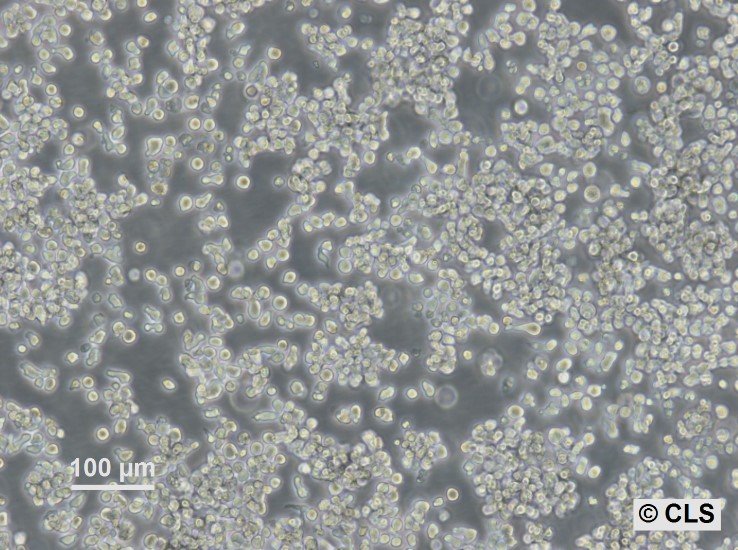
HuT-78 Cells































USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The HuT-78 cell line is a human T-cell lymphoma line derived from a patient with Sézary syndrome, a leukemic variant of cutaneous T-cell lymphoma (CTCL). These cells are characterized by their mature T-helper phenotype, expressing CD4 and lacking CD8 surface markers, consistent with their origin from a malignant T-cell population. The HuT-78 cells are particularly significant in studies of T-cell biology, immune response, and lymphoma, offering insights into the molecular and cellular mechanisms underlying T-cell leukemias and lymphomas. HuT-78 cells exhibit a range of abnormal karyotypes, including complex chromosomal rearrangements and aneuploidy, which are commonly associated with their malignant phenotype. These cells are responsive to mitogenic stimulation, which can be utilized in research involving T-cell activation and signaling pathways. Additionally, HuT-78 cells are sensitive to various chemotherapeutic agents, making them a valuable model for testing anti-cancer drugs, particularly those targeting T-cell lymphomas. Researchers also use HuT-78 cells to study the interactions between lymphoma cells and the immune system, providing a better understanding of immune evasion mechanisms. This cell line is cultured in suspension, requiring specific conditions to maintain viability and growth. HuT-78 cells are vital in advancing the understanding of CTCL pathogenesis and in the development of potential therapeutic strategies targeting malignant T-cells. |
|---|---|
| Organism | Human |
| Tissue | Blood |
| Disease | Mycosis fungoides and Sezary syndrome |
| Synonyms | Hut 78, HUT 78, HuT 78, HUT-78, HuT78, Hut78, HUT78, NCI-H78 |
Characteristics
| Age | 53 years |
|---|---|
| Gender | Male |
| Ethnicity | Caucasian |
| Morphology | Round cells |
| Cell type | T lymphoblast |
| Growth properties | Suspension |
Regulatory Data
| Citation | HuT-78 (Cytion catalog number 300338) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0337 |
Biomolecular Data
| Receptors expressed | Interleukin-2 (interleukin 2, IL-2) |
|---|---|
| Protein expression | P53 negative |
| Antigen expression | CD4 |
| Products | Interleukin-2 (interleukin 2, IL-2), tumor necrosis factor alpha (TNF alpha) |
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% heat-inactivated FBS |
| Subculturing | Maintain cultures by periodically adding or replacing the medium. Initiate cultures with a density of 5 x 105 cells/ml and keep the cell concentration within the range of 3 x 105 to 1 x 106 cells/ml for optimal growth. |
| Seeding density | 1 x 105 cells/ml |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | Allow the cells to recover from the freezing process for 24 to 48 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300338-030323 | Certificate of Analysis | 23. May. 2025 | 300338 |
| 300338-130125 | Certificate of Analysis | 23. May. 2025 | 300338 |
