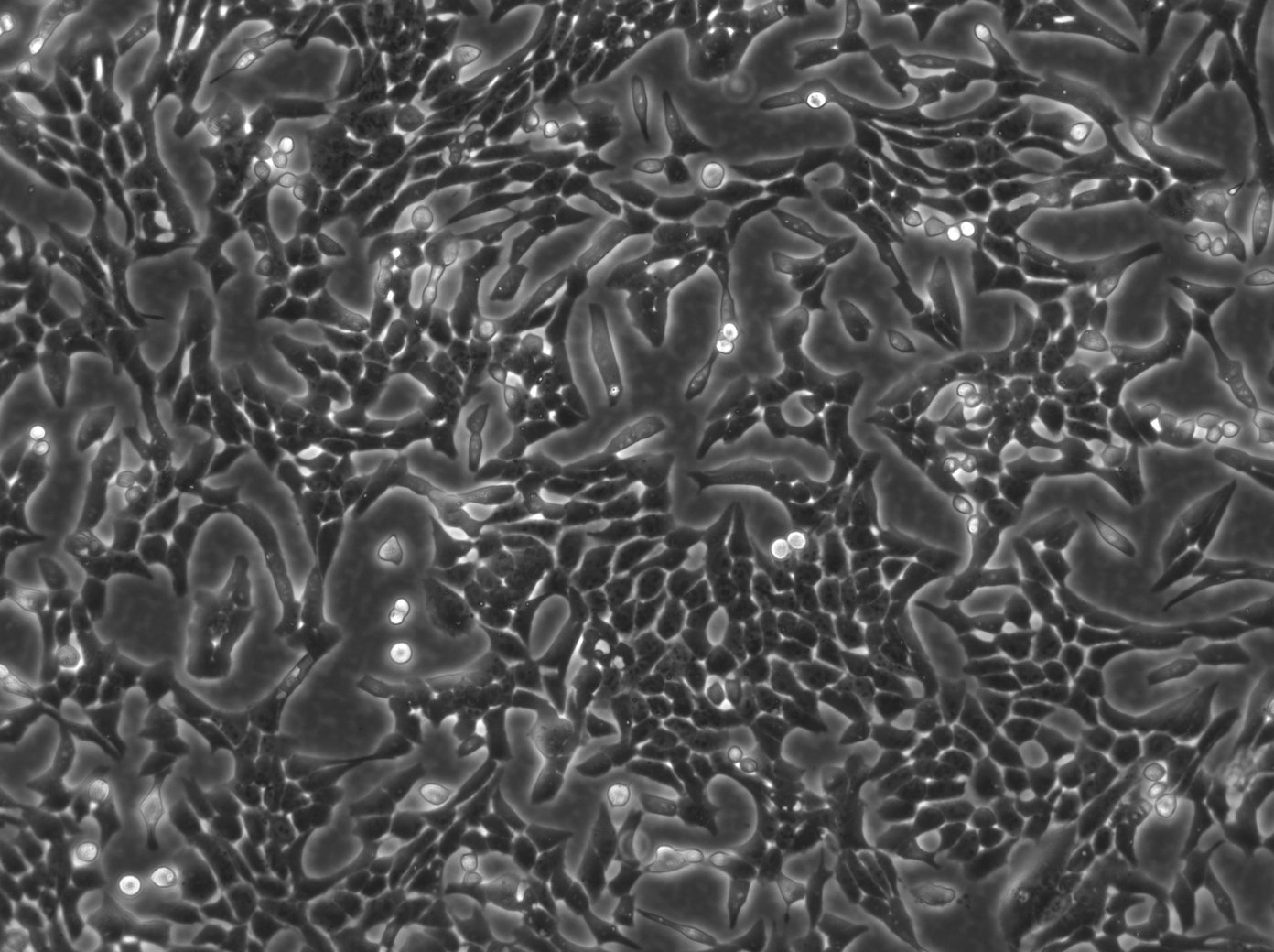
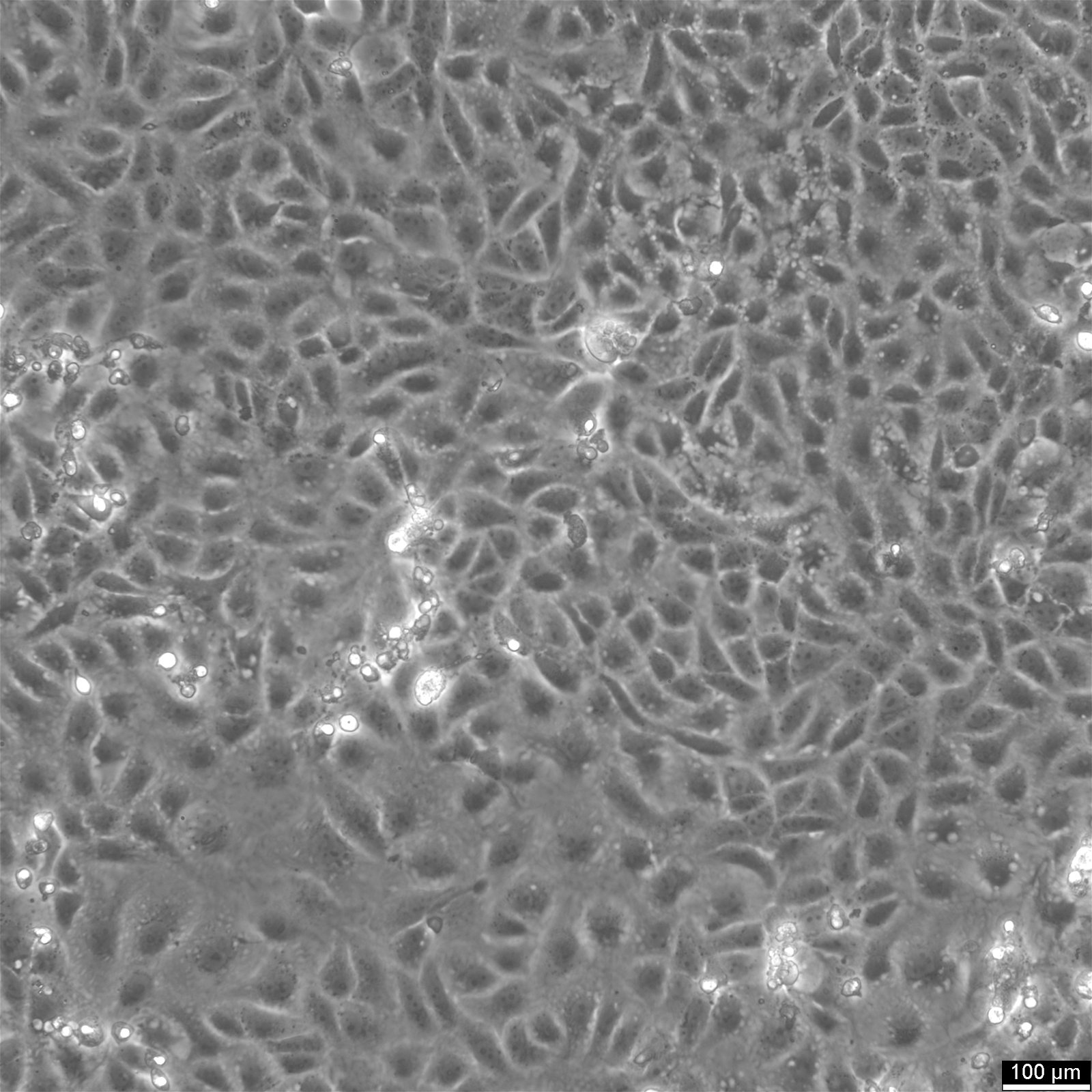
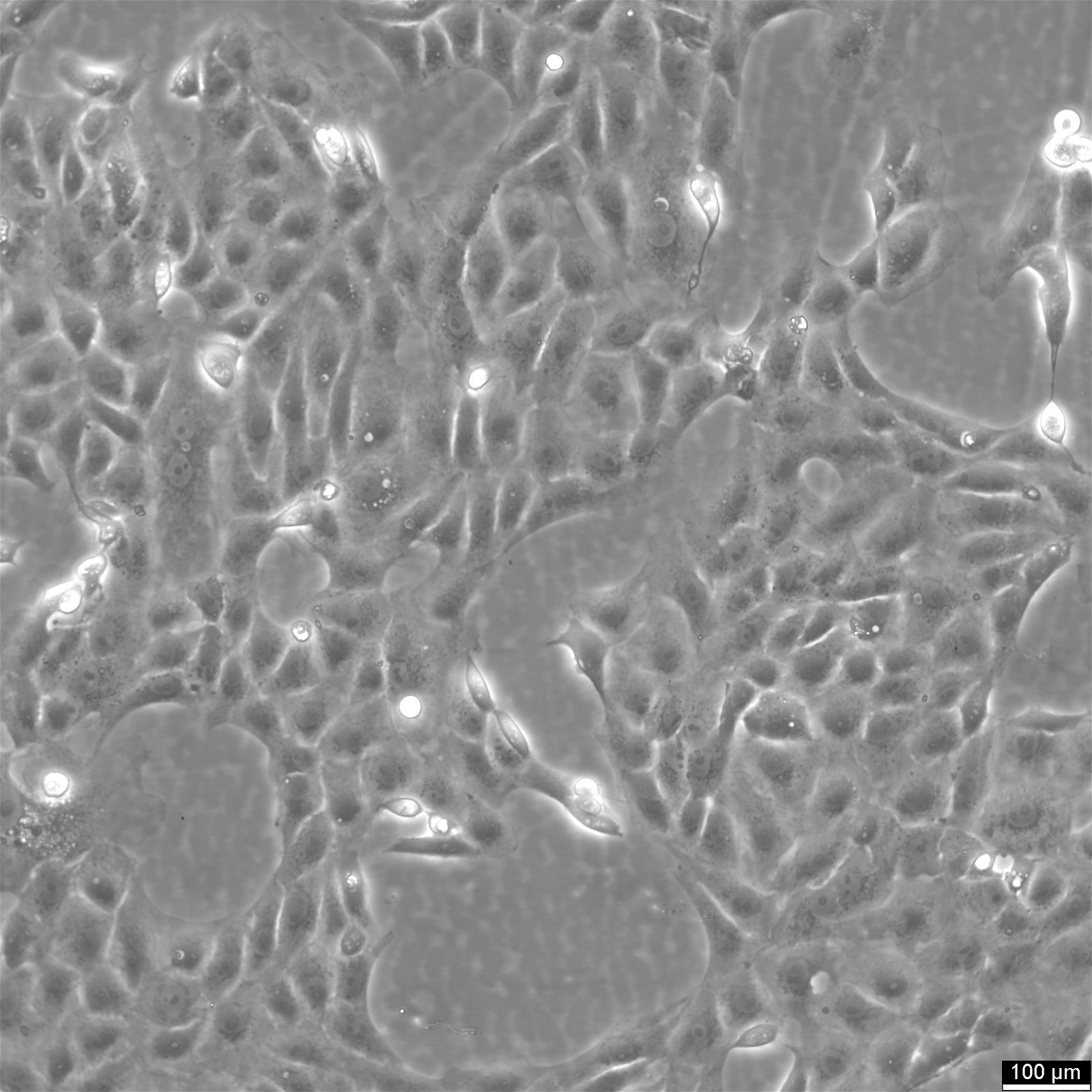
Vero E6 Cells






USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
Insights on the Vero E6 cell line
| Description | Vero E6 cells, also known as Vero C1008 or Vero 76 clone E6, are a continuous line of epithelial cells derived from the kidney of the African green monkey, Chlorocebus sabaeus. The Vero clone E6, a subline of Vero cells, is particularly noted for its utility in virology research due to its high susceptibility to a wide range of viruses, including coronaviruses like SARS-CoV and SARS-CoV-2, the Ebola virus, and the Zika virus. The cell line is crucial in the production of vaccines, such as those for Japanese Encephalitis vaccine, due to their capacity for virus culture and isolation. The cells have played a pivotal role in the development of COVID therapeutics, including the testing of the polymerase inhibitor remdesivir. With their ability to support the replication of a variety of viruses, Vero E6 cells facilitate compound screening and the evaluation of antiviral efficacy. Their role in clinical trials extends to the assessment of anti-inflammatory drugs like dexamethasone and the study of gene products like the P-glycoprotein (pgp protein) encoded by the pgp gene. Vero E6 cells lack the interferon-β gene, which partly explains their high susceptibility to viral infections; this deficiency prevents them from mounting an effective innate antiviral response. In summary, Vero E6 cells are a valuable resource in the field of virology and biomedicine, providing a versatile platform for antiviral screening, the study of replication in Vero, and aiding in the quest for understanding retroviral sequences. |
|---|---|
| Organism | Chlorocebus sabaeus (Green monkey) |
| Tissue | Normal Kidney |
Features
| Age | Adult |
|---|---|
| Morphology | Epithelial |
| Growth properties | Adherent |
Documentation about Vero E6 cells
| Citation | Vero E6 (Cytion catalog number 305008) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9534 |
| CellosaurusAccession | CVCL_0574 |
Handling Vero C1008 cells
Quality control on Vero 76 clone E6 cells
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Doubling time | 22 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305008-200225 | Certificate of Analysis | 23. May. 2025 | 305008 |