AAV-293 Cells























USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
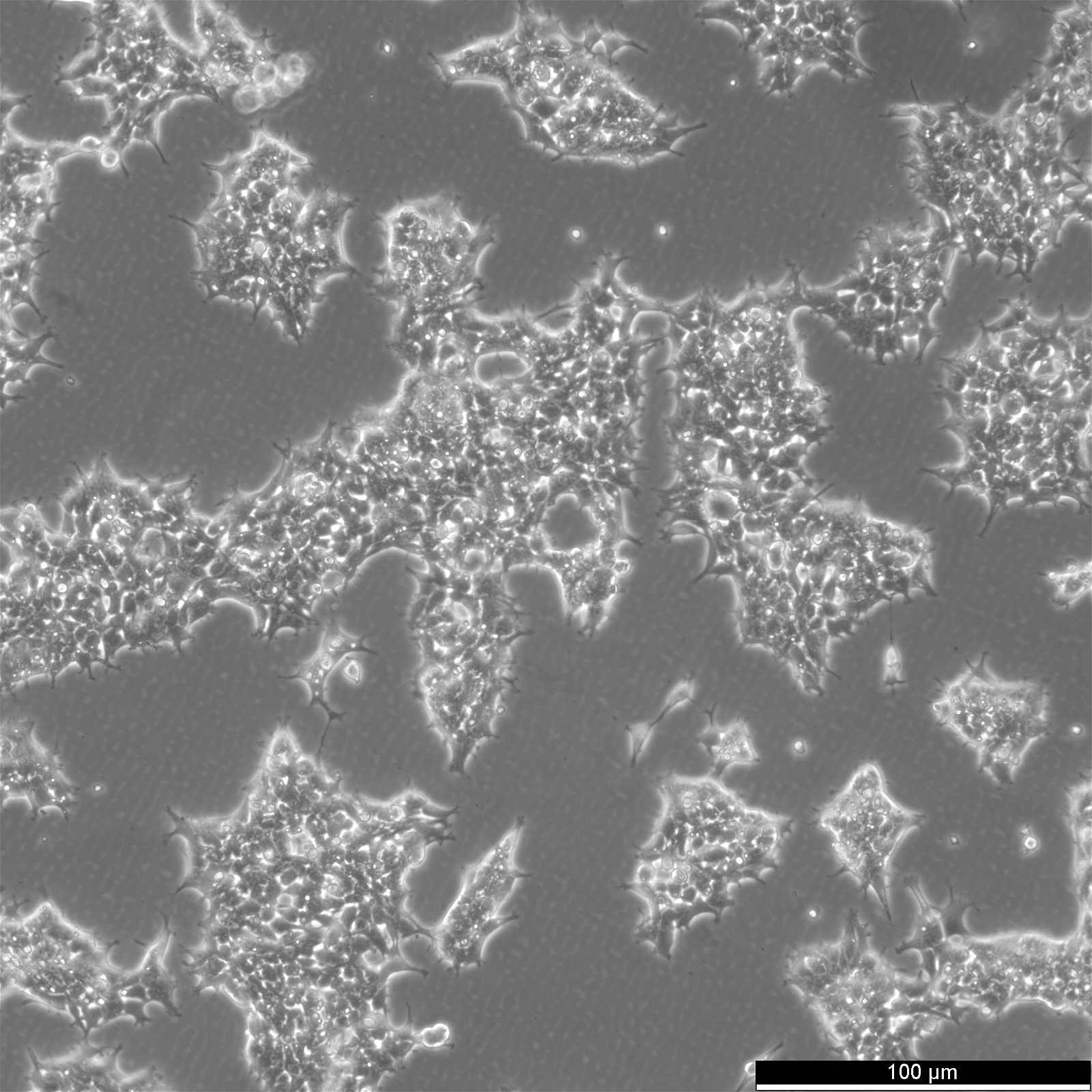
| Description | The AAV-293 cell line is a permanent line established from primary embryonic human kidney transformed with human adenovirus type 5 DNA. The genes encoded by the E1 region of adenovirus (E1a and E1b) are expressed in these cells and participate in transactivation of viral promoters, allowing these cells to produce high levels of protein. AAV-293 is derived from the parental 293 cell line, through cloning and multiple rounds of testing, AAV-293 is specifically selected for a high level of AAV production in a helper-free system. It offers several advantages over the regular 293 cells: Larger cell surface area resulting higher transfection and better yield of AAV. The advantages are a flattened morphology, firm attachment to culture plate, and the cells are ideal for large-scale culture and AAV production. Adeno-associated virus (AAV) belongs to the family of Parvoviridae, a group of viruses among the smallest of single-stranded and non-enveloped DNA viruses. There are nine different AAV serotypes reported to date. AAV can infect both dividing and non-dividing cells and can be maintained in the human host cell, creating the potential for long-term gene transfer. Recombinant AAV-2 is the most common serotype used in gene delivery, and can be produced at high titers with a helper virus or AAV-293 cells. |
|---|---|
| Organism | Human |
| Tissue | Embryonic kidney |
| Synonyms | AAV293 |
Characteristics
| Age | Fetus |
|---|---|
| Gender | Female |
| Morphology | Epithelial |
| Growth properties | Adherent |
Regulatory Data
| Citation | AAV-293 (Cytion catalog number 305127) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_6871 |
| GMO Status | GMO-S1: This HEK293-derived AAV-293 line contains clonal modifications supporting AAV vector production. This classification applies only within Germany and may differ elsewhere. |
Biomolecular Data
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 3.7 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS, 0.1 mM NEAA |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 5 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305127-120825 | Certificate of Analysis | 22. Oct. 2025 | 305127 |
