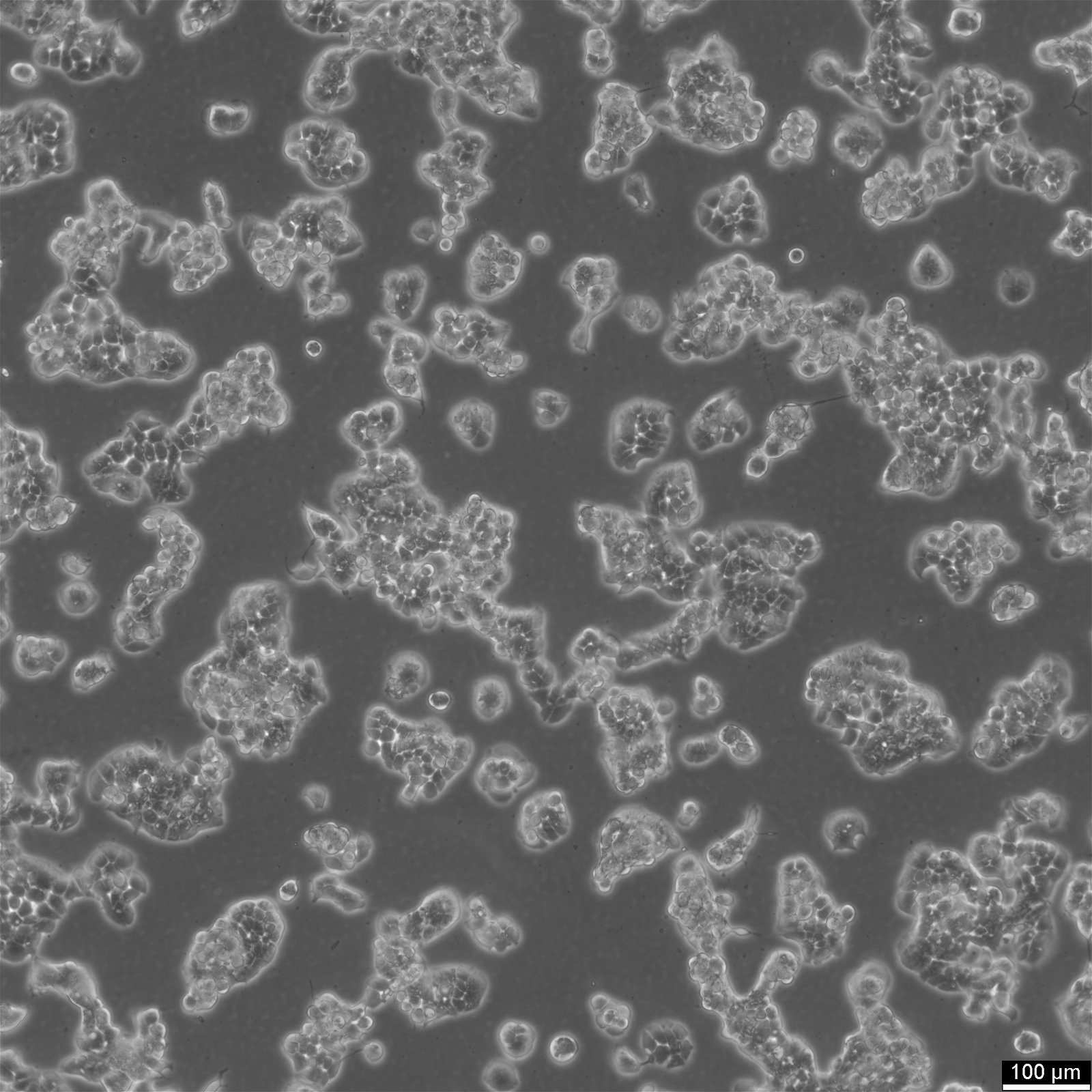
ZR-75-30 Cells



























CAD$759.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | ZR-75-30 is a human breast cancer cell line derived from a ductal carcinoma. Genomic profiling studies have shown that ZR-75-30 harbors amplification of the ERBB2/HER2 gene, a key driver in a subset of breast cancers. This amplification results in elevated HER2 protein expression, which has been linked to increased proliferation and resistance to certain therapies. Additionally, ZR-75-30 exhibits alterations in the epidermal growth factor receptor (EGFR) signaling pathway, including gains of EGFR-related genes, suggesting that the cell line may be useful in studying HER2-targeted therapies and their resistance mechanisms. Transcriptomic analyses have placed ZR-75-30 within the luminal subtype of breast cancer, supporting its relevance for studying endocrine therapy responses. The cell line has been included in studies evaluating precision medicine approaches, where molecular profiling has helped predict responses to targeted treatments. Given its molecular characteristics, ZR-75-30 is widely used as a preclinical model for evaluating hormone receptor-targeted therapies and HER2 inhibitors, making it a valuable tool in breast cancer research. |
|---|---|
| Organism | Human |
| Tissue | Breast, Mammary gland |
| Disease | Invasive breast carcinoma of no special type |
| Metastatic site | Ascites |
| Synonyms | ZR75-30, ZR7530 |
Characteristics
| Age | 47 years |
|---|---|
| Gender | Female |
| Ethnicity | African American |
| Morphology | Epithelial |
| Cell type | Epithelial |
| Growth properties | Adherent |
Regulatory Data
| Citation | ZR-75-30 (Cytion catalog number 305389) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_1661 |
Biomolecular Data
| Mutational profile | Mutation: Gene fusion, APPBP2 + HGNC, PHF20L1, Name(s)=APPBP2-PHF20L1.Gene fusion, BCAS3 + HGNC, HOXB9, Name(s)=BCAS3-HOXB9. Gene fusion, COL14A1 + HGNC, SKAP1, Name(s)=COL14A1-SKAP1. Gene fusion, DDX5 + HGNC, DEPTOR, Name(s)=DDX5-DEPTOR. Gene fusion, BCAS3 + HGNC, ERBB2, Name(s)=ERBB2-BCAS3. Gene fusion, ENPP2 + HGNC, PLEC, Name(s)=PLEC-ENPP2, PLEC1-ENPP2. Gene fusion, PCGF2 + HGNC, TAOK1, Name(s)=TAOK1-PCGF2. Gene fusion, NRIP1 + HGNC, TIAM1, Name(s)=TIAM1-NRIP1. Gene fusion, ARHGAP32 + HGNC, TIMM23, Name(s)=TIMM23-ARHGAP32. Gene fusion, LASP1 + HGNC, TRPS1, Name(s)=TRPS1-LASP1. Gene fusion, CWC25 + HGNC, USP32, Name(s)=USP32-CWC25, USP32-CCDC49. Gene fusion, OPRD1 + HGNC, ZMYM4, Name(s)=ZMYM4-OPRD1. Mutation, BRAF, Simple, p.Ile326Thr (c.977T>C), Heterozygous, CDH1, Simple, p.Glu243Ter (c.727G>T), Homozygous. |
|---|
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS, 10 µg/ml Insulin |
| Doubling time | 110 hours |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305389-180325 | Certificate of Analysis | 23. May. 2025 | 305389 |