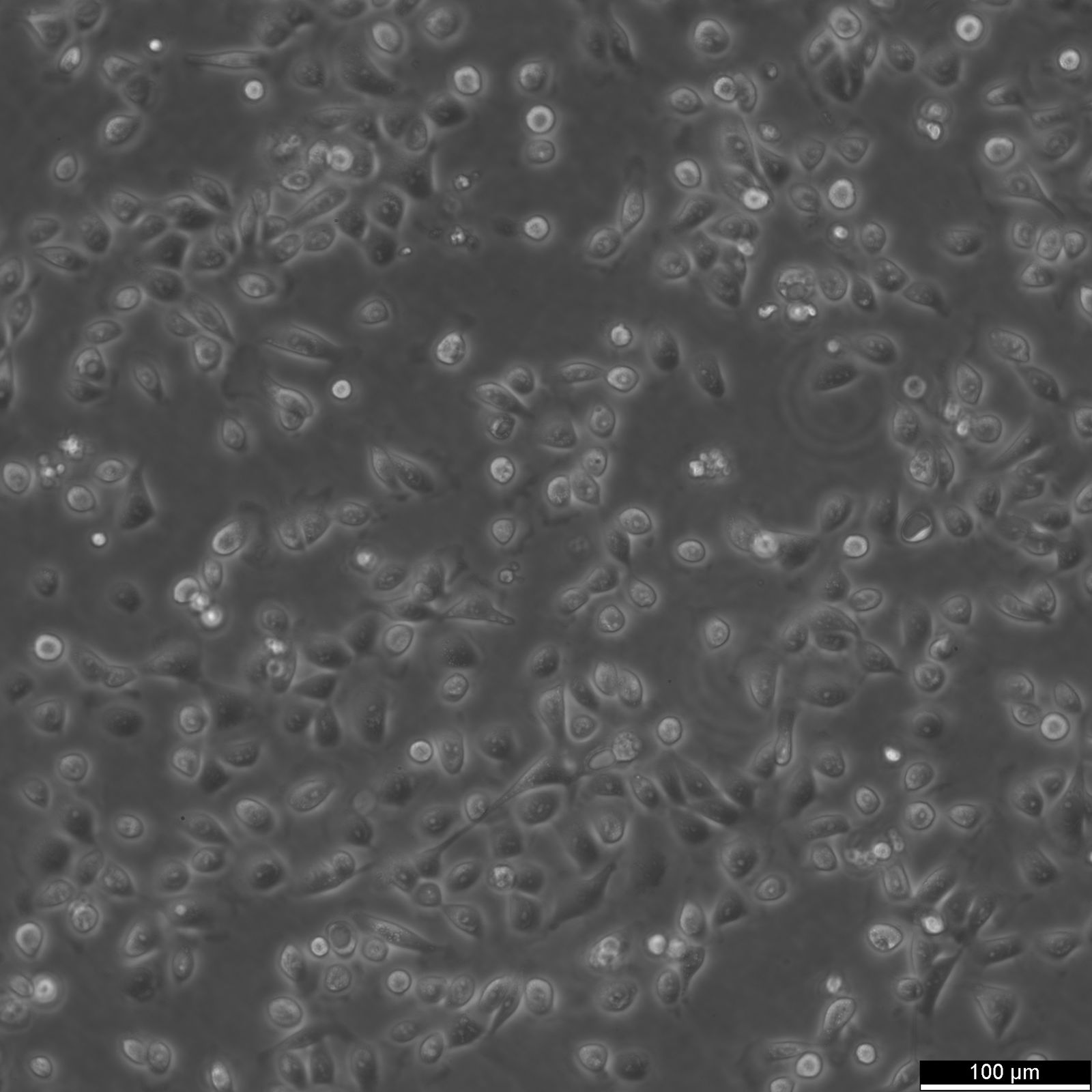
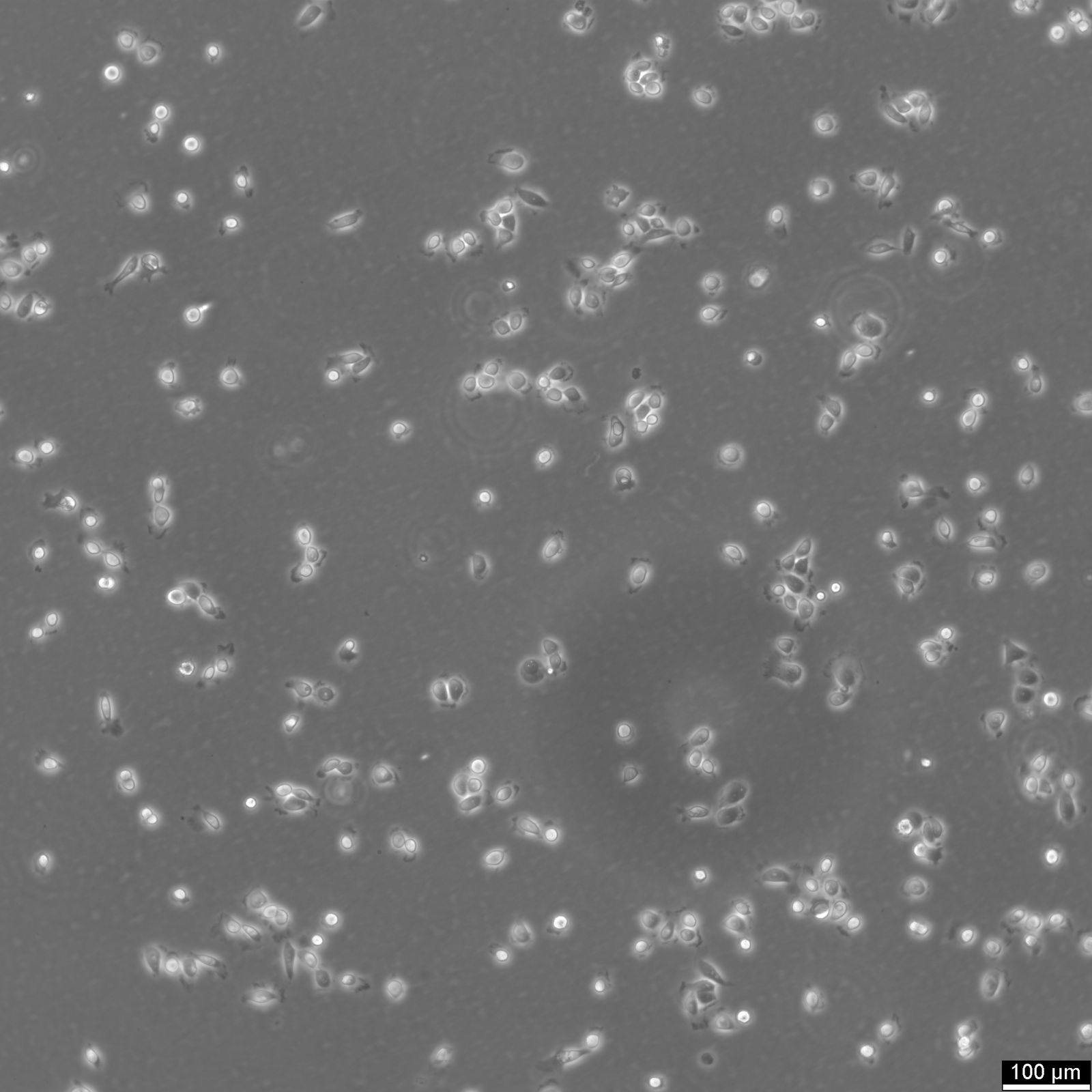
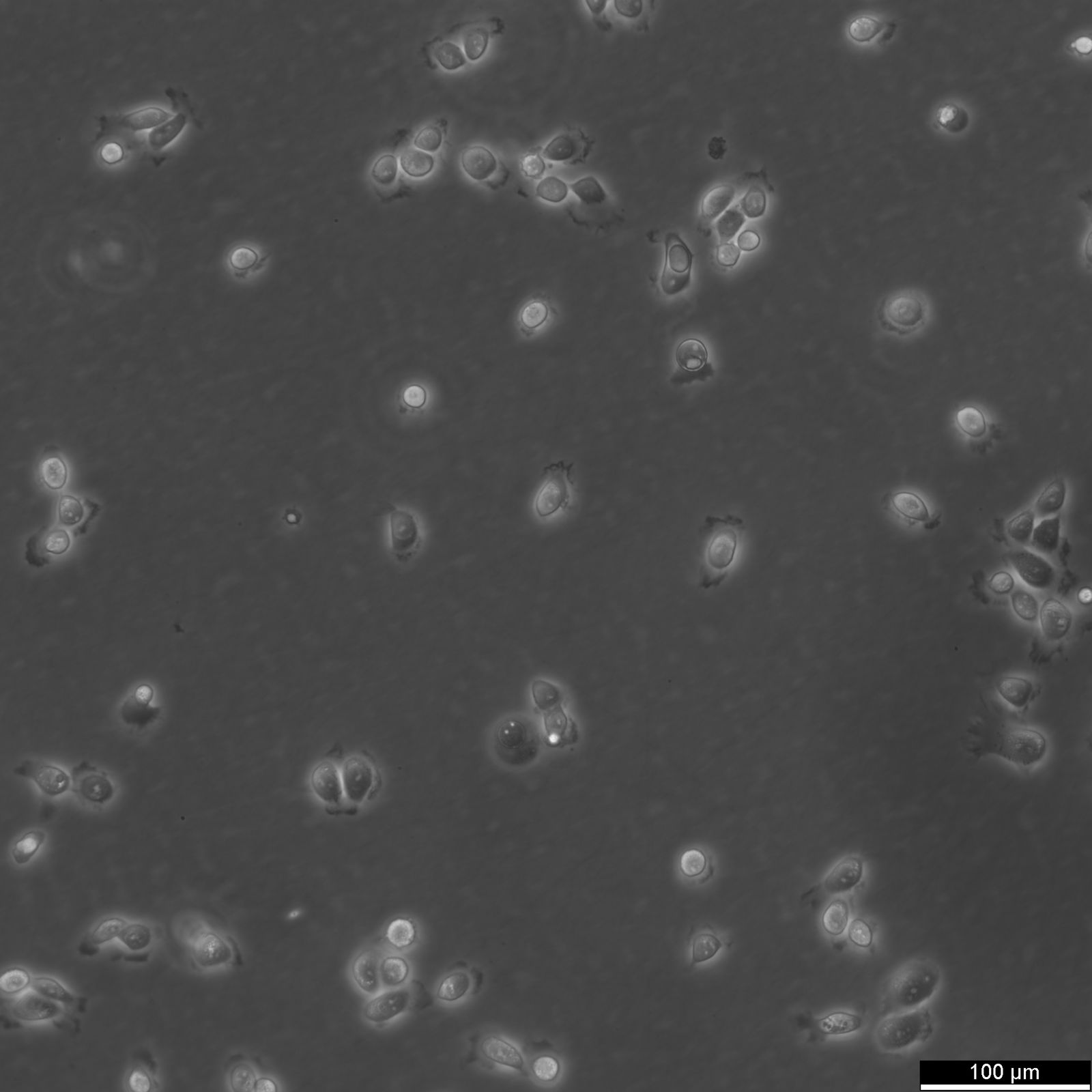
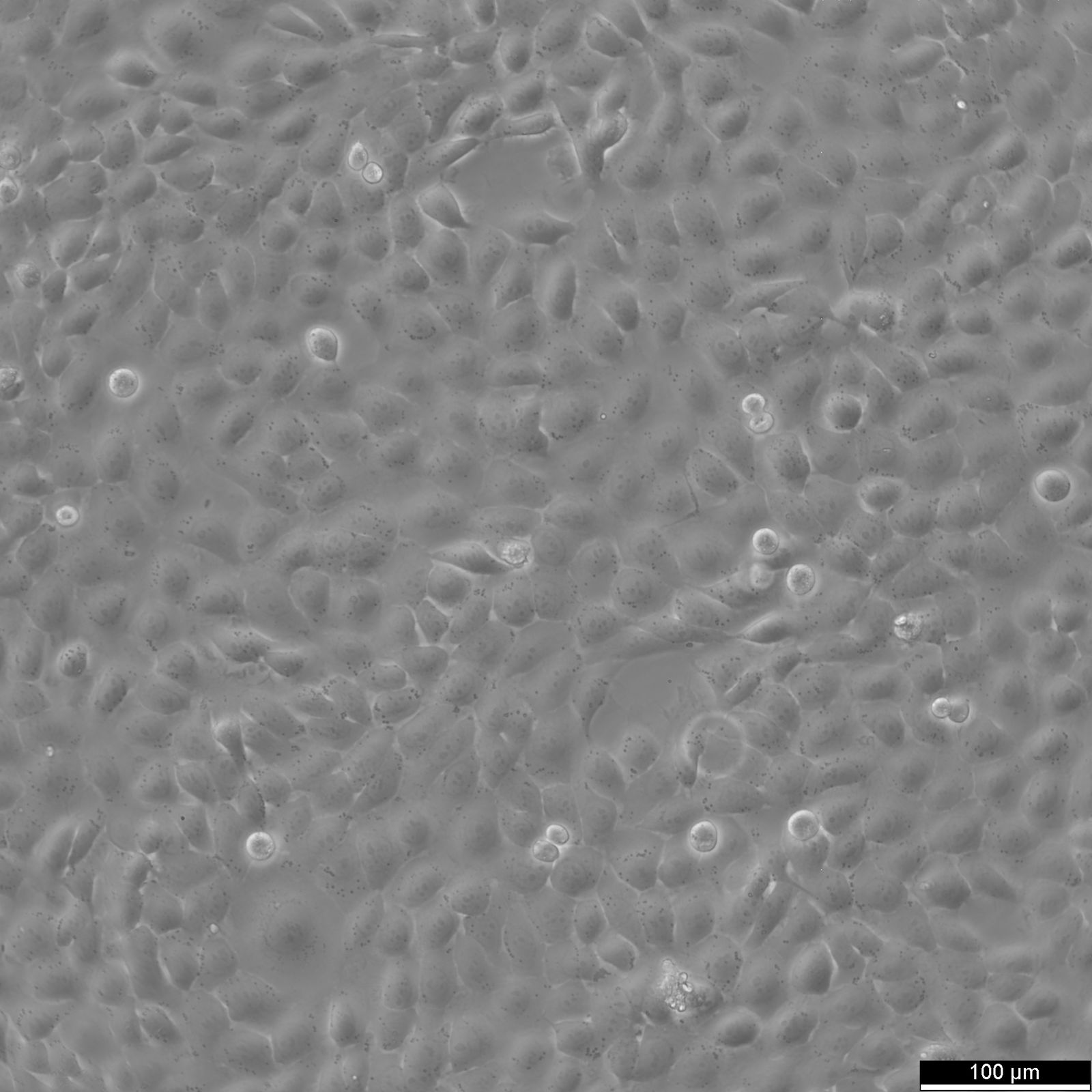
5637 Cells




















USD$395.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | 5637 is a bladder carcinoma cell line isolated from the urinary bladder of a 68-year-old man with grade II carcinoma. 5637 cells produce and secrete several growth factors, such as SCF, IL-1, IL-6, G-CSF, and GM-CSF. These cytokines are functionally active and can be a valuable source for the culture of growth factor-responsive or dependent hematopoietic primary cells and cell lines. The karyotype modal chromosome number of 5637 cells is 67, ranging from 59 to 71. The stemline modal chromosome number is 67 at 36% and polyploidy at 0.6%. Fourteen marker chromosomes are common to these cells, including 3q+, 11q+, i(13q), t(9q21q), i(17q), i(21q). Additional markers, like der(5)t(5;7)(q31;p11) and 1p, were only found specific to a minor subpopulation, as well as microchromosomes and double minutes (DM). Some cells include one or occasionally two Y chromosomes. 5637 cells are tumorigenic and have been shown to induce tumours in nude mice inoculated subcutaneously. The doubling time of 5637 cells is approximately 24 hours. The isoenzyme profile of 5637 cells consists of isoform 1 of AK-1, ES-D, Me-2 and PGM1, isoform 1 and 2 of GLO-I, isoform B of G6PD, as well as isoform 2 of PGM3. In terms of oncogenes, 5637 cells are positive for FGFR3, PIK3CA, HRAS, KRAS, NRAS, TERT, and CDKN2A but negative for TP53 and belong to the molecular bladder cancer subtype l5637 is a bladder carcinoma cell line isolated from the urinary bladder of a 68-year-old man with grade II carcinoma. 5637 cells produce and secrete several growth factors, such as SCF, IL-1, IL-6, G-CSF, and GM-CSF. These cytokines are functionally active and can be a valuable source for the culture of growth factor-responsive or dependent hematopoietic primary cells and cell lines. The karyotype modal chromosome number of 5637 cells is 67, ranging from 59 to 71. The stemline modal chromosome number is 67 at 36% and polyploidy at 0.6%. Fourteen marker chromosomes are common to these cells, including 3q+, 11q+, i(13q), t(9q21q), i(17q), i(21q). Additional markers, like der(5)t(5;7)(q31;p11) and 1p, were only found specific to a minor subpopulation, as well as microchromosomes and double minutes (DM). Some cells include one or occasionally two Y chromosomes. 5637 cells are tumorigenic and have been shown to induce tumours in nude mice inoculated subcutaneously. The doubling time of 5637 cells is approximately 24 hours. The isoenzyme profile of 5637 cells consists of isoform 1 of AK-1, ES-D, Me-2 and PGM1, isoform 1 and 2 of GLO-I, isoform B of G6PD, as well as isoform 2 of PGM3. In terms of oncogenes, 5637 cells are positive for FGFR3, PIK3CA, HRAS, KRAS, NRAS, TERT, and CDKN2A but negative for TP53 and belong to the molecular bladder cancer subtype luminal. In conclusion, 5637 cells are a valuable tool for cancer research, especially with respect to the study of growth factors, cell division, oncogenes, and bladder cancer. |
|---|---|
| Organism | Human |
| Tissue | Bladder |
| Disease | Carcinoma |
| Applications | This cell line is an optimal choice for transfection. |
Characteristics
| Age | 68 years |
|---|---|
| Gender | Male |
| Ethnicity | Caucasian |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | 5637 (Cytion catalog number 300105) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_0126 |
Biomolecular Data
| Isoenzymes | Me-2, 1, PGM3, 2, PGM1, 1, ES-D, 1, AK-1, 1, GLO-1, 1-2, G6PD, B |
|---|---|
| Tumorigenic | Yes, In nude mice. |
| Products | IL-1, IL-6, G-CFS, GM-CSF, SCF |
| Ploidy status | The modal chromosome number of the stemline cells is 67, comprising 36% of the total. Polyploidy occurs in 0.6% of these cells. Each cell typically had one or occasionally two Y chromosomes. |
| Karyotype | Phenotype Frequency Product: 0.0056. |
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Doubling time | 24 hours |
| Subculturing | First, remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Seeding density | 1 x 104 cells/cm2 will result in a confluent monolayer within 3 days. |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | After thawing, plate the cells at 5 x 104 cells/cm2 and allow the cells to recover from the freezing process and to adhere for at least 24 hours. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 300105-140425 | Certificate of Analysis | 23. May. 2025 | 300105 |
| 300105-715SF | Certificate of Analysis | 23. May. 2025 | 300105 |
| 300105-613 | Certificate of Analysis | 05. Dec. 2025 | 300105 |
