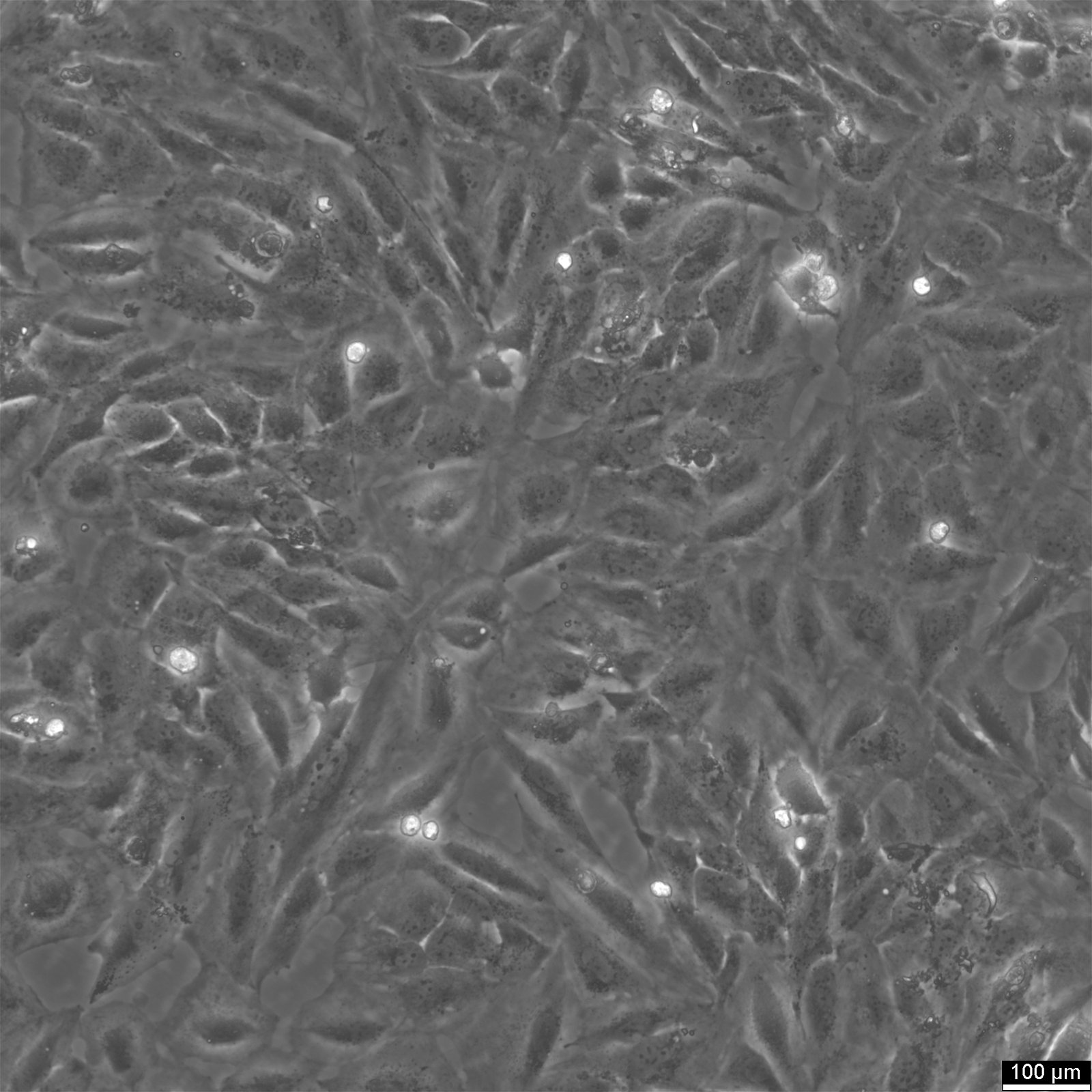
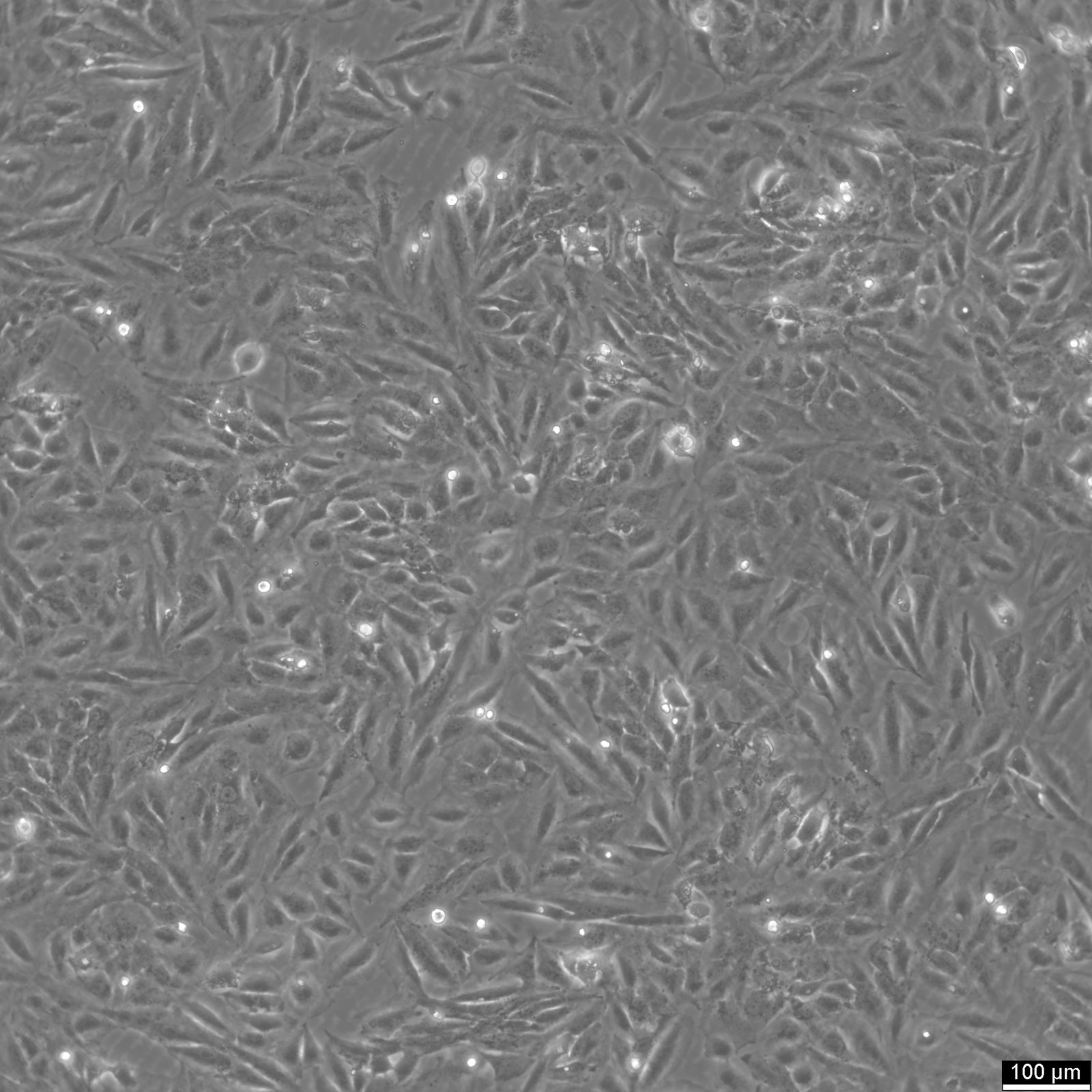
TCCSUP Cells






USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The TCCSUP cell line was established from a Grade IV transitional cell carcinoma (TCC). The cell line was derived from a highly anaplastic carcinoma with characteristics of aggressive malignancy, including rapid proliferation and poor differentiation. Cytogenetic analysis revealed an abnormal karyotype with a lack of a clear modal number, and distinct marker chromosomes were observed throughout its in vitro passages. Morphologically, TCCSUP cells display epithelial-like and fibroblast-like features, consistent with the heterogeneity of aggressive TCC tumors. In vitro, TCCSUP cells exhibit robust growth in monolayer cultures. The cell line has been extensively used in cancer research, particularly in studies of bladder cancer biology and therapeutic response. Notably, TCCSUP cells retain tumor-associated antigens, making them a valuable model for immunological studies and for developing antigen-targeting therapies. Further molecular characterization has highlighted its utility in high-throughput drug screening and genetic studies. TCCSUP cells have been included in large-scale proteomic and genomic analyses, including reverse-phase protein array studies, revealing alterations in signaling pathways such as PI3K/AKT and MAPK. These findings align with the cell line's tumorigenic properties and its relevance as a model for understanding the molecular underpinnings of bladder cancer progression. |
|---|---|
| Organism | Human |
| Tissue | Urinary bladder |
| Disease | Bladder carcinoma |
| Synonyms | TCCSuP, TCC-SUP, TCC Sup |
Characteristics
| Age | 67 years |
|---|---|
| Gender | Female |
| Ethnicity | European |
| Morphology | Epithelial |
| Growth properties | Adherent |
Regulatory Data
| Citation | TCCSUP (Cytion catalog number 305073) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_1738 |
Biomolecular Data
Handling
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Doubling time | 30 to 40 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305073-290224 | Certificate of Analysis | 23. May. 2025 | 305073 |
