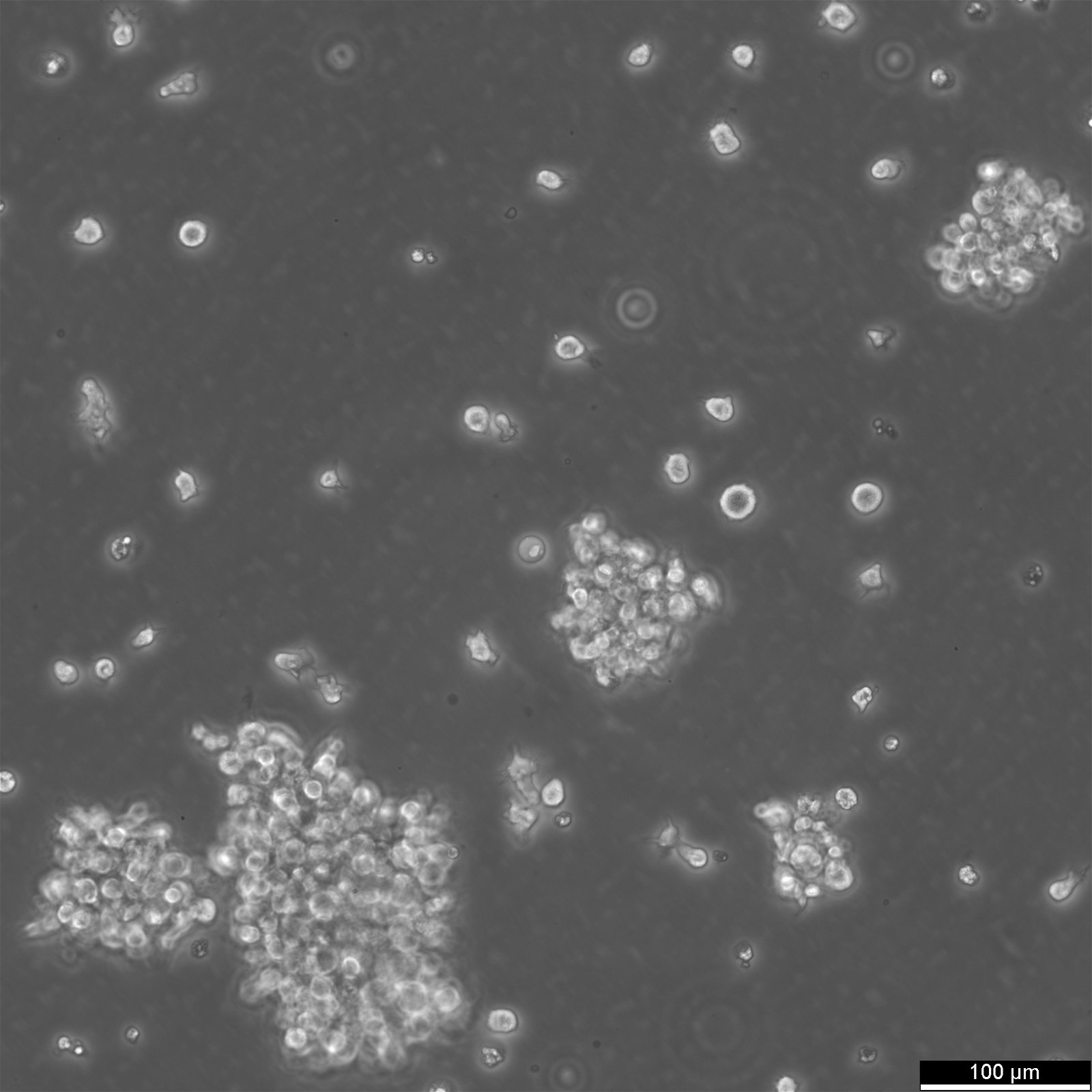
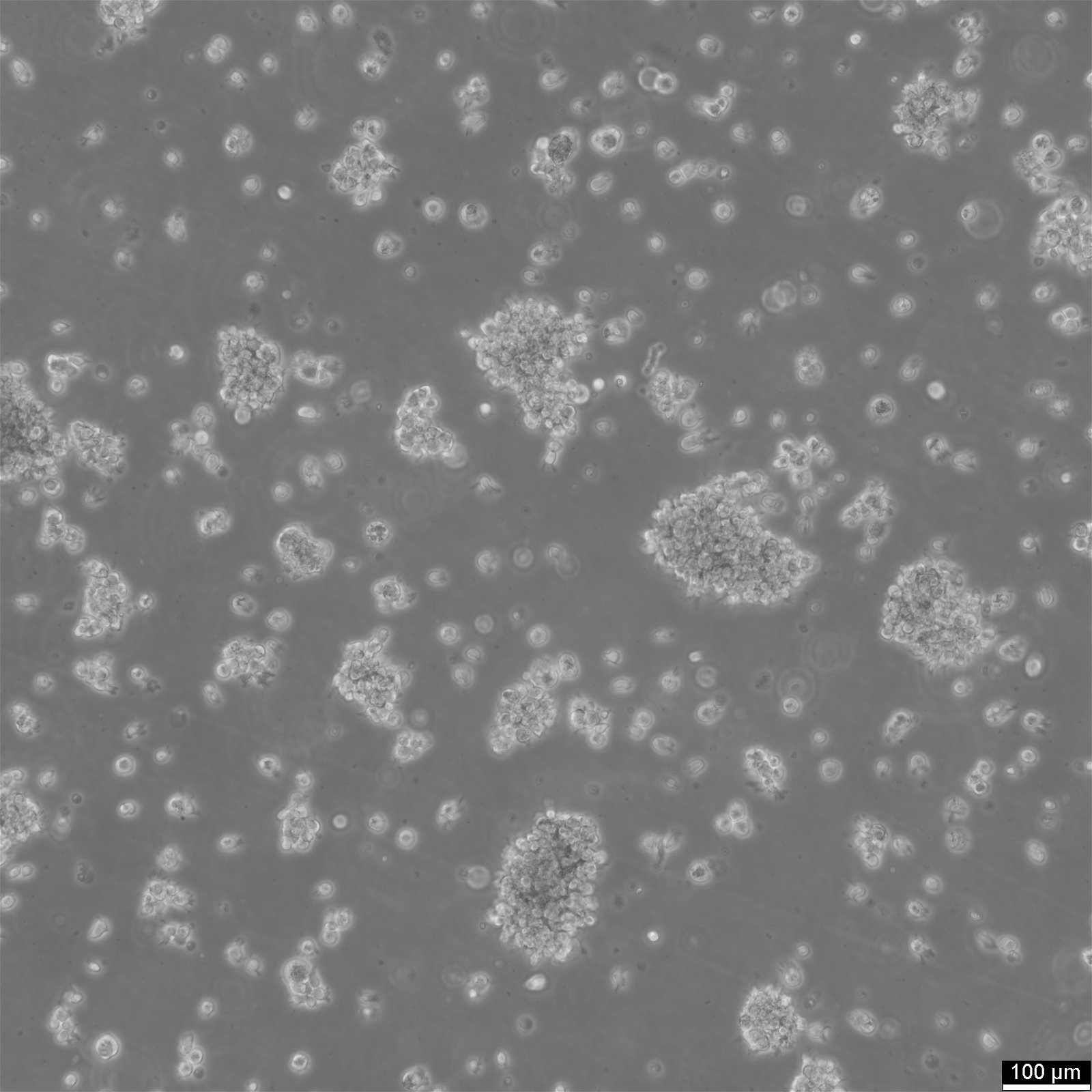
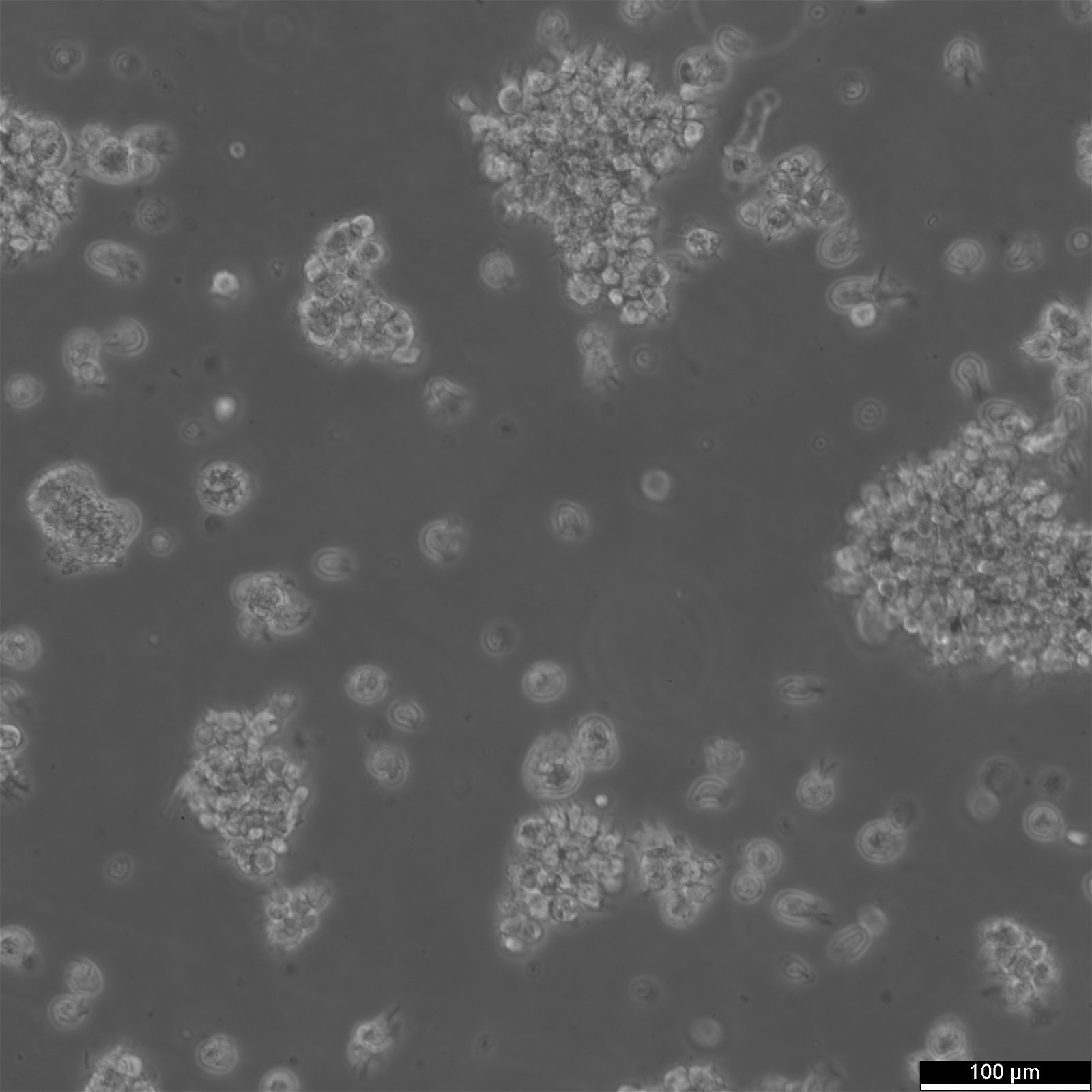
COX Cells













USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The COX cell line is a reference B-lymphoblastoid cell line (B-LCL) derived from a human donor and transformed with Epstein-Barr virus (EBV). It is frequently used in immunogenetics and histocompatibility research due to its inclusion in the International Histocompatibility Working Group (IHWG) panels. The COX cell line represents a specific major histocompatibility complex (MHC) haplotype, HLA-A1-B8-Cw7-DR3-DQ2, associated with susceptibility to autoimmune diseases such as type 1 diabetes, systemic lupus erythematosus, and myasthenia gravis. This haplotype is notable for its high degree of linkage disequilibrium, making the cell line an essential model for studying MHC-related genetic associations. The genomic sequence of the COX haplotype has been completely characterized as part of the MHC Haplotype Project. It spans approximately 4.8 Mb, encompassing the class I, II, and III regions of the MHC, as well as the extended class I region. Detailed sequencing revealed over 16,000 single nucleotide polymorphisms (SNPs) and numerous structural variations, providing insights into the genetic architecture of this region. The COX cell line's comprehensive MHC characterization makes it a key resource for understanding immune system function and the genetic basis of HLA-associated diseases. In research, the COX cell line is used for fine mapping of disease-associated loci within the MHC, as well as for functional studies on antigen processing and presentation. Its well-defined genetic profile allows for comparative studies with other MHC haplotypes, aiding in the identification of disease risk variants and potential therapeutic targets. Additionally, the cell line is involved in the evaluation of new sequencing and genotyping technologies, serving as a standard reference in immunogenetic studies. |
|---|---|
| Organism | Human |
| Tissue | Peripheral blood |
| Disease | Burkitt lymphoma |
| Synonyms | LCL (DR3) |
Characteristics
| Age | Age unspecified |
|---|---|
| Gender | Male |
| Ethnicity | Caucasian |
| Morphology | Round cells |
| Cell type | B lymphoblast |
| Growth properties | Suspension |
Regulatory Data
| Citation | COX (Cytion catalog number 302138) |
|---|---|
| Biosafety level | 2 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_E534 |
Biomolecular Data
| Viruses | Transformed by EBV |
|---|
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% heat-inactivated FBS |
| Subculturing | Gently homogenize the cell suspension in the flask by pipetting up and down, then take a representative sample to determine the cell density per ml. Dilute the suspension to achieve a cell concentration of 1 x 105 cells/ml with fresh culture medium, and aliquot the adjusted suspension into new flasks for further cultivation. |
| Seeding density | 5 x 105 cells/cm2 |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 105 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 302138-081223 | Certificate of Analysis | 23. May. 2025 | 302138 |
| 302138-230925 | Certificate of Analysis | 05. Dec. 2025 | 302138 |
