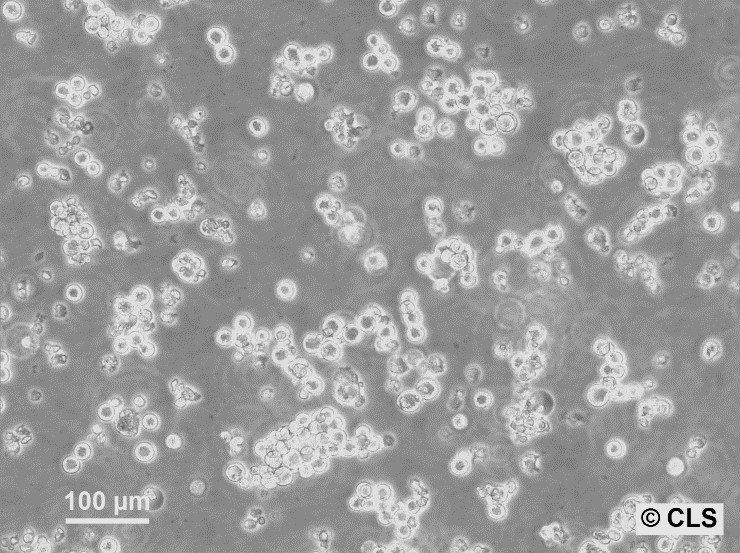
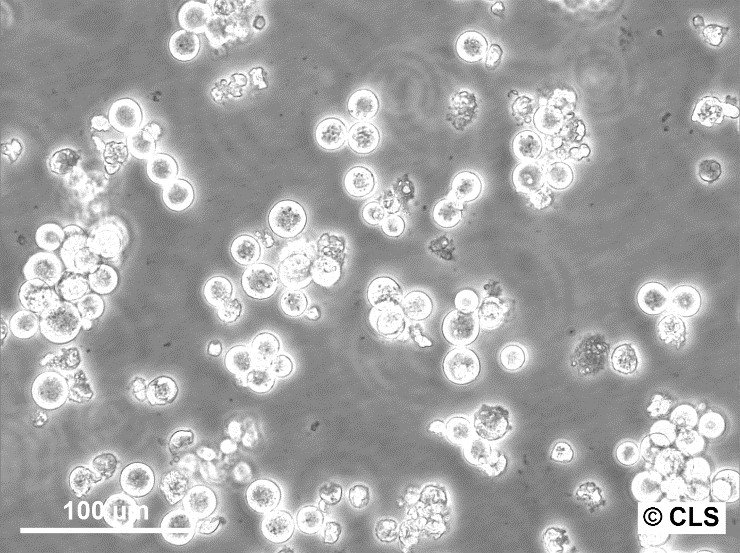
AH-130 FN Cells






USD$550.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | AH-130 FN is a variant of the AH-130 rat ascites tumor cell line, used extensively in studies related to coagulation, fibrinolysis, and metastasis. These cells were derived from rats and are typically maintained by serial intraperitoneal implantation in male Donryu rats. The AH-130 line itself is known for its high thromboplastic and fibrinolytic activities, which are linked to its role in promoting blood-borne metastasis, especially in the lungs. In contrast, the AH-130 FN variant has lower thromboplastic and fibrinolytic activity. This difference in enzymatic activity between AH-130 and AH-130 FN is crucial as it influences the formation of thrombi and the number of metastatic foci in the lungs following intravenous inoculation. Research has shown that after intravenous injection, AH-130 cells cause a significant reduction in platelet count and fibrinogen levels, indicative of increased thrombus formation. This effect is notably more pronounced than in AH-130 FN. Histological studies demonstrate that AH-130 forms more abundant metastatic foci in the lungs compared to AH-130 FN, both at 72 hours and 7 days post-inoculation. AH-130 is associated with the formation of thrombi composed of platelets and fibrin around embolized tumor cells, whereas AH-130 FN shows sparse thrombus formation. These findings suggest that AH-130’s higher thromboplastic activity plays a significant role in promoting metastasis through platelet aggregation and fibrin deposition around the tumor cells, a process less prominent in AH-130 FN. |
|---|---|
| Organism | Rat |
| Tissue | Liver |
| Disease | Hepatocellular carcinoma |
| Synonyms | AH130FN-TC, AH130FN, AH-130F(N), AH-130FN, AH 130 FN |
Characteristics
| Morphology | Round cells in suspension, Epithelial-like when adherent |
|---|---|
| Growth properties | Suspension, few adherent |
Regulatory Data
| Citation | AH-130 FN (Cytion catalog number 500451) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 10116 |
| CellosaurusAccession | CVCL_5683 |
Biomolecular Data
| Tumorigenic | Yes, in Wistar rats. |
|---|---|
| Viruses | RAP-test negative. . |
Handling
| Culture Medium | DMEM:Ham's F12 (1:1), w: 3.1 g/L Glucose, w: 2.5 mM L-Glutamine, w: 15 mM HEPES, w: 0.5 mM Sodium pyruvate, w: 1.2 g/L NaHCO3 (Cytion article number 820400a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Subculturing | Gently homogenize the cell suspension in the flask by pipetting up and down, then take a representative sample to determine the cell density per ml. Dilute the suspension to achieve a cell concentration of 1 x 105 cells/ml with fresh culture medium, and aliquot the adjusted suspension into new flasks for further cultivation. |
| Seeding density | 1 x 106 cells/cm2 |
| Fluid renewal | Every 3 to 5 days |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
