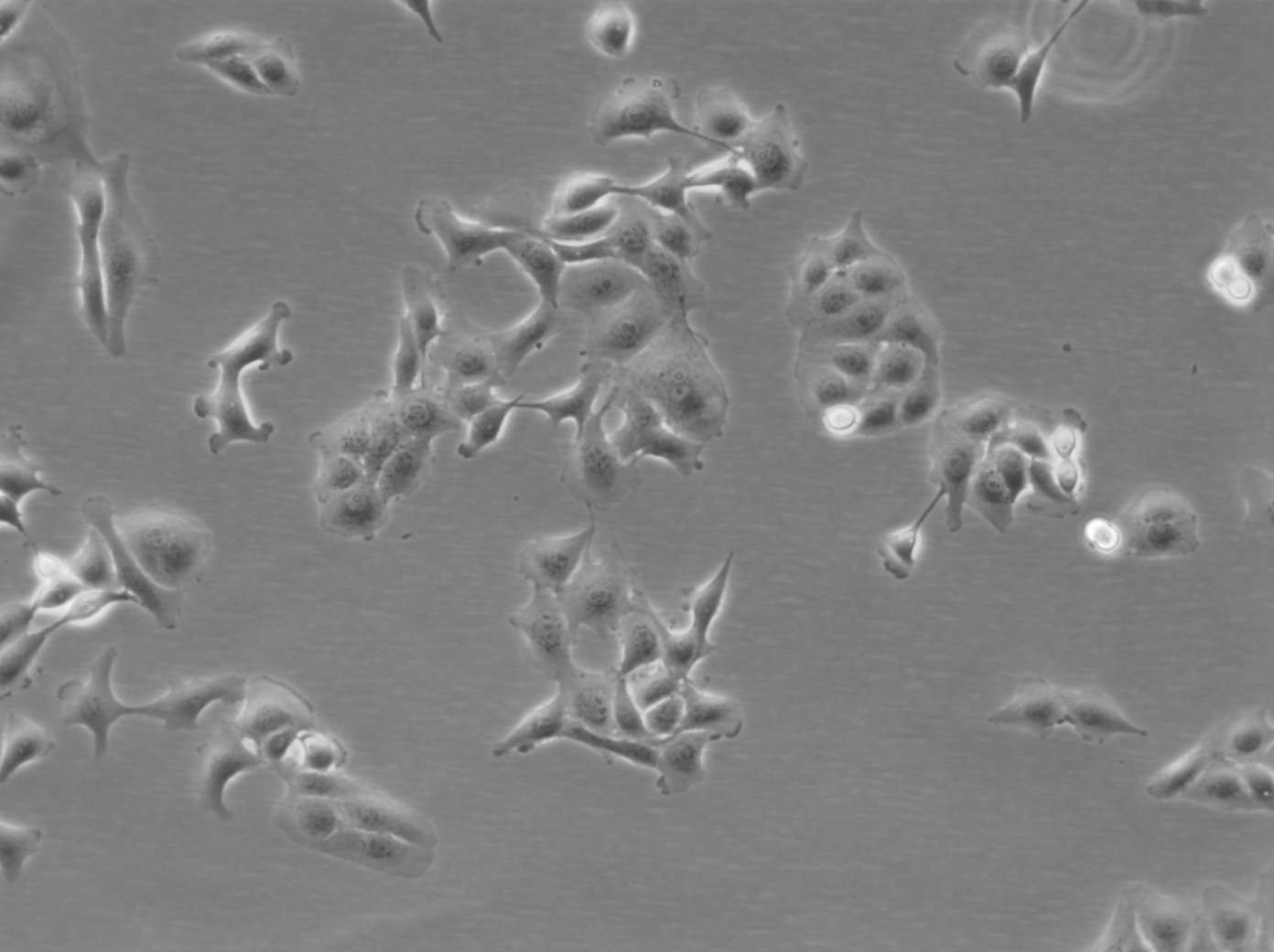
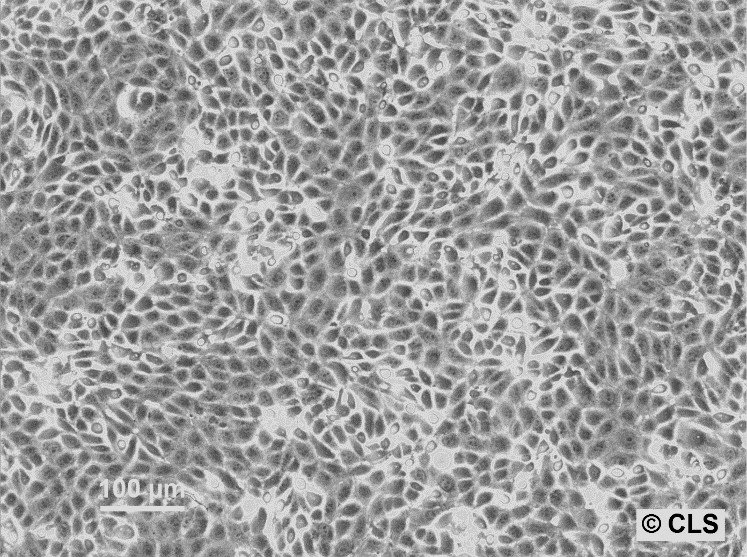
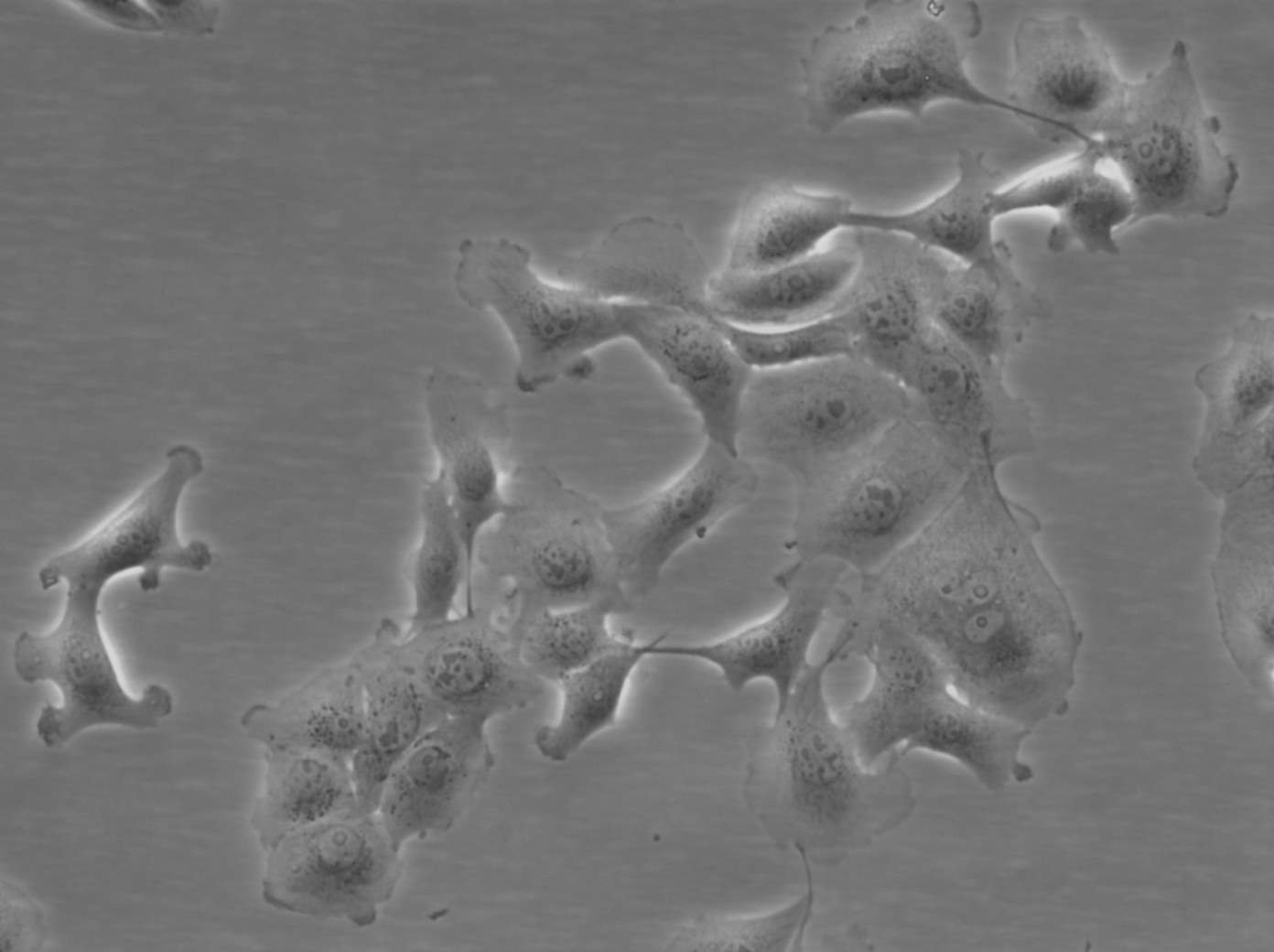
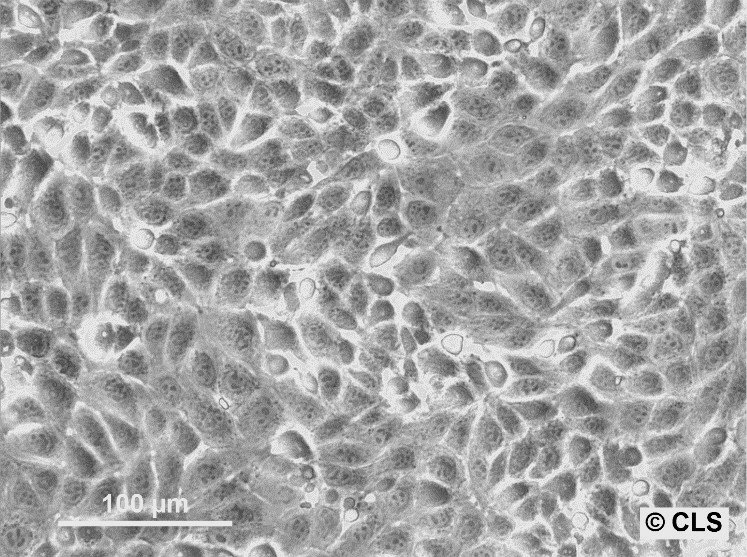
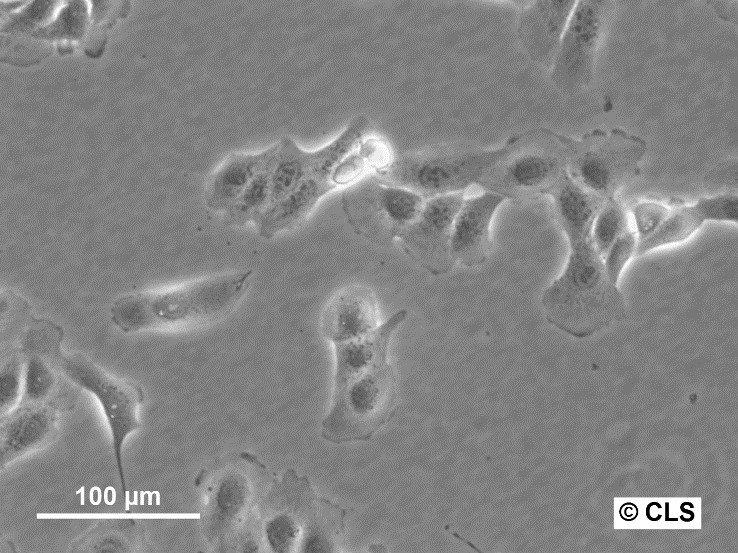
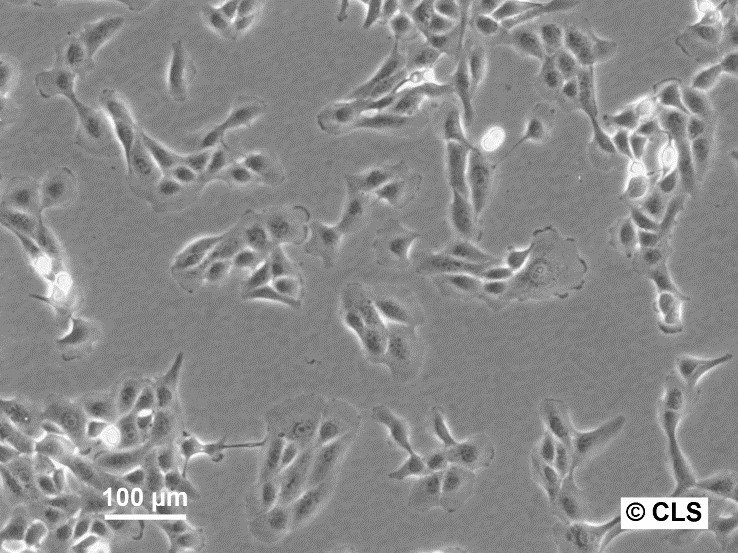
MDBK (NBL-1) Cells


















USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | MDBK cells, short for Madin-Darby Bovine Kidney cells (also known as NBL-1), are an exceptional biological resource derived from the kidneys of apparently healthy adult Bos taurus, specifically male individuals. These cells grow adherently and possess an epithelial-like morphology. One of the remarkable applications of MDBK cells lies in their ability to facilitate in vitro studies on the expression of Eimeria bovis-derived antigens on the host cell surface membrane. With an average doubling time ranging from 24 to 35 hours, MDBK cells exhibit a moderate proliferation rate. The establishment of the MDBK cell line dates back to February 18, 1957, when S.H. Madin and N.B. Darby successfully derived it from the kidney of a healthy adult steer. Since then, these cells have become a cornerstone in biological research, enabling numerous breakthroughs in various scientific fields. The karyotype analysis of MDBK cells reveals a modal chromosome number of 51, indicating a hypodiploid state. Within the cell population, the hypodiploid condition manifests as a stemline chromosome number of 2n = 60, with a 2S component occurring in approximately 5% of the cells. Moreover, 11-14 marker chromosomes are typically present, comprising a combination of metacentric, submetacentric, and acro-telocentric chromosomes. Notably, the x chromosome appears monosomic, while no HSR chromosomes or DM's (double minutes) are observed. MDBK cells exhibit an array of applications in the realm of biological research. Their utility extends to 3D cell culture, enabling scientists to recreate complex tissue-like structures for advanced studies. Furthermore, MDBK cells are invaluable in high-throughput screening, facilitating the rapid and efficient screening of compounds or agents for various purposes. Additionally, these cells play a crucial role in toxicology studies, essential for evaluating the safety and potential adverse effects of substances on living organisms. |
|---|---|
| Organism | Bovine |
| Tissue | Kidney |
| Synonyms | MDBK (NBL-1), NBL-1, Madin-Darby Bovine Kidney, Madin Darby Bovine Kidney |
Characteristics
| Breed/Subspecies | Bos taurus |
|---|---|
| Age | Adult |
| Gender | Male |
| Morphology | Epithelial-like |
| Growth properties | Monolayer, adherent |
Regulatory Data
| Citation | MDBK (NBL-1) (Cytion catalog number 600396) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9913 |
| CellosaurusAccession | CVCL_0421 |
Biomolecular Data
| Viruses | The line was tested and shown to be free of bovine diarrhoea virus (BVD). |
|---|---|
| Virus susceptibility | The cells are susceptibled to bovine diarrhea virus, vesicular stomatitis (Indiana strain), infectious bovine rhinotracheitis virus, bovine parvovirus, bovine adenovirus I and III, and parainfluenza virus 3. |
| Virus resistance | Poliovirus 2 |
| Reverse transcriptase | Negative |
| Products | Keratin |
Handling
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 |
| Fluid renewal | Every 3 days |
| Post-Thaw Recovery | Fast |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 600396-090123 | Certificate of Analysis | 21. Jul. 2025 | 600396 |
