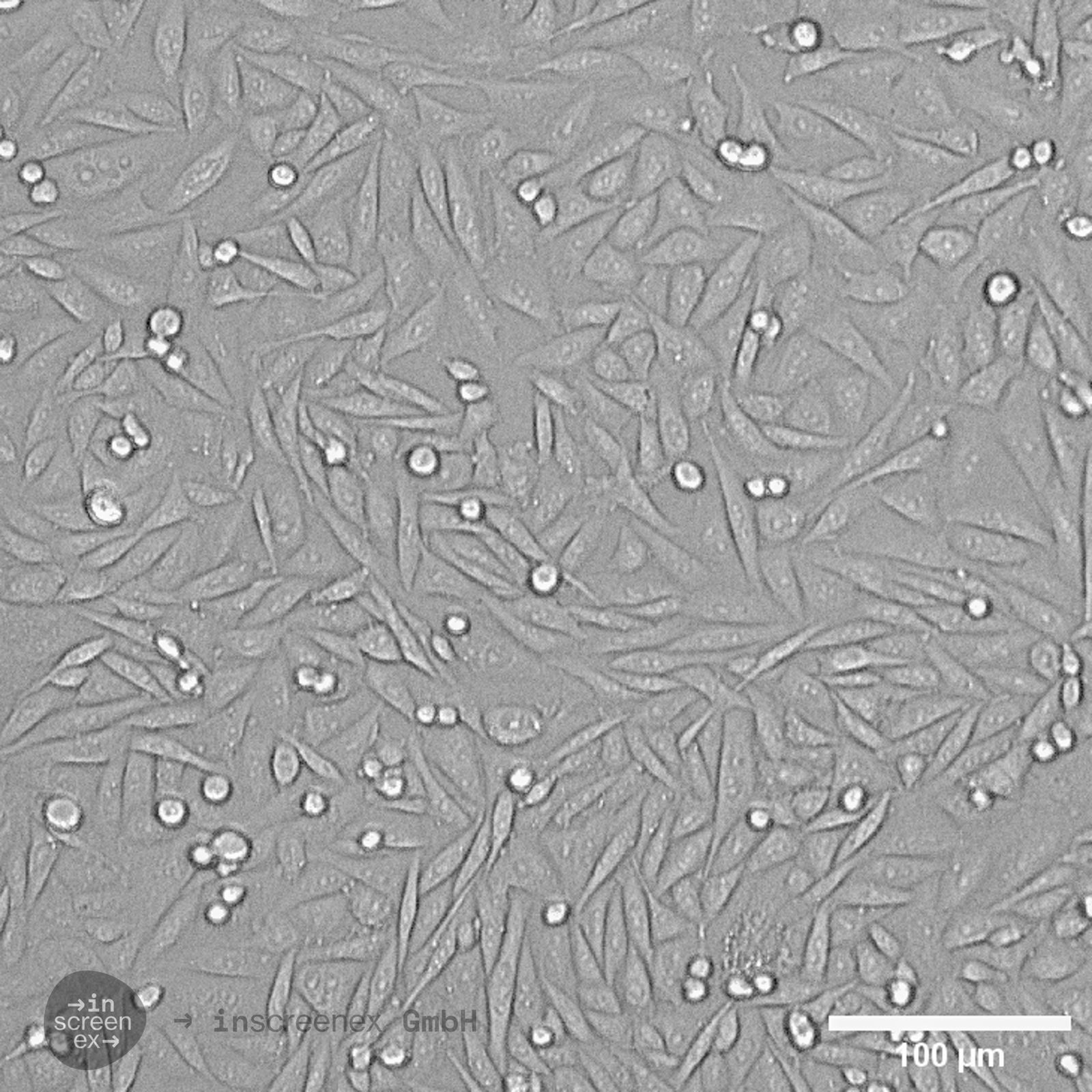
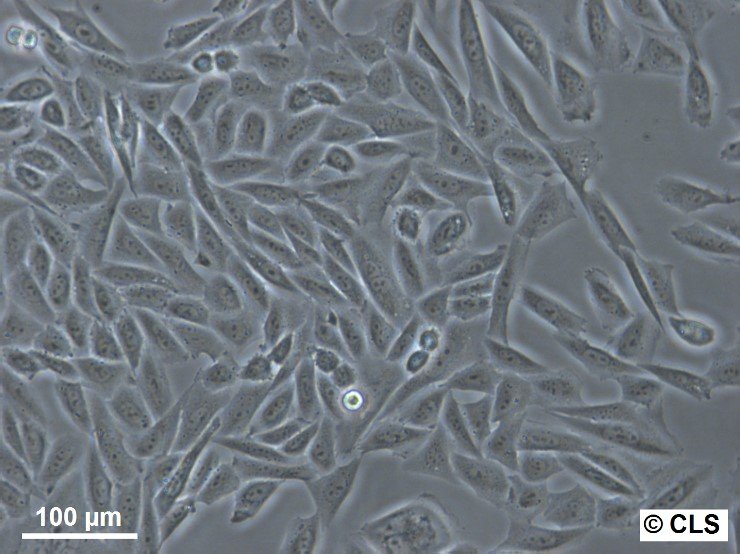
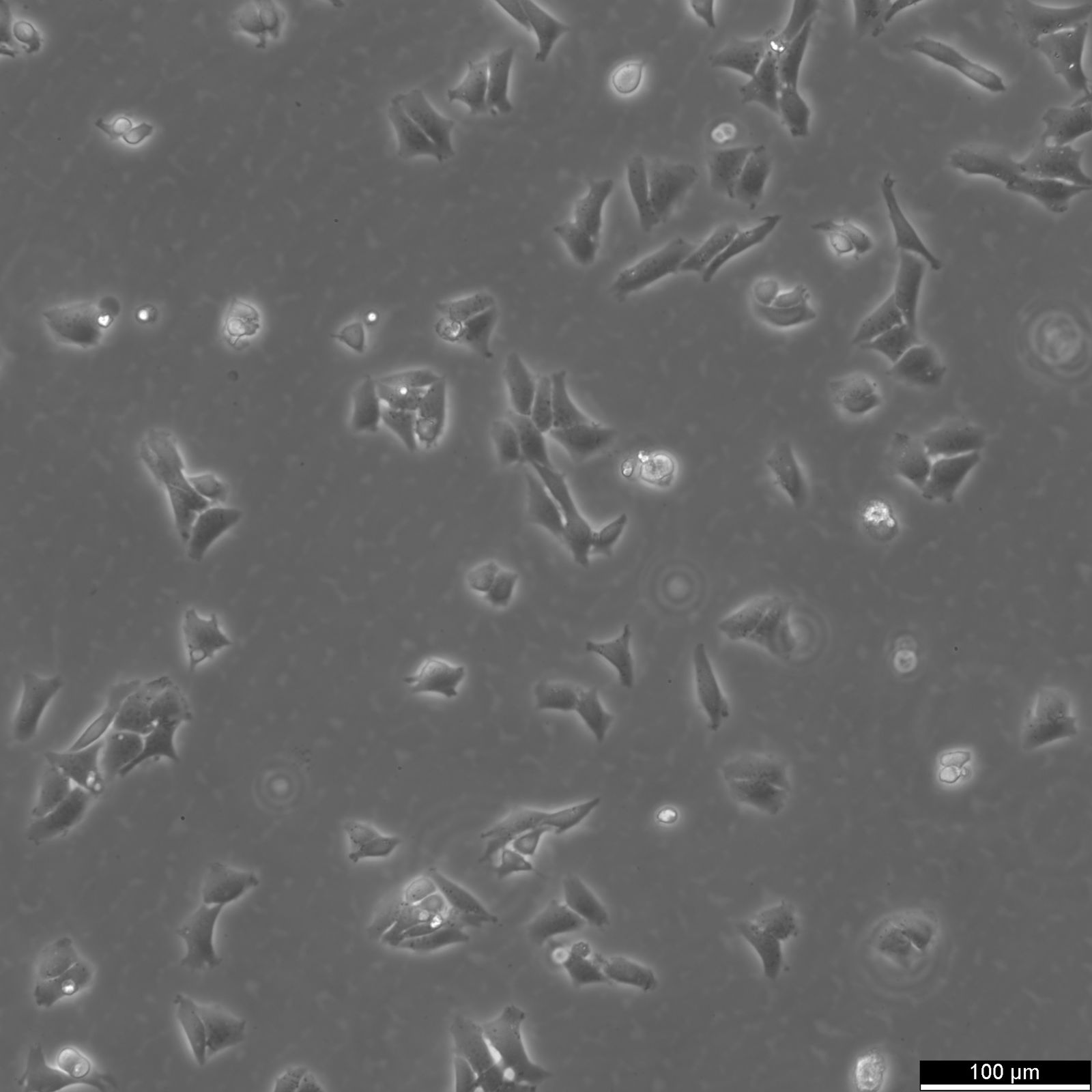
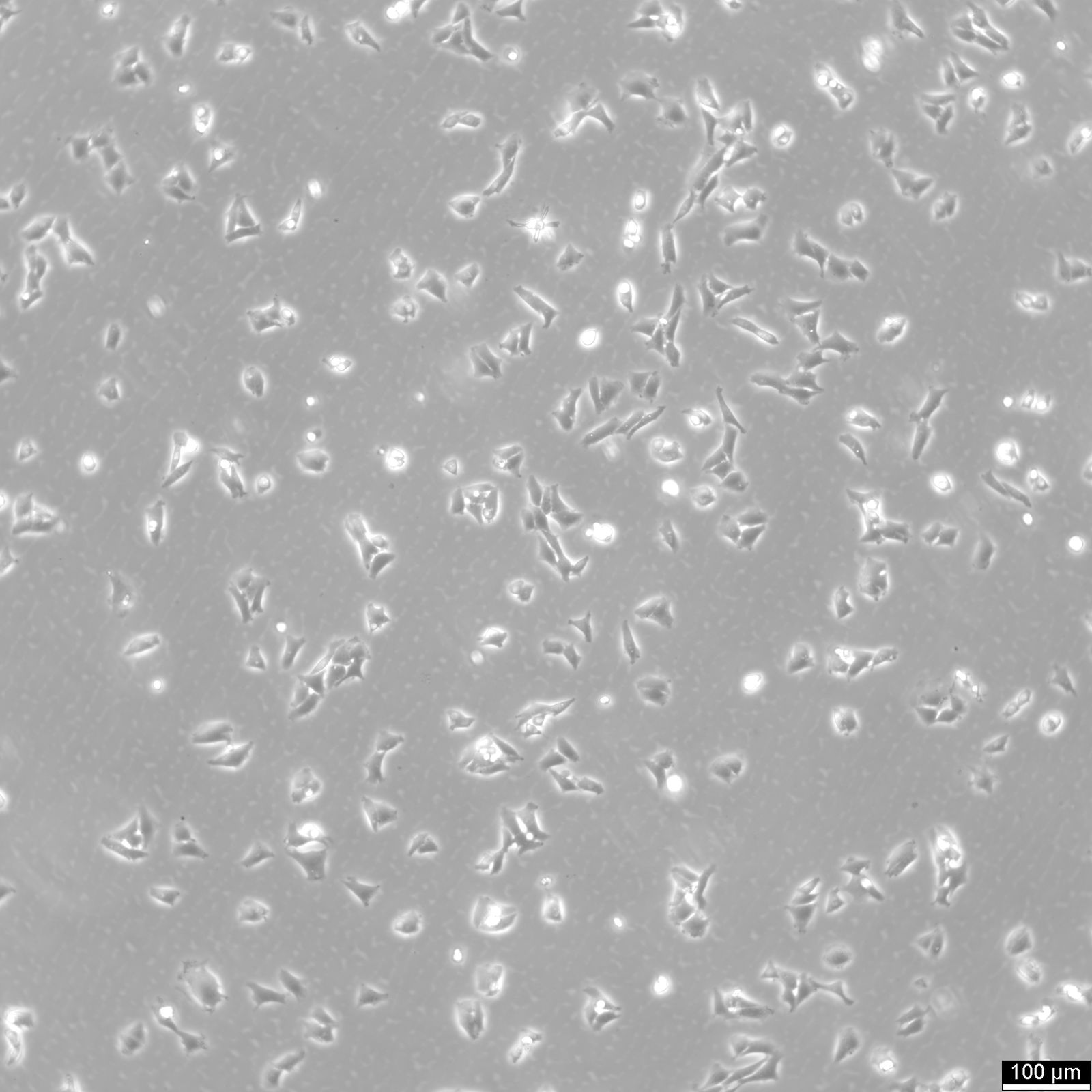
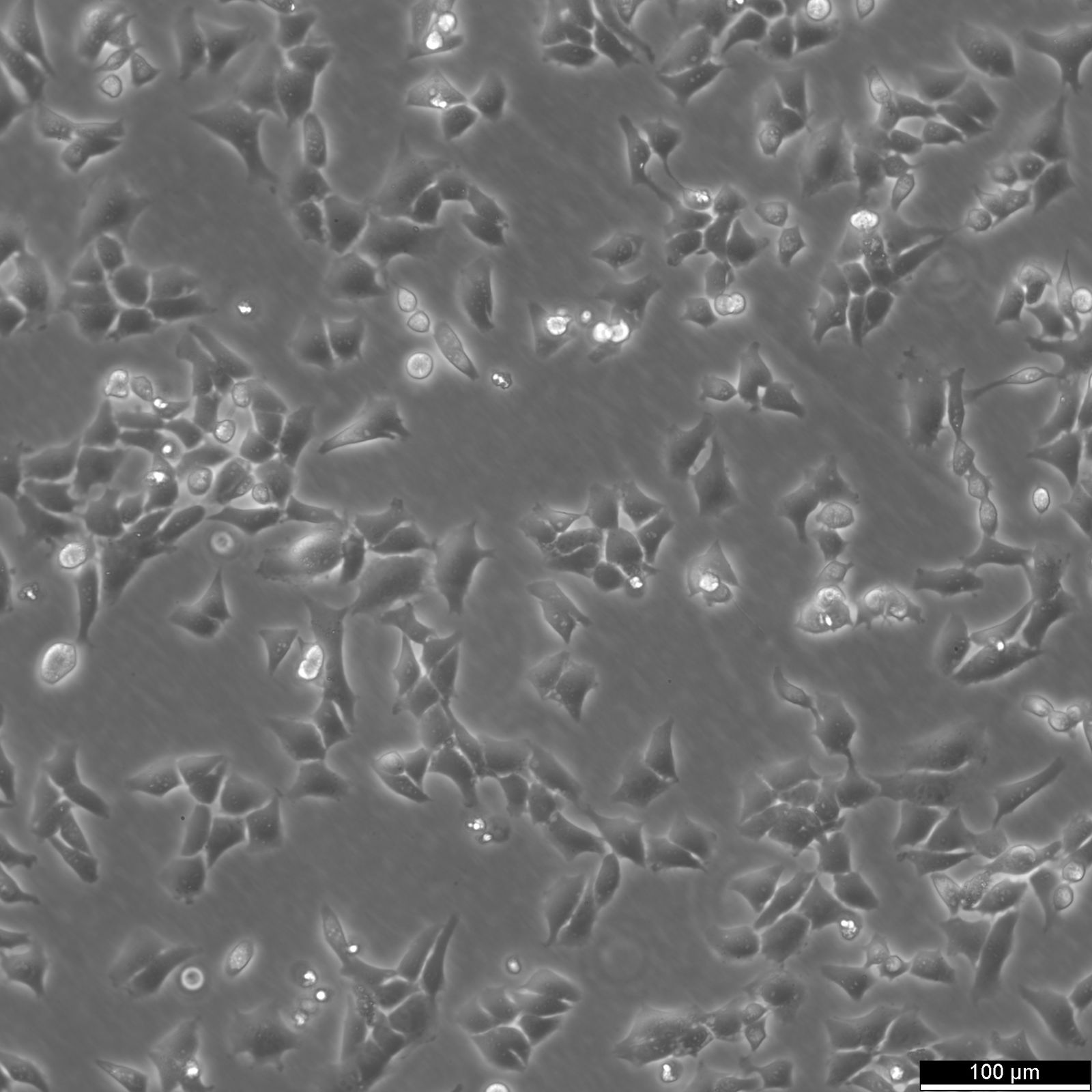
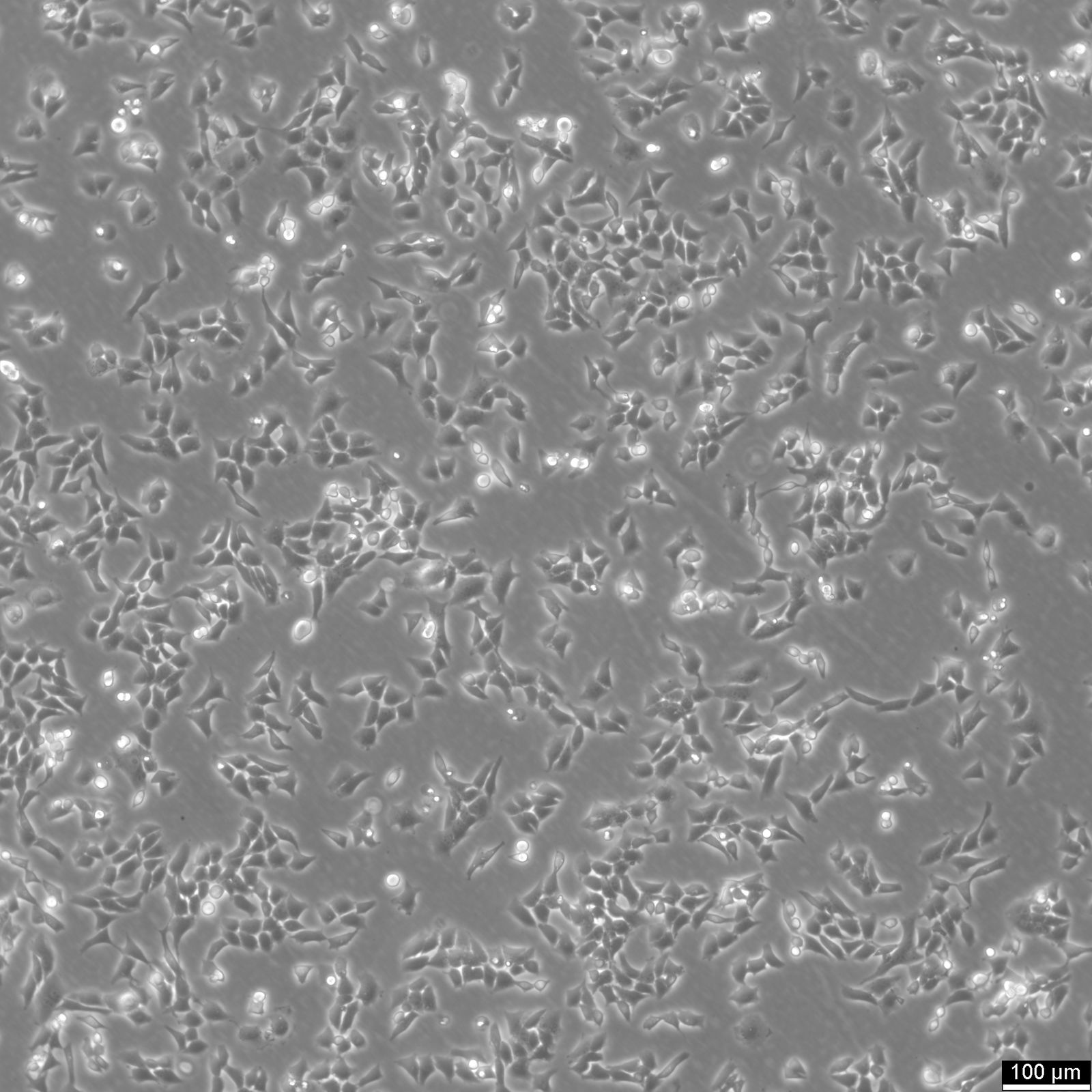
CHO Cells


















USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
Introduction to CHO cells
| Description | Chinese hamster ovary (CHO) cells are a cornerstone in the field of biotechnology and are heavily utilized in the process of CHO cell line development for the manufacture of biopharmaceuticals. These include monoclonal antibodies, recombinant antibody expression, and vaccines. The many advantages of CHO cells underscore their popularity in biomanufacturing, positioning them as a robust and versatile animal cell line with a proven track record in genetics, molecular biology, toxicity screening, nutrition, and gene expression studies. The contribution of CHO cells to the biopharmaceutical industry is immense, with their role in the development of recombinant antibodies and monoclonal antibody production being particularly significant. Nearly 50 biotherapeutics developed using these cells have been approved in the USA and EU, which speaks to the efficacy of CHO cells and their integral role in antibody development. Their hamster origin contributes to lower susceptibility to viruses, enhancing biosafety in biomanufacturing settings and reducing batch-to-batch variation. CHO cells are well-suited to produce proteins that undergo post-translational modifications, which is critical for therapeutic protein production. The versatility of the Chinese Hamster Ovary-derived cells is further highlighted by their fast proliferation rates and high protein expression rates of 1-5 grams per liter of culture. The ease of cultivating CHO cells and their ability to be genetically modified makes CHO cells an optimal choice for both transient and stable expression studies. The CHO-K1 cell line, a derivative of the original Chinese hamster ovary (CHO) cells is frequently utilized in expressing recombinant proteins, especially for the production of therapeutic proteins and recombinant antibodies. They excel in producing therapeutic proteins and antibodies due to efficient post-translational modification, notably glycosylation. Researchers modify CHO-K1 cells to enhance protein expression and tailor glycosylation for specific therapies, crucial in biomedicine. In conclusion, the Chinese hamster ovary cell line, known for its remarkable ability to mimic human post-translational modifications, is an invaluable scientific resource. Whether overcoming the difficulty of expressing challenging proteins or monoclonal antibody production, CHO cells have revolutionized the development and production of recombinant protein therapeutics. They remain pivotal in modern medicine, serving as a cornerstone for biopharmaceutical production and reflecting the advancements in biotechnology. |
|---|---|
| Organism | Chinese hamster |
| Tissue | Ovary |
| Applications | This cell line is an optimal choice for toxicology, industrial biotechnology and bioproduction. |
| Synonyms | Chinese Hamster Ovary, CHO-ori |
Specifications
| Age | Adult |
|---|---|
| Gender | Female |
| Morphology | Epithelial-like |
| Growth properties | Monolayer, adherent |
Documentation
| Citation | CHO (Cytion catalog number 603479) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 10029 |
| CellosaurusAccession | CVCL_0213 |
Genetic profile of the CHO cell line
Handling
| Culture Medium | Ham's F12, w: 1.0 mM stable Glutamine, w: 1.0 mM Sodium pyruvate, w: 1.1 g/L NaHCO3 (Cytion article number 820600a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 3 x 104 cells/cm2 will yield in a confluent layer in about 4 days |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality assurance of CHO cells
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 603479-060325 | Certificate of Analysis | 22. Oct. 2025 | 603479 |
-
Related products
Related products
CHO-CXCR4 Cells (medium-high)Variants: Medium-highOrganism Chinese hamster Tissue Ovary USD$2,200.00*CHO-CXCR7 Cells (medium-high)Variants: Medium-highOrganism Chinese hamster Tissue Ovary USD$2,200.00*