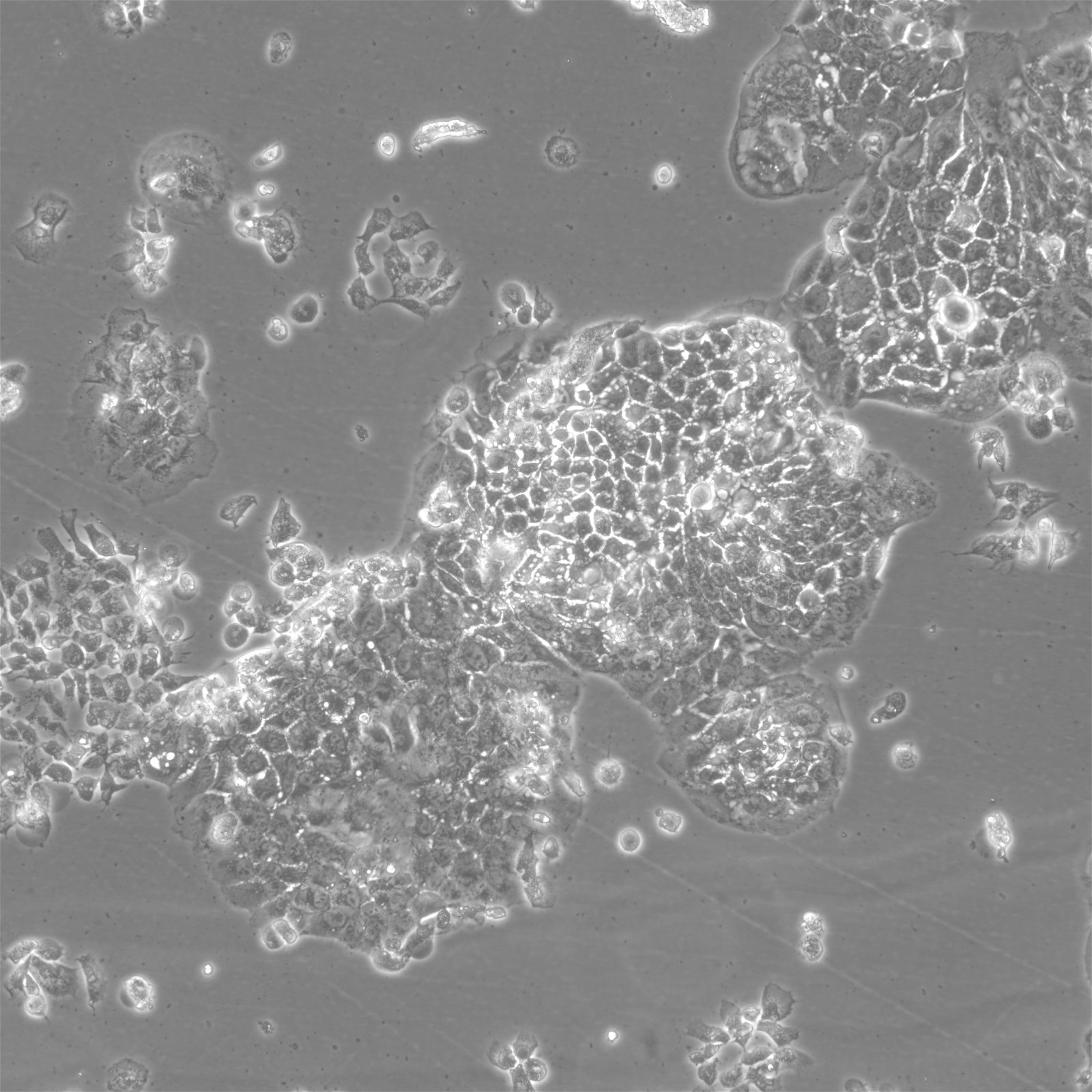
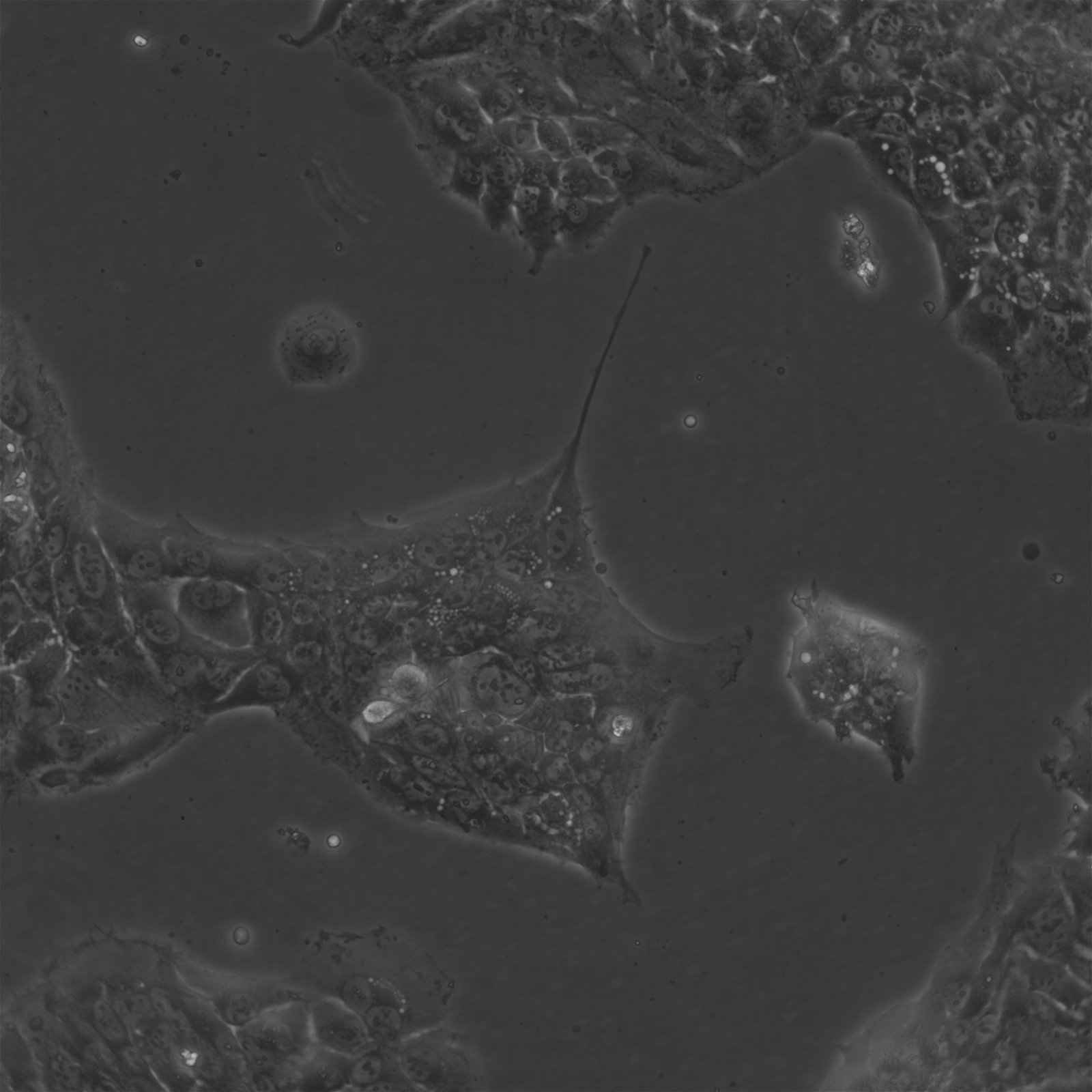
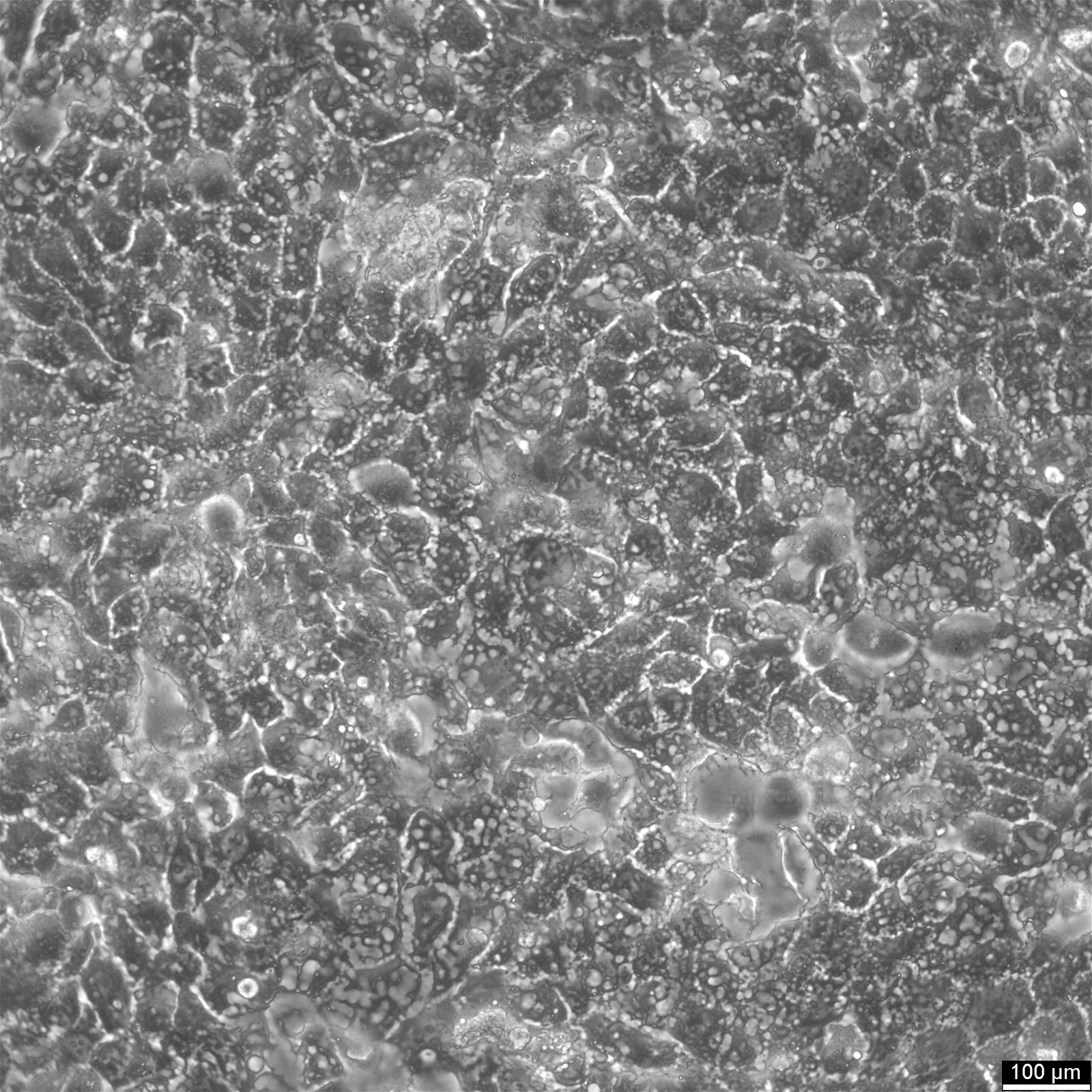
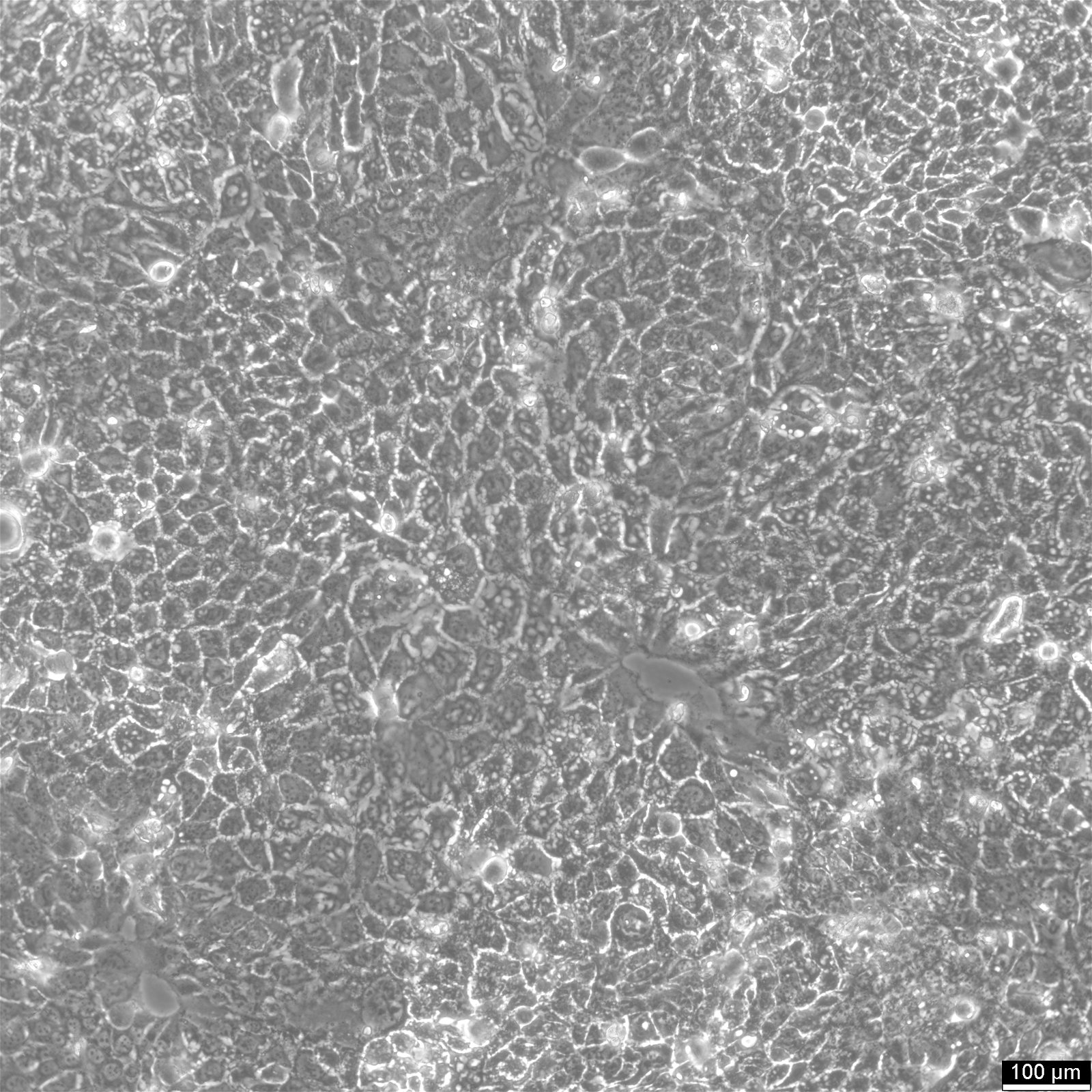
BEWO Cells












USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | BeWo cells, a cell line derived from malignant gestational choriocarcinoma of the fetal male placenta, have become a widely used in vitro model for studying the placenta. The cell-cell fusion during the human trophoblast syncytialization phase during placental development is one of the most significant yet least understood events. Due to the difficulty of studying this process in a placenta in vivo, BeWo cells are utilized as a cell culture model to simulate in vivo syncytialization of the placental villous trophoblast. These cells exhibit an epithelial-like phenotype and are adherent. The b30 subclone of BeWo cells is particularly useful for studying nutrient uptake and transport due to its dense growth on permeable membranes. CK 7 and E-cadherin are molecular markers that are expressed by BeWo cells. VE-cadherin is found in BeWo cells and is enhanced upon treatment with forskolin. The cells also express keratin and are positive for G6PD, B isoenzyme. The karyotype of BeWo cells is modal number = 86, with a range of 71 to 178, and the stemline number is hypotetraploid. The karyotype is relatively stable within the stemline number. BeWo cells secrete various hormones, including human chorionic gonadotropin (hCG), human chorionic somatomammotropin (placental lactogen), and steroid hormones as estrone, estriol, and estradiol. However, the levels of β-hCG and estradiol secreted by BeWo cells are lower than those secreted by other choriocarcinoma-derived cell lines such as JEG-3. Upon Forskolin treatment, the secretion of β-hCG in BeWo cells increases to a level similar to that observed in the other choriocarcinoma-derived cell lines. Furthermore, Forskolin treatment also increases the progesterone levels secreted by BeWo cells. In summary, BeWo cells are a widely used in vitro model for studying placental development and the human trophoblast syncytialization process. They exhibit an epithelial-like phenotype, express various molecular markers, and secrete multiple hormones, including hCG, placental lactogen, and steroid hormones. Overall, BeWo cells are a valuable tool for investigating the complex processes involved in placental development. |
|---|---|
| Organism | Human |
| Tissue | Placenta |
| Disease | Choriocarcinoma |
| Metastatic site | Brain |
| Synonyms | BeWo, Be Wo, Be-Wo |
Characteristics
| Age | Fetus |
|---|---|
| Gender | Male |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | BEWO (Cytion catalog number 300123) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0044 |
Biomolecular Data
| Isoenzymes | G6PD, B |
|---|---|
| Virus susceptibility | Poliovirus 3, vesicular stomatitis (Indiana) |
| Reverse transcriptase | Negative |
| Products | Progesterone, human chorionic somatomammotropin (placental lactogen), estrogen, estrone, estriol, estradiol, keratin |
Handling
| Culture Medium | Ham's F12K Medium, w: 2.0 mM L-Glutamine, w: 2.0 mM Sodium pyruvate, w: 2.5 g/L NaHCO3 (Cytion article number 820608a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | A seeding density of 1 x 104 cells/cm2 is recommended. |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300123-1022 | Certificate of Analysis | 23. May. 2025 | 300123 |
| 300123-010425 | Certificate of Analysis | 23. May. 2025 | 300123 |
