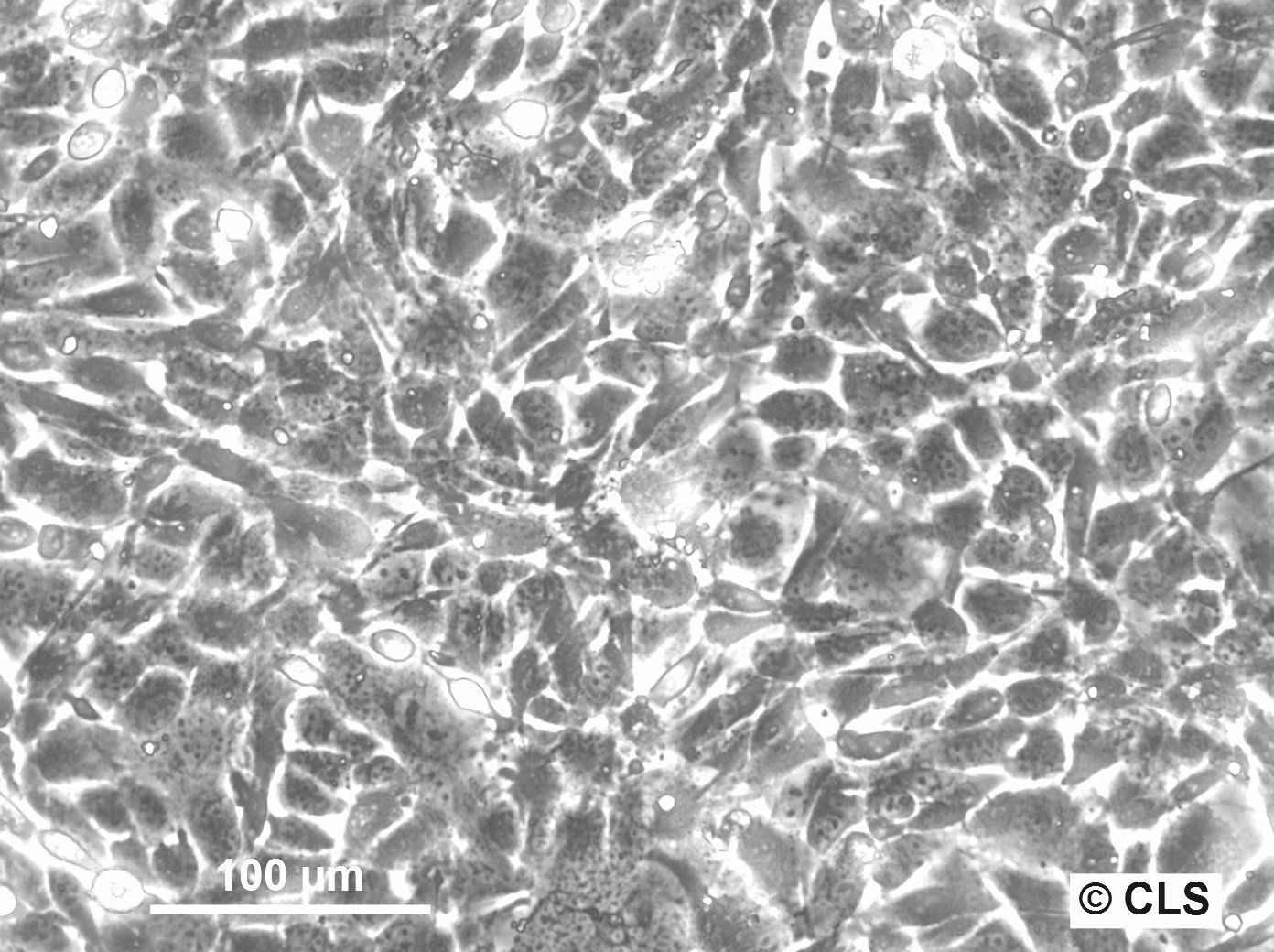
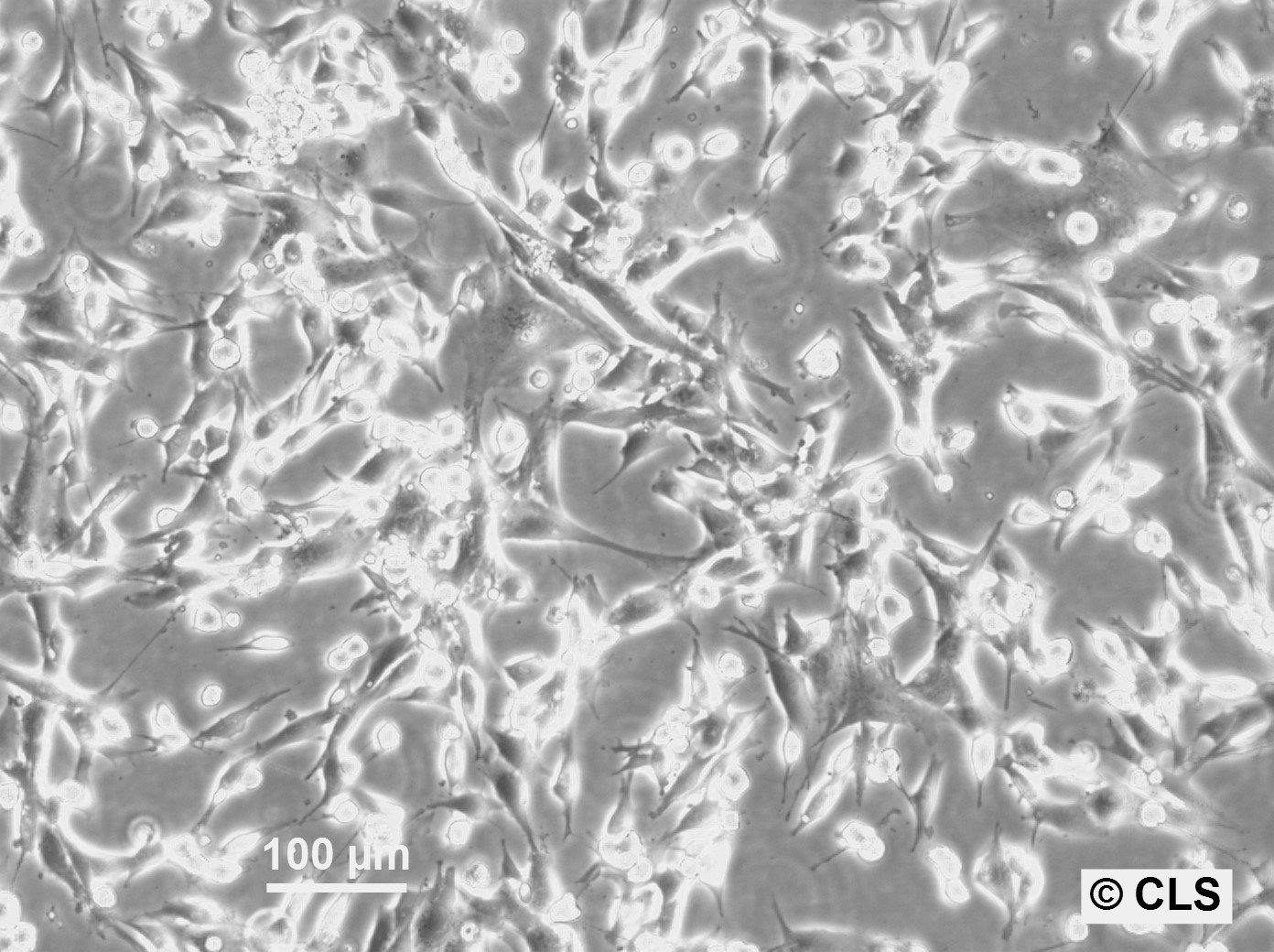
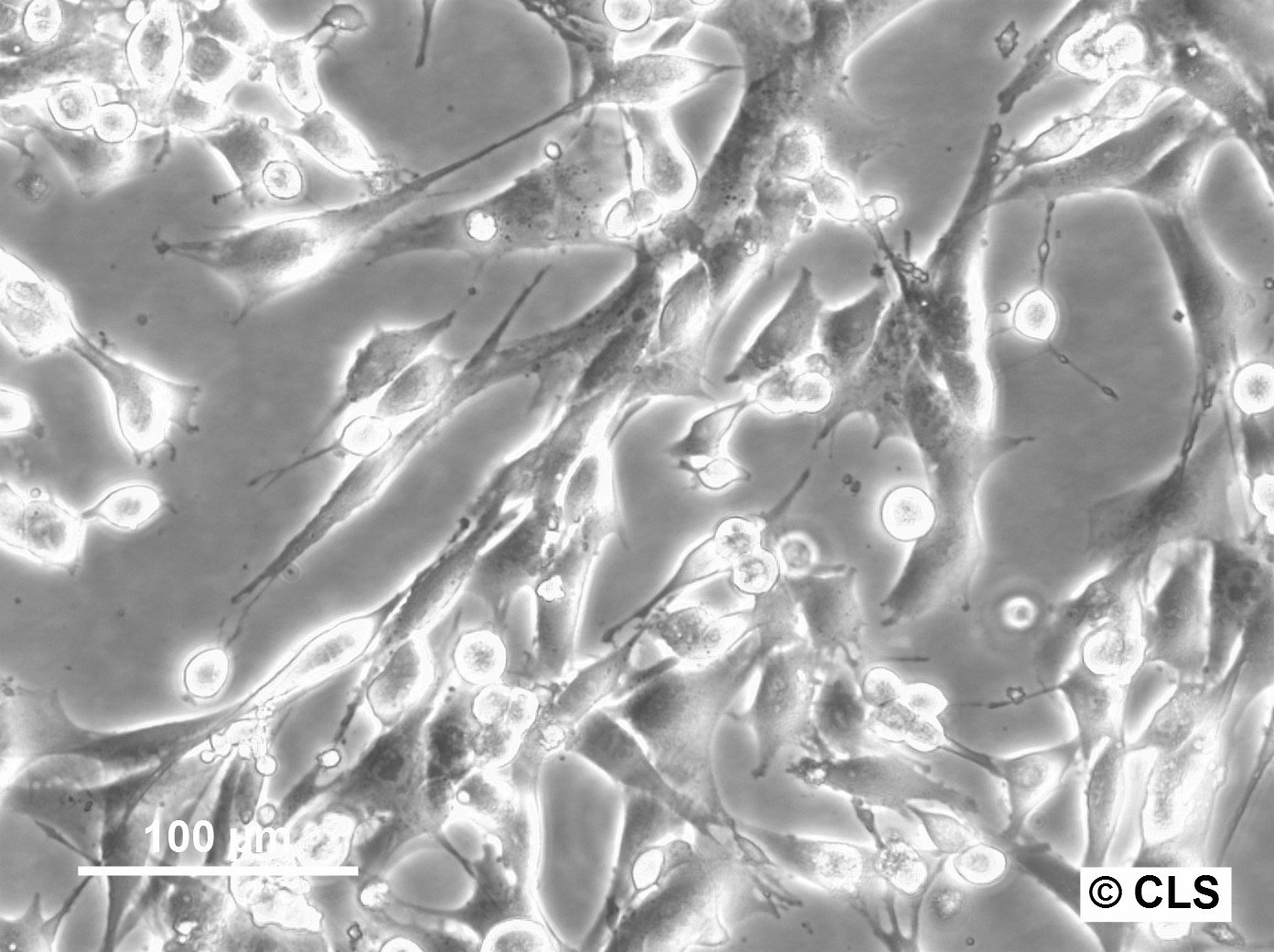
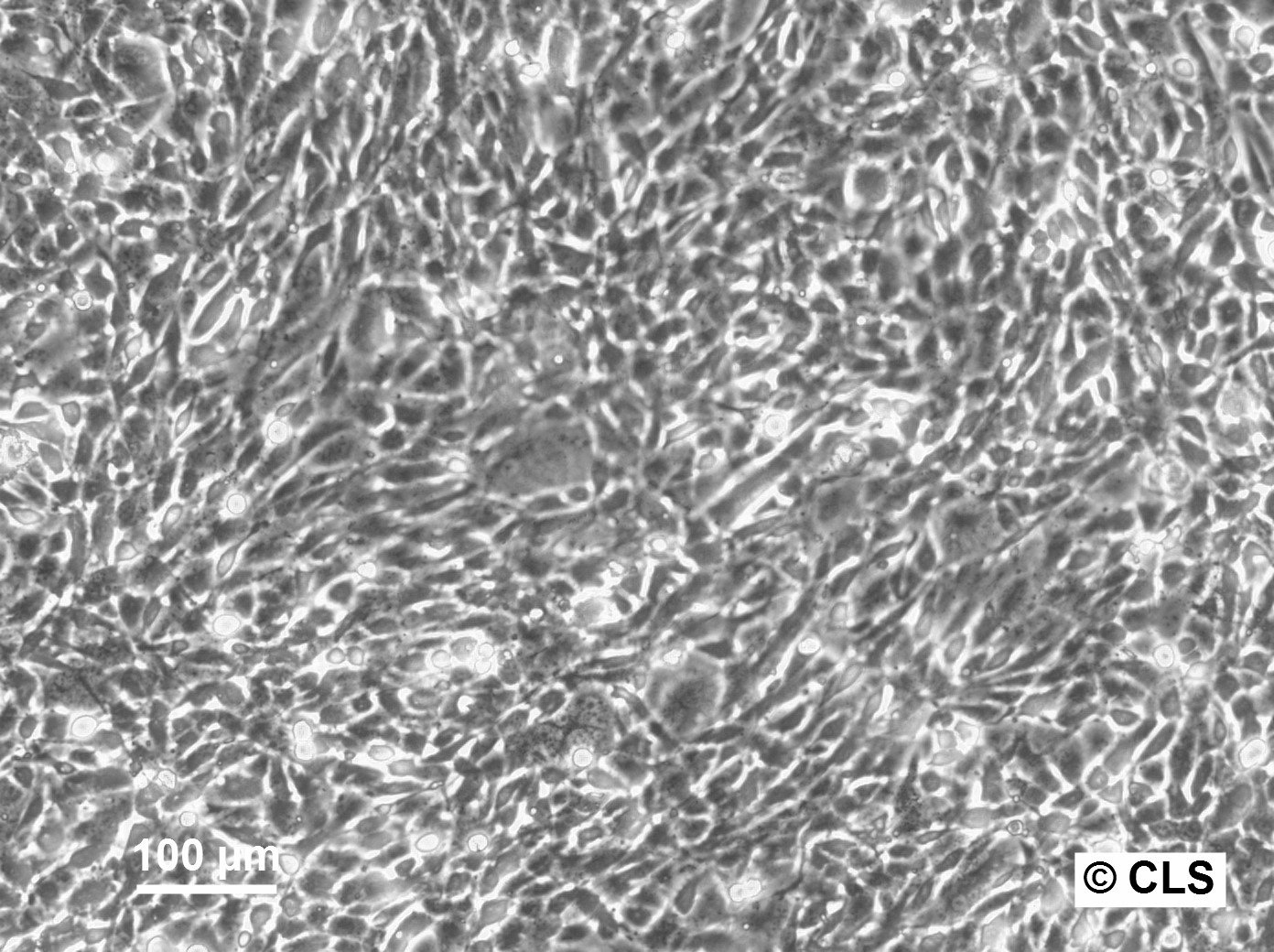
FS-C3H Cells












USD$800.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The FS-C3H cell line, derived from the C3H/HeJ mouse strain, plays a pivotal role in studying host responses to endotoxins, particularly in the context of cancer research. This strain is notable for its resistance to endotoxin due to a specific insensitivity to lipopolysaccharide (LPS), a major component of bacterial endotoxin. This characteristic has made FS-C3H an invaluable model for dissecting the biochemical and genetic pathways involved in immune response regulation. Researchers have extensively used this cell line to examine the dynamics of B lymphocytes and macrophages, focusing on their unique non-responsiveness to LPS, which contrasts with typical immune cell reactions to such stimuli. The non-responsiveness of FS-C3H cells to LPS is attributed to the absence or alteration of a crucial receptor responsible for LPS signal transduction. Studies have shown that despite the non-reactivity to LPS, these cells can be activated through alternative pathways such as protein kinase C (PKC) and tyrosine kinase signaling mechanisms, similar to those activated in LPS-responsive cells. The interaction and regulatory roles of these kinases in signaling pathways highlight complex intra-cellular mechanisms, suggesting that the PKC and tyrosine kinase pathways could compensate for the defective LPS signaling. This observation opens avenues for exploring how tyrosine kinase-modulated phosphorylation affects overall cellular responses in these mice. Continued research on FS-C3H cells is critical to understanding the molecular basis of their hyporesponsiveness to LPS, potentially linked to a genetic defect in the Lpsn gene. By delving into the phosphorylation profiles of these cells compared to LPS responders, scientists aim to unravel the specific molecular defects that lead to altered gene activation and proliferation responses. The isolation and characterization of the gene product responsible for LPS interaction could provide deeper insights into immune system dysfunctions and pave the way for novel therapeutic approaches in treating related immune and inflammatory disorders. |
|---|---|
| Organism | Mouse |
| Tissue | Skin |
| Disease | Fibrosarcoma |
Characteristics
| Breed/Subspecies | C3H |
|---|---|
| Growth properties | Adherent |
Regulatory Data
| Citation | FS-C3H (Cytion catalog number 400418) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 10090 |
| CellosaurusAccession | CVCL_5755 |
Biomolecular Data
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 3.7 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 2 x 104 cells/cm2 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
