2V6.11 Cells












USD$800.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
Insights on 2v6.11 cells
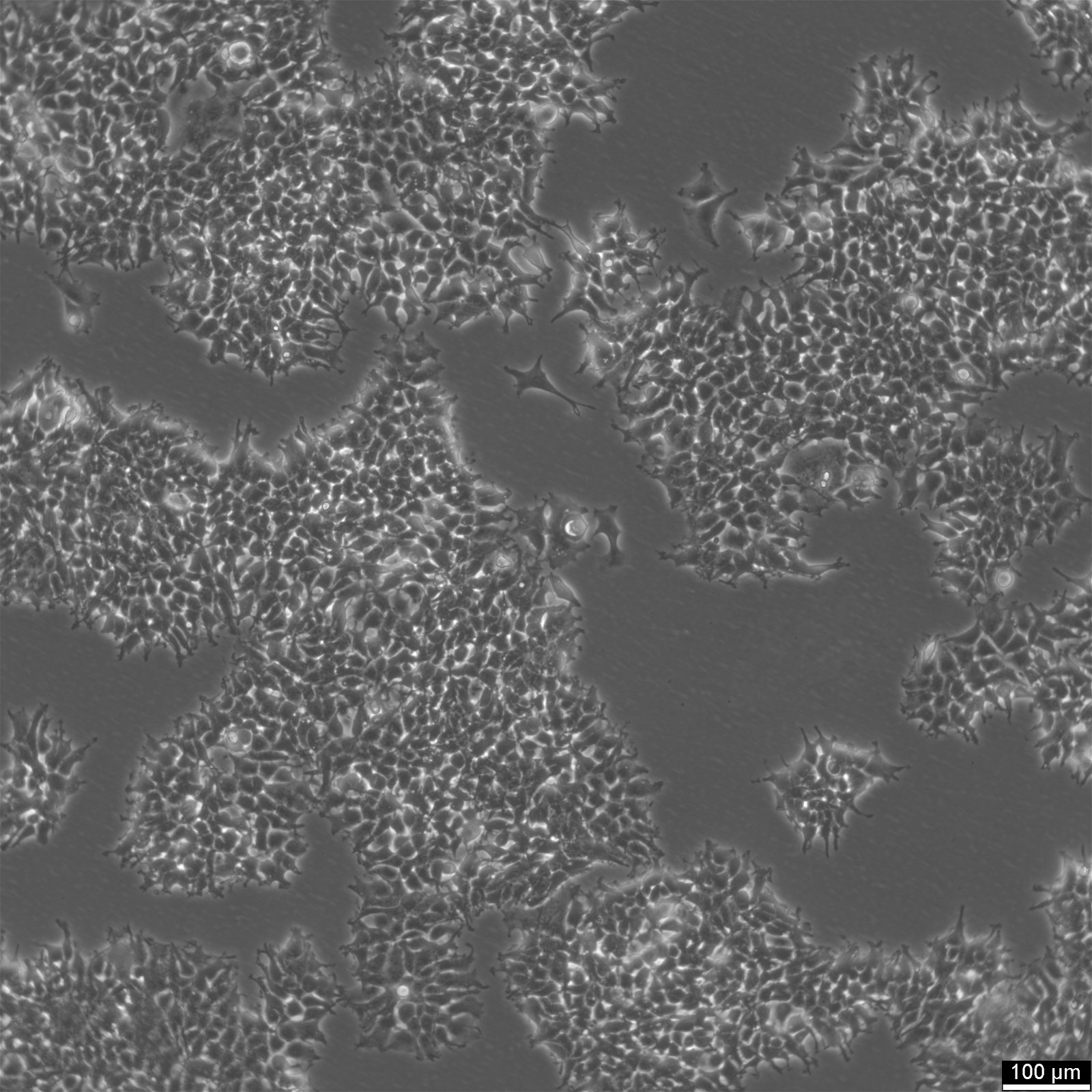
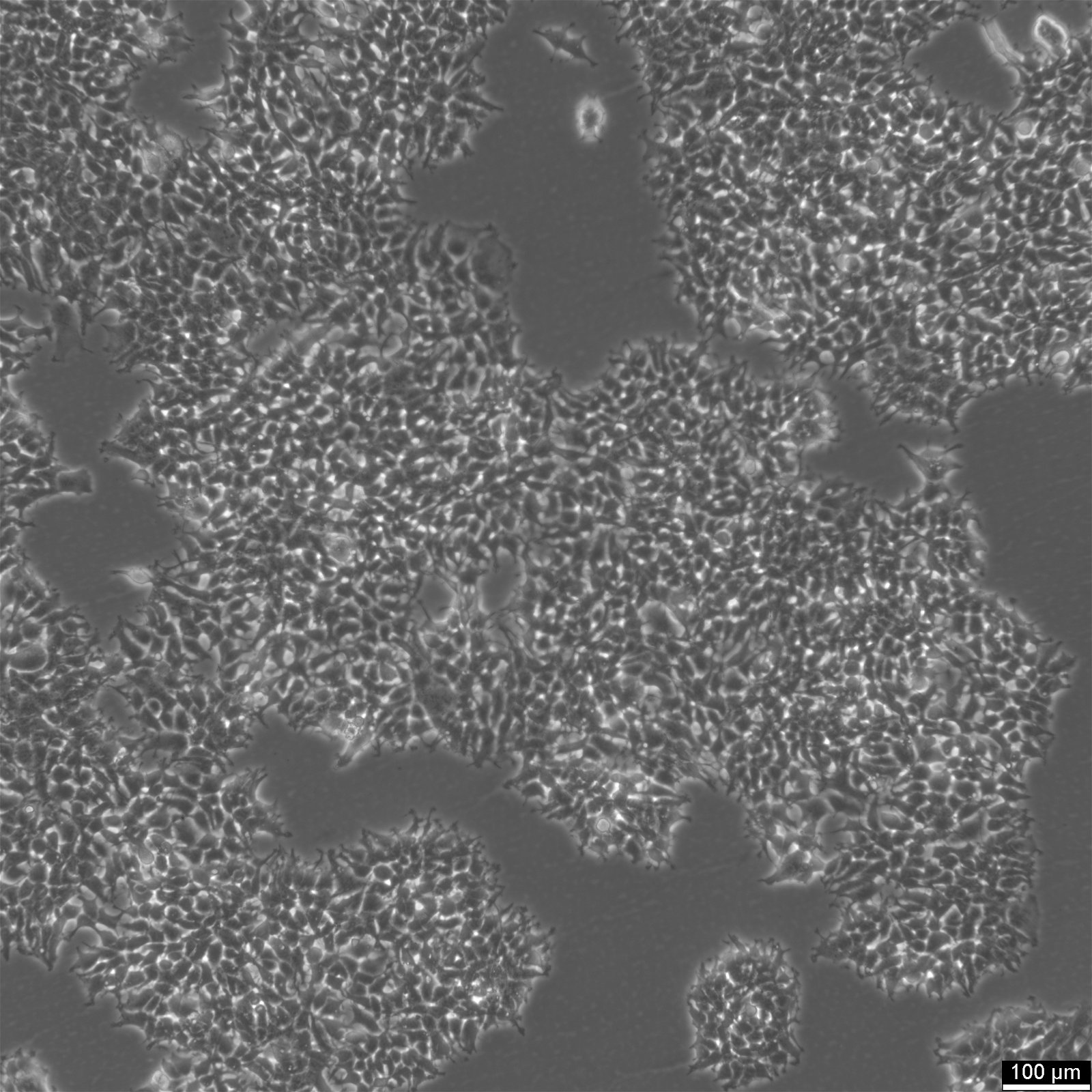
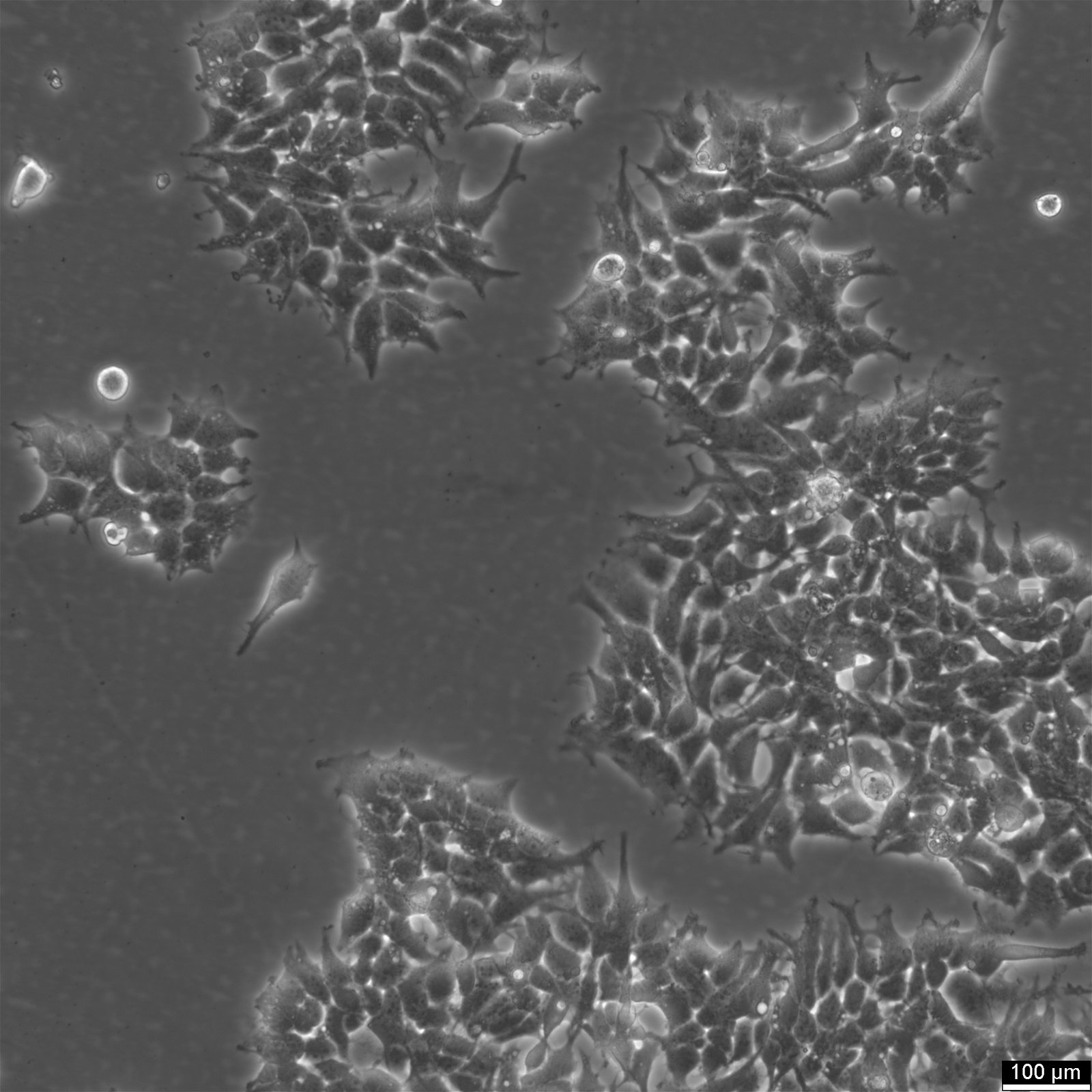
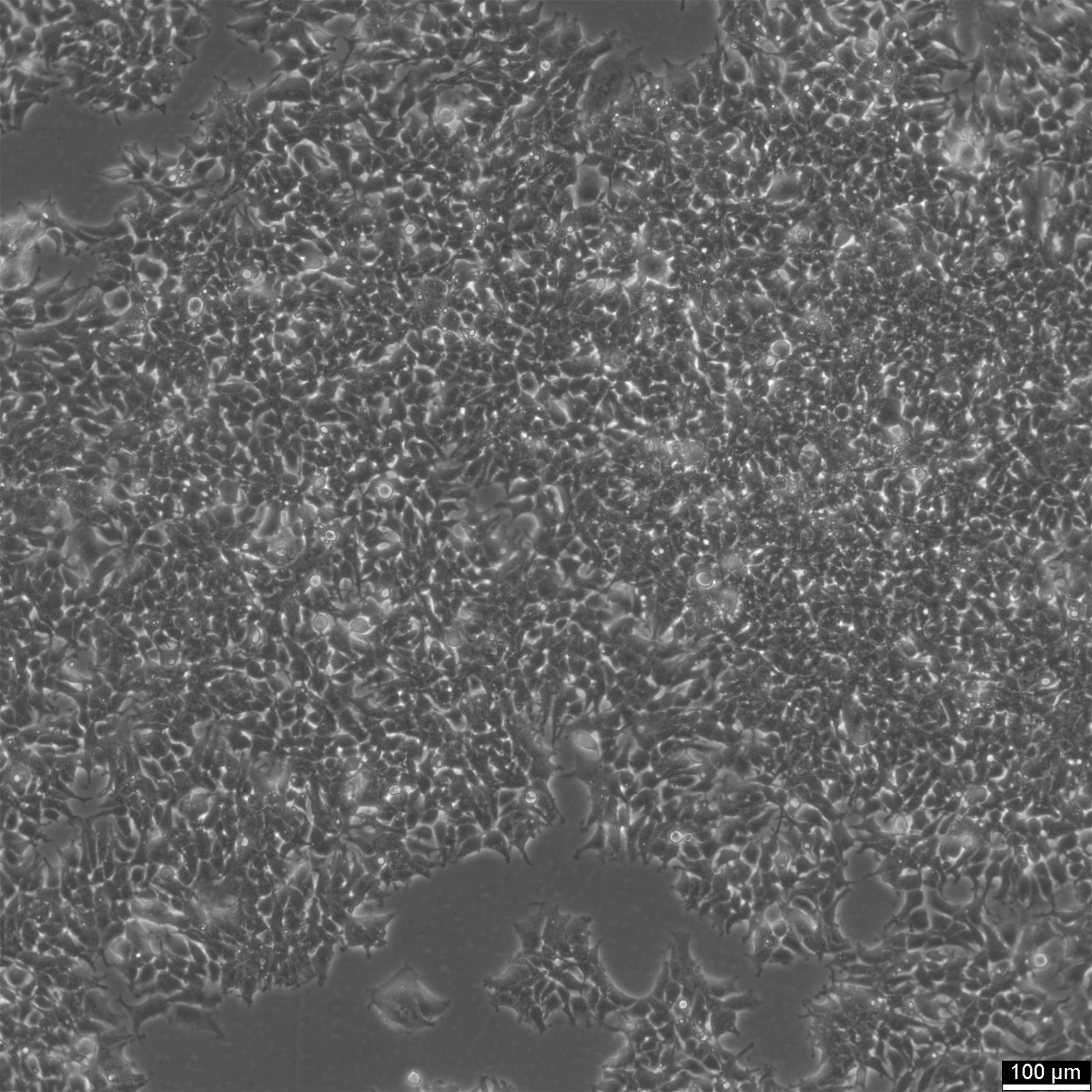
| Description | 2v6.11 cells were derived from the human embryonic kidney line HEK-293 in 2001. The 2V6.11 cell line is a valuable resource for studying the adenoviral E4 oncoprotein, particularly the E4 34K protein known to be involved in cellular genome maintenance and repair. 2V6.11 cells, obtained through transfection with the plasmid pVgRxR followed by pEKORF6, result in the inducible expression of the E4 34K protein, which is linked to the inhibition of cellular mechanisms that repair double strand-breaks in DNA. The 2V6.11 cell line demonstrated that the adenoviral proteins E4 34k and E1b 55k inhibit chromosomal DNA repair by disrupting non-homologous end joining (NHEJ) and destabilizing DNA repair proteins, extending their effect from extrachromosomal to cellular genomic DNA. The 2V6.11 inducible cell line, with their adherent epithelial morphology, are ideal for investigating the behavior and characteristics of kidney-derived epithelial cells, including their response to infections by human adenovirus 40. This versatile cell line, which can be detected by western blot, enables researchers to delve into the molecular mechanisms by which the adenovirus E4 oncoprotein inhibits repair processes, thus contributing to our understanding of adenovirus pathology and the potential for developing new therapeutic strategies. |
|---|---|
| Organism | Human |
| Tissue | Fetal Kidney |
Features
| Age | Fetus |
|---|---|
| Gender | Female |
| Morphology | Epithelial |
| Growth properties | Adherent |
Specifications of the 2V6.11 cell line
| Citation | 2V6.11 (Cytion catalog number 305147) |
|---|---|
| Biosafety level | 2 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_6355 |
| GMO Status | GMO-S1: This HEK293-derived line contains an adenovirus 5 E4-34k expression construct controlled by an ecdysone-inducible promoter, enabling regulated E4 protein production. This classification applies only within Germany and may differ elsewhere. |
Genetic profile
Handling
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality assurance
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305147-170424 | Certificate of Analysis | 23. May. 2025 | 305147 |
| 305147-100323SF | Certificate of Analysis | 23. May. 2025 | 305147 |
| 305147-100323 | Certificate of Analysis | 18. Aug. 2025 | 305147 |
