Farage Cells






CAD$545.10*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
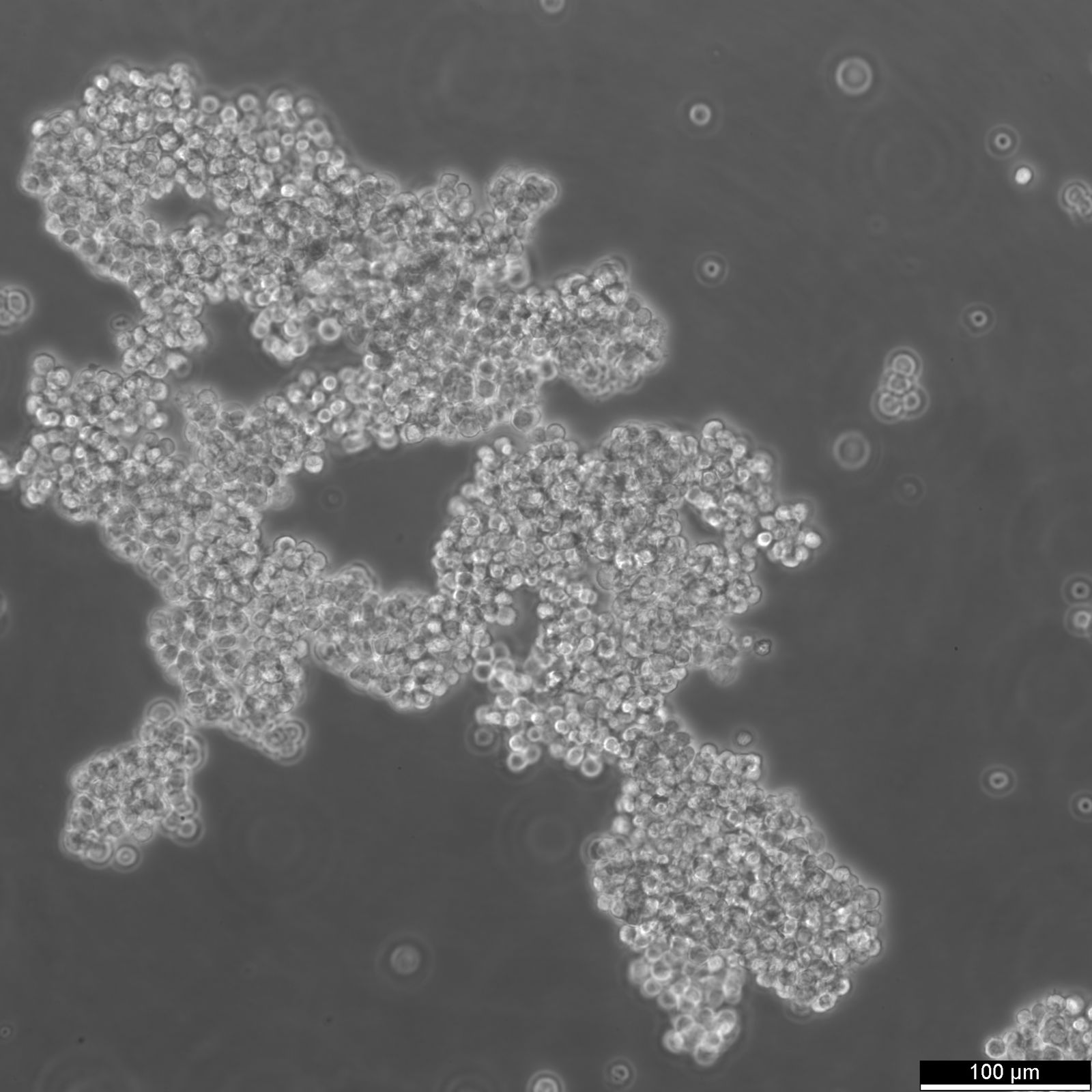
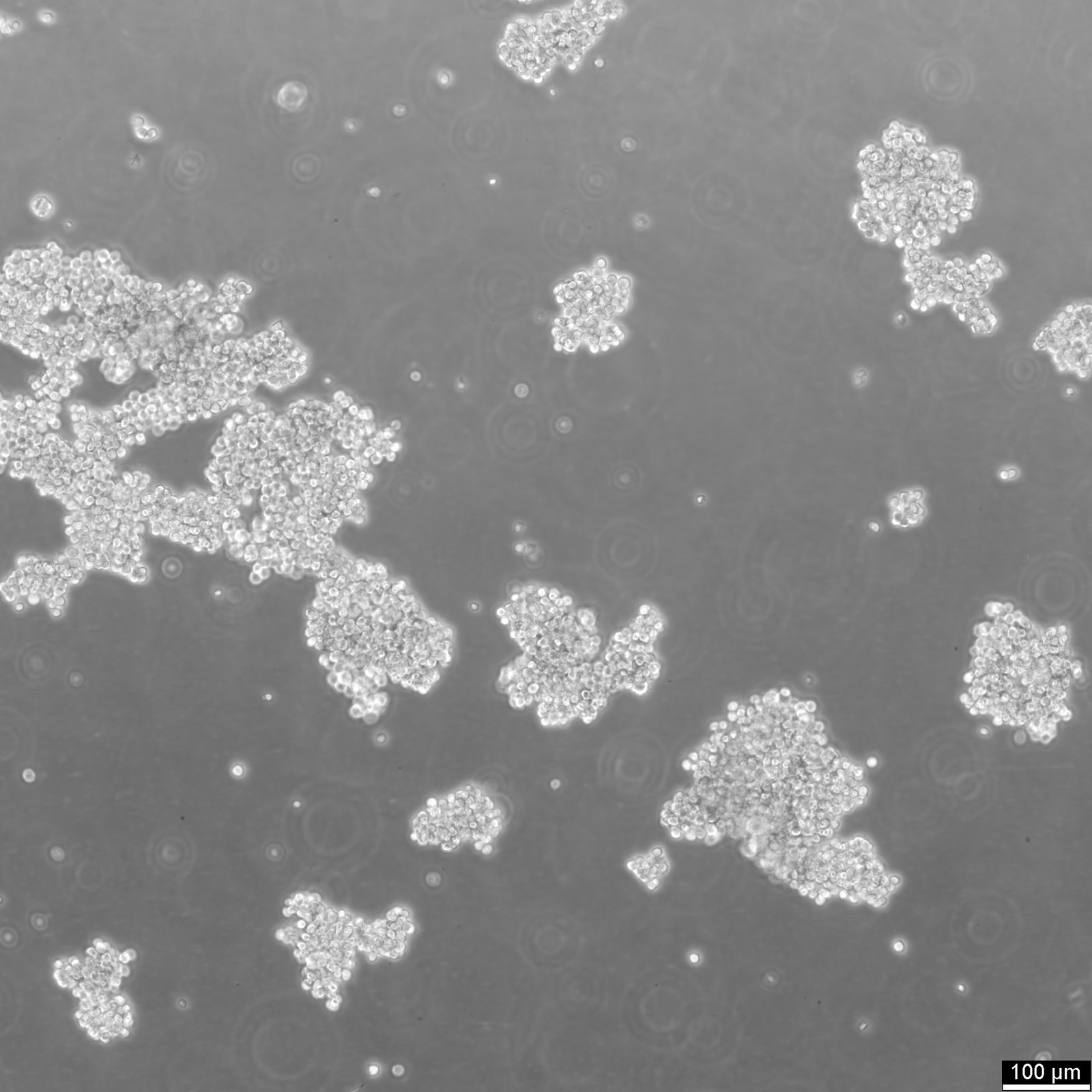
| Description | The Farage cell line originates from a B lymphocyte derived from an adult female diagnosed with non-Hodgkin's B-cell lymphoma. This cell line is particularly valuable in immunological studies due to its unique characteristics and reactions to various stimuli. Farage cells grow in suspension and are notable for not expressing surface or cytoplasmic immunoglobulins, highlighting their utility in studies focused on immune response without the interference of these proteins. When treated with interleukin-4 (IL-4), Farage cells exhibit an increase in the expression of several markers including CD23, CD54, and CD58, while showing a reduction in CD21, CD22, and CD38 levels. This modulation of surface markers suggests IL-4’s role in influencing B-cell behavior and provides a useful model for exploring the signaling pathways and regulatory mechanisms in B-cells. Moreover, the response to phorbol 12-myristate 13-acetate (PMA) treatment, which results in the down-regulation of CD21 and CD23, further supports its application in studying kinase-driven signaling in B-cells. The absence of terminal deoxynucleotidyl transferase (TdT) and recombination activating genes (RAG-1 and RAG-2) in Farage cells confirms their classification as mature B-cells rather than pre-B cells. This aspect is crucial for research targeting the mature stages of B-cell development or function. Additionally, the presence of Epstein-Barr virus (EBV) in these cells can be leveraged in studies investigating viral interactions with host cellular mechanisms, particularly in the context of oncogenic processes in lymphocytes. |
|---|---|
| Organism | Human |
| Tissue | Lymphatic system |
| Disease | Diffuse large B-cell lymphoma germinal center B-cell type |
| Metastatic site | Lymph node |
| Synonyms | FARAGE, Farage OL, Farage Original Line |
Characteristics
| Age | 70 years |
|---|---|
| Gender | Female |
| Ethnicity | European |
| Morphology | Lymphoblast |
| Growth properties | Suspension |
Regulatory Data
| Citation | Farage (Cytion catalog number 305071) |
|---|---|
| Biosafety level | 2 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_3302 |
Biomolecular Data
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% heat-inactivated FBS, add 2.5 g/L glucose and 10 mM HEPES |
| Doubling time | 48 hours |
| Subculturing | Can be cultivated to 1.5-2 x 106 cells/ml. Gently homogenize the cell suspension in the flask by pipetting up and down, then take a representative sample to determine the cell density per ml. Dilute the suspension to achieve a cell concentration of 5 x 105 cells/ml with fresh culture medium, and aliquot the adjusted suspension into new flasks for further cultivation. |
| Seeding density | 5 x 105 cells/ml |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305071-220424 | Certificate of Analysis | 23. May. 2025 | 305071 |
