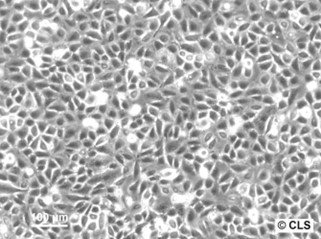
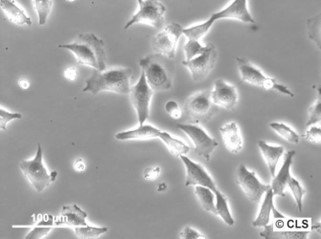
Chang Liver (HeLa) Cells






USD$550.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The Chang Liver cell line, originally believed to be derived from normal human liver tissue, has undergone significant reclassification following advanced genetic profiling. STR PCR DNA profiling techniques have demonstrated that the Chang Liver cell line is indistinguishable from the HeLa cell line, suggesting that it is not derived from hepatocyte cells as previously thought, but rather should be considered a HeLa derivative. This revelation has important implications for researchers using this cell line, emphasizing the need for careful interpretation of experimental results derived from its use. HeLa cells, originally taken from Henrietta Lacks, a Black woman, in the early 1950s, are known for their robust growth and genetic stability in vitro, characteristics likely shared by the Chang Liver cell line given its genetic similarity. This background necessitates that studies employing the Chang Liver cell line in research related to liver function or diseases may need to be re-evaluated or confirmed with additional hepatocyte-specific models. The misidentification also highlights broader issues in cell culture practices, including cross-contamination and mislabeling, underscoring the importance of regular authentication of cell lines used in research settings. |
|---|---|
| Organism | Human |
| Tissue | Liver |
| Disease | Adenocarcinoma |
| Synonyms | Chang-liver, Chang Cells, Chang, CHL |
Characteristics
| Age | 30 years |
|---|---|
| Gender | Female |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | Chang Liver (HeLa) (Cytion catalog number 300139) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0238 |
Biomolecular Data
| Isoenzymes | G6PD, A |
|---|---|
| Tumorigenic | Yes, in Syrian hamsters |
| Viruses | Tested MHV (mouse hepatitis virus) negative |
| Virus susceptibility | Poliovirus 1, 2, 3, adenovirus 3, vesicular stomatitis (Indiana) |
| Reverse transcriptase | Negative |
| Products | Keratin |
Handling
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 will yield in a confluent layer in about 4 days |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300139-713 | Certificate of Analysis | 23. May. 2025 | 300139 |
