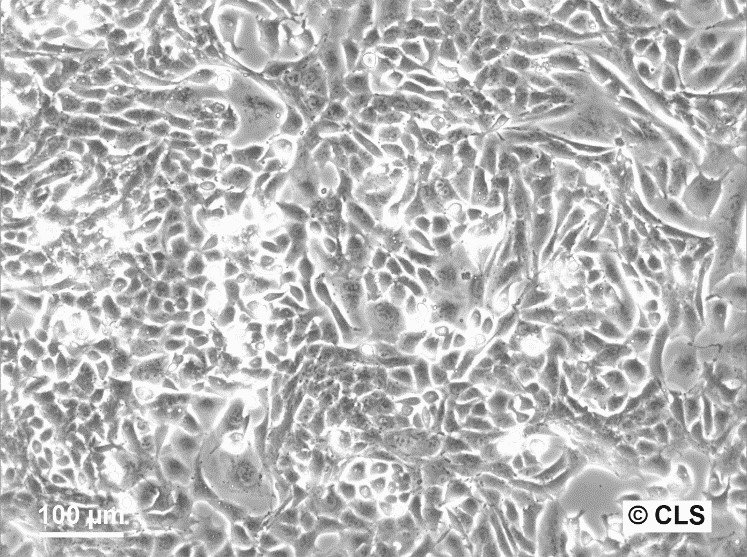
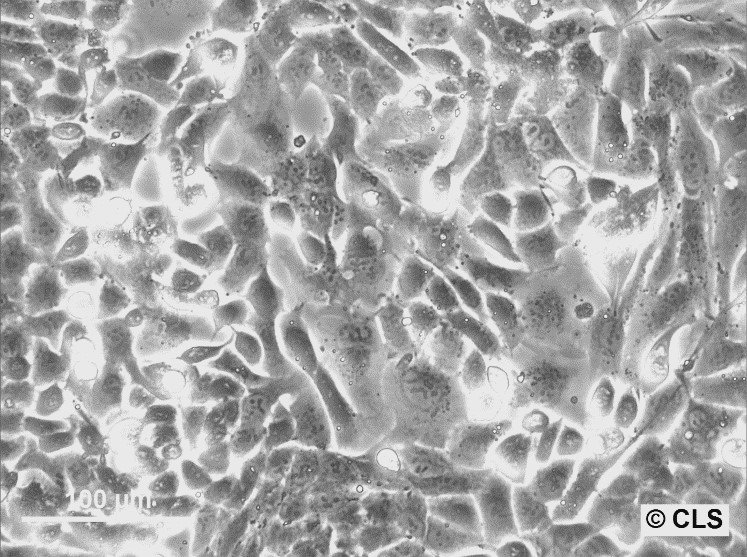
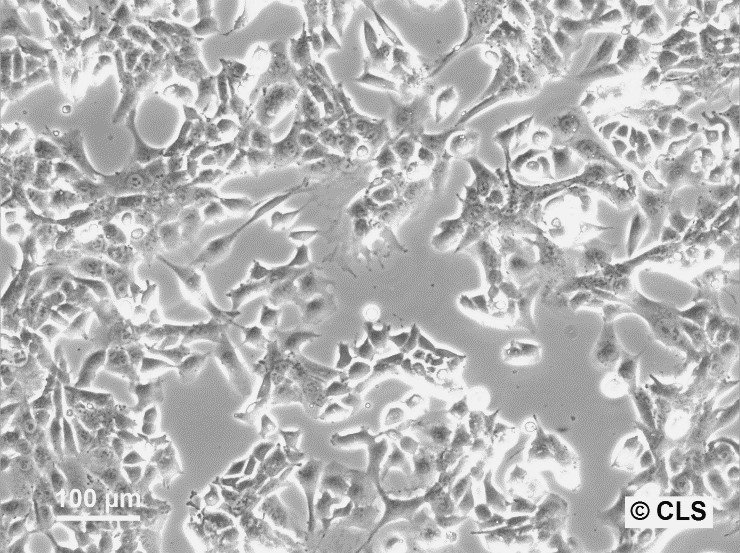
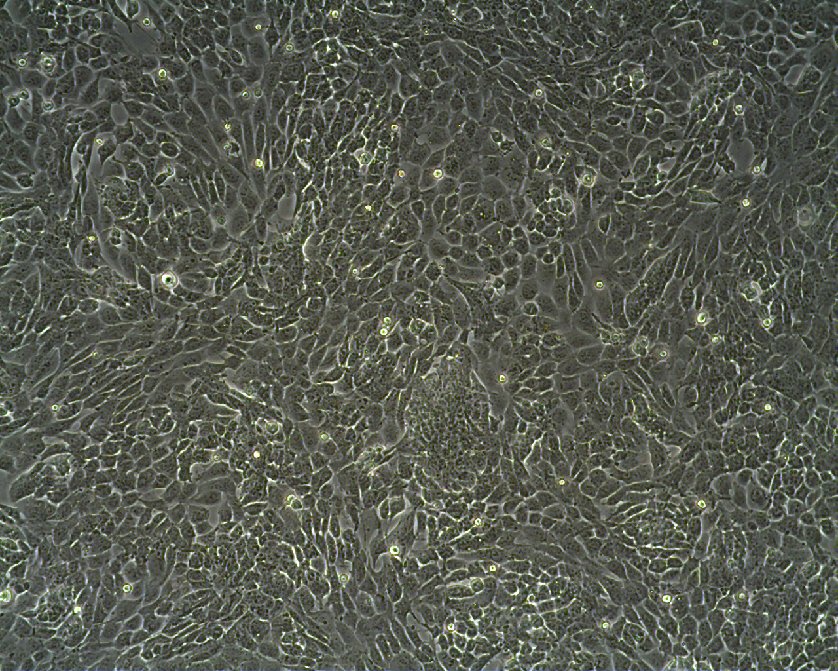
WT-CLS1 Cells


















USD$650.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The WT-CLS1 cell line was established from a primary Wilms’ tumor by CLS in 1998. However, the cells have rhabdoid characteristics, as demonstrated by E. Kunce Stroup et al. in 2017. WT-CLS1 cells are sensitive to miR-16, as a result cyclin D genes expression decreases. In addition, the cells showed a unique resistance to IGF1R inhibition, in contrast to true Wilm’s tumor cells. |
|---|---|
| Organism | Human |
| Tissue | Kidney |
| Disease | Rhabdoid tumor |
| Synonyms | CLS1 |
Characteristics
| Age | 5 years |
|---|---|
| Gender | Female |
| Ethnicity | Caucasian |
| Morphology | Epithelial-like |
| Cell type | B lymphoblast |
| Growth properties | Monolayer, adherent |
Regulatory Data
| Citation | WT-CLS1 (Cytion catalog number 300379) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_5904 |
Biomolecular Data
| Tumorigenic | Yes, in nude mice. Forms tumor with small cells consistent with Wilms' tumor (xenografts may not represent Wilm’s tumors completely, see E. Kunce Stroup 2017) |
|---|---|
| Viruses | HIV-1: negative, HBV: negative, HCV: negative |
| Mutational profile | WT1 mutation status: wild type, CTNNB1 mutation status: wild type, no LOH. |
Handling
| Culture Medium | IMDM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 25 mM HEPES, w: 1.0 mM Sodium pyruvate, w: 3.024 g/L NaHCO3 (Cytion article number 820800a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 to 3 x 105 cells/cm2 |
| Fluid renewal | Every 3 to 4 days |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300379-614SF | Certificate of Analysis | 23. May. 2025 | 300379 |
