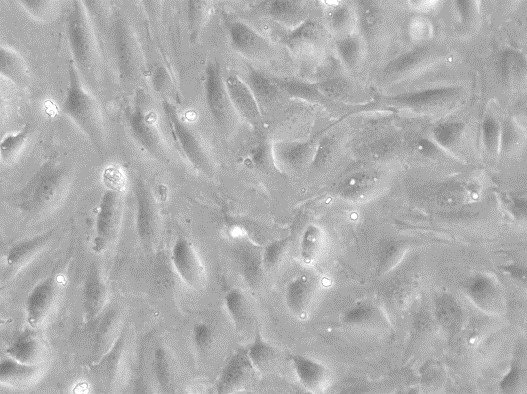
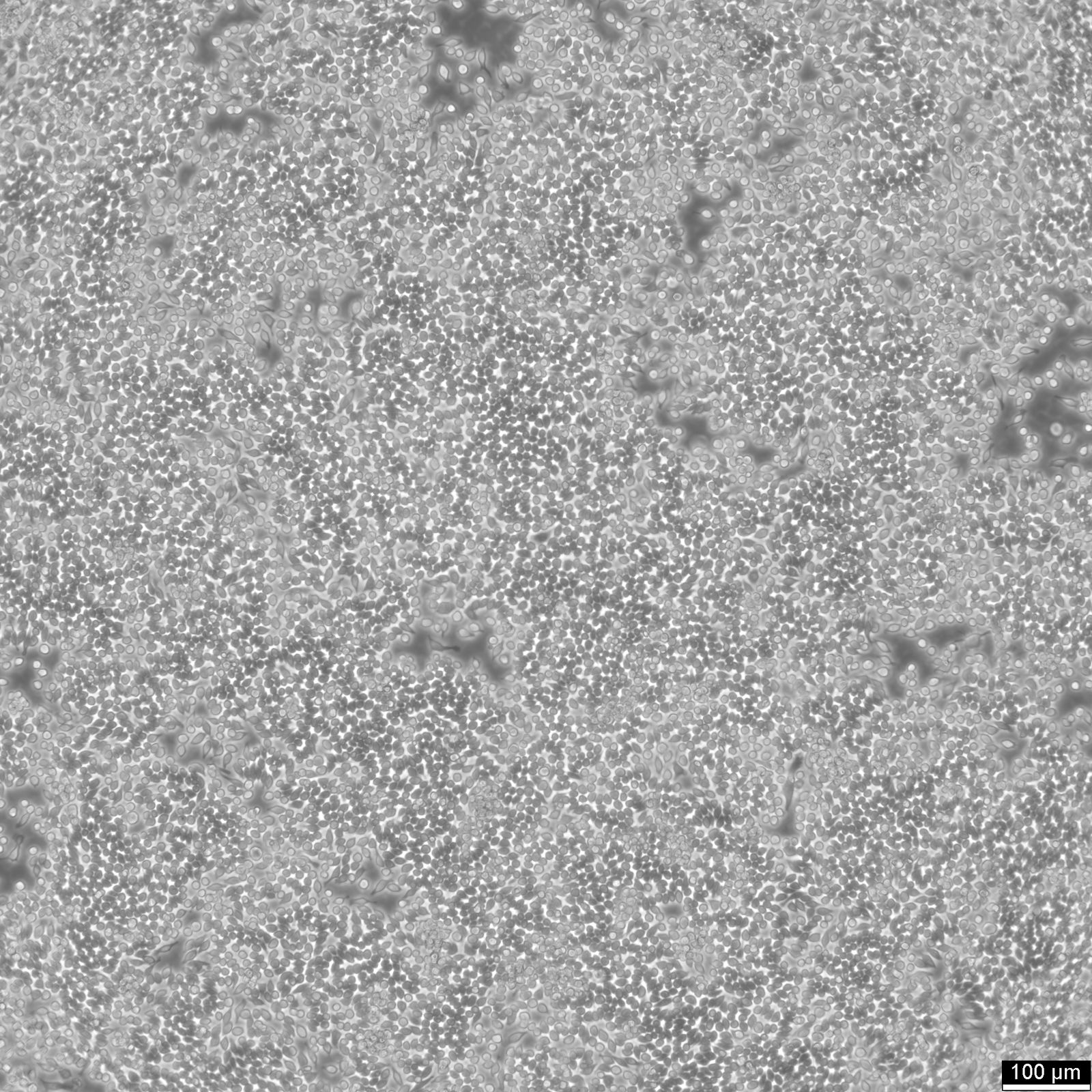
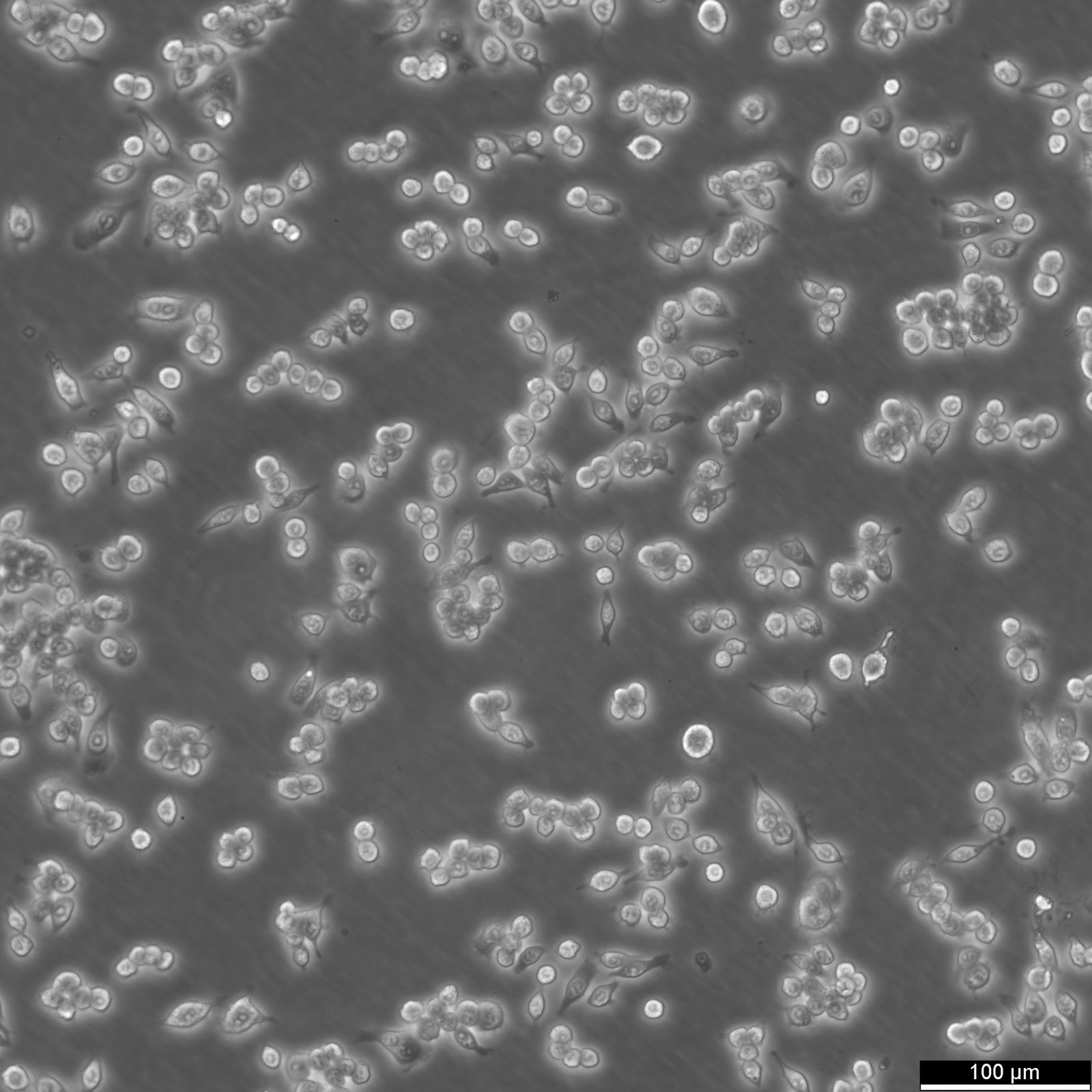
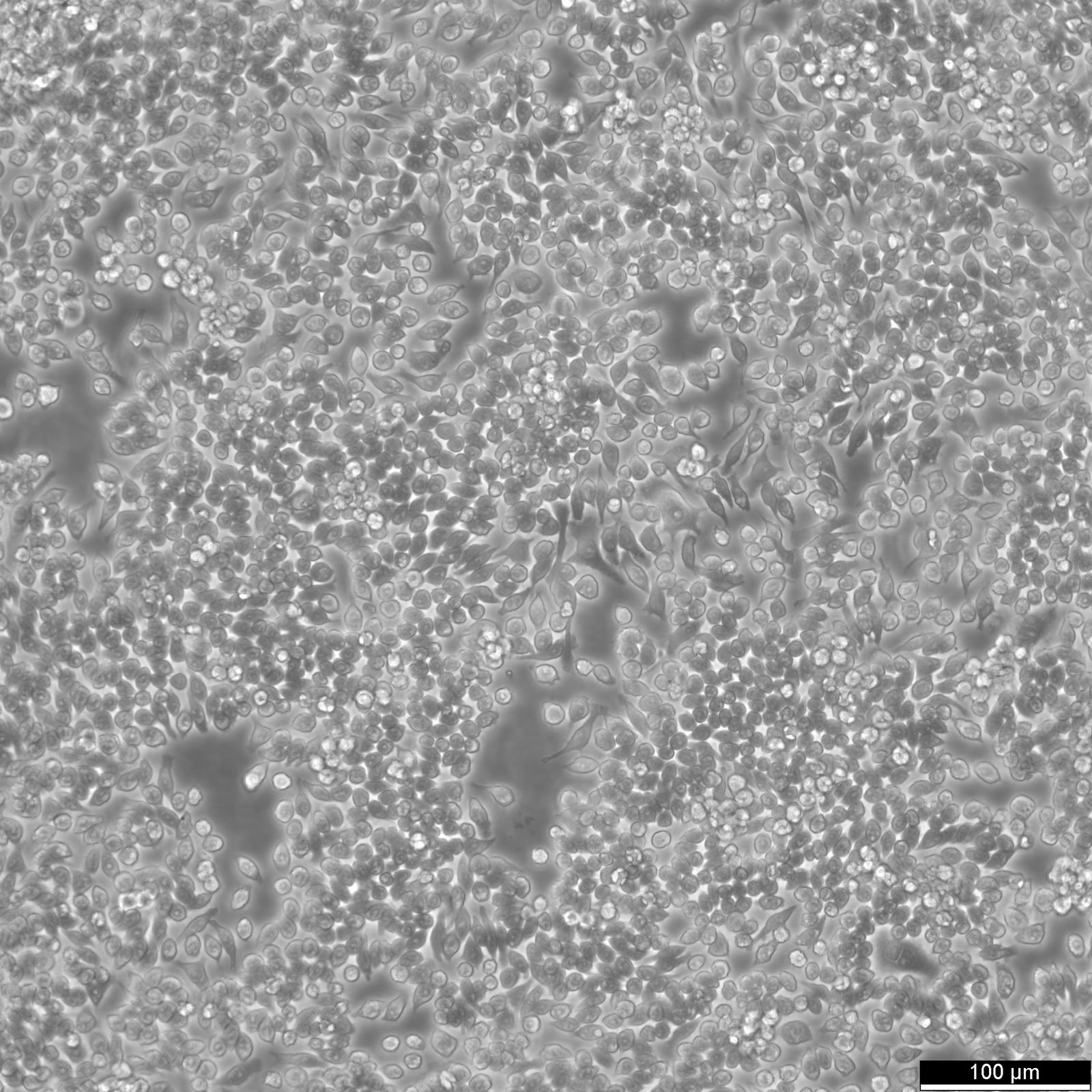
RAW 264.7 Cells





















USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information about the RAW264.7 cell line
| Description | RAW 264.7 cells are a widely used murine macrophage cell line derived from the ascites of a male mouse with a tumor induced by the Abelson murine leukemia virus and are commonly used in immunological and infectious disease research. As an immortalized cell line, RAW264.7 cells are a key model system for studying macrophage biology, including immune responses to pathogens, signal transduction, and gene expression. RAW264.7 cells are particularly valuable for their ability to differentiate into macrophage-like cells. These cells can be polarized into M1 macrophages, associated with inflammatory responses, or M2 macrophages, linked to tissue repair and anti-inflammatory processes. This polarization capacity, along with their ability to perform essential macrophage functions like pinocytosis and phagocytosis, underscores their relevance in studying macrophage biology and the complex interplay between immune responses and pathogens. RAW 264.7 cells are instrumental in studying the immune system's interactions with various factors, including pathogens and bone biology. RAW264.7 cells can be induced to differentiate into osteoclast-like cells under certain conditions, such as exposure to RANKL (Receptor Activator of Nuclear Factor κB Ligand), making them a model for studying certain aspects of osteoclast biology and bone resorption. The RAW264.7 cell line's response to various stimuli, including the induction of pyroptosis, an inflammatory cell death process triggered by factors such as LPS (lipopolysaccharide), is instrumental in dissecting the pathways leading to inflammatory cytokine production. The impact of environmental conditions, such as extracellular glucose levels on cell function and phenotype, offers insights into cellular metabolism and the potential downregulation of inflammatory responses. RAW264.7 cells, with their origins in murine leukemia and their extensive use in immunological research, serve as a crucial tool in advancing our understanding of macrophage biology, immune system-pathogen dynamics, osteoimmunology, and inflammatory responses, highlighting their indispensable role in both basic and applied biomedical research. |
|---|---|
| Organism | Mouse |
| Tissue | Ascites |
| Disease | Leukemia |
| Synonyms | RAW264, RAW2647, RAW264.7, RAW-264.7, Raw 264.7, Raw264.7 |
Properties
| Breed/Subspecies | BALB/c |
|---|---|
| Age | Adult |
| Gender | Male |
| Cell type | Macrophage |
| Growth properties | Adherent |
Documentation
| Citation | RAW 264.7 (Cytion catalog number 400319) |
|---|---|
| Biosafety level | 2 |
| NCBI_TaxID | 10090 |
| CellosaurusAccession | CVCL_0493 |
Genetic profile of the mouse macrophage cell line RAW264.7
| Receptors expressed | Immunoglobulin (Fc), complement (C3) |
|---|---|
| Antigen expression | H-2d |
| Viruses | The cell line was tested and found positive for Reverse Transcriptase (RT) activity from C-Type retroviruses in cell culture supernatant and cell extract. Ectromelia virus (mousepox) may be secreted. |
| Products | Lysozyme |
Cell maintenance techniques
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Strongly adhesive cells, use of cell scraper |
| Doubling time | RAW264.7 cells exhibit a doubling time ranging from 11 to 30 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 4 x 104 cells/cm2 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality control
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 400319-140225 | Certificate of Analysis | 23. May. 2025 | 400319 |
| 400319-180324 | Certificate of Analysis | 23. May. 2025 | 400319 |
| 400319-180823 | Certificate of Analysis | 23. May. 2025 | 400319 |
-
Related products
Related products
THP-1 CellsOrganism Human Tissue The tissue of origin is peripheral blood Disease Leukemia USD$395.00*