RAG Cells










CAD$545.10*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
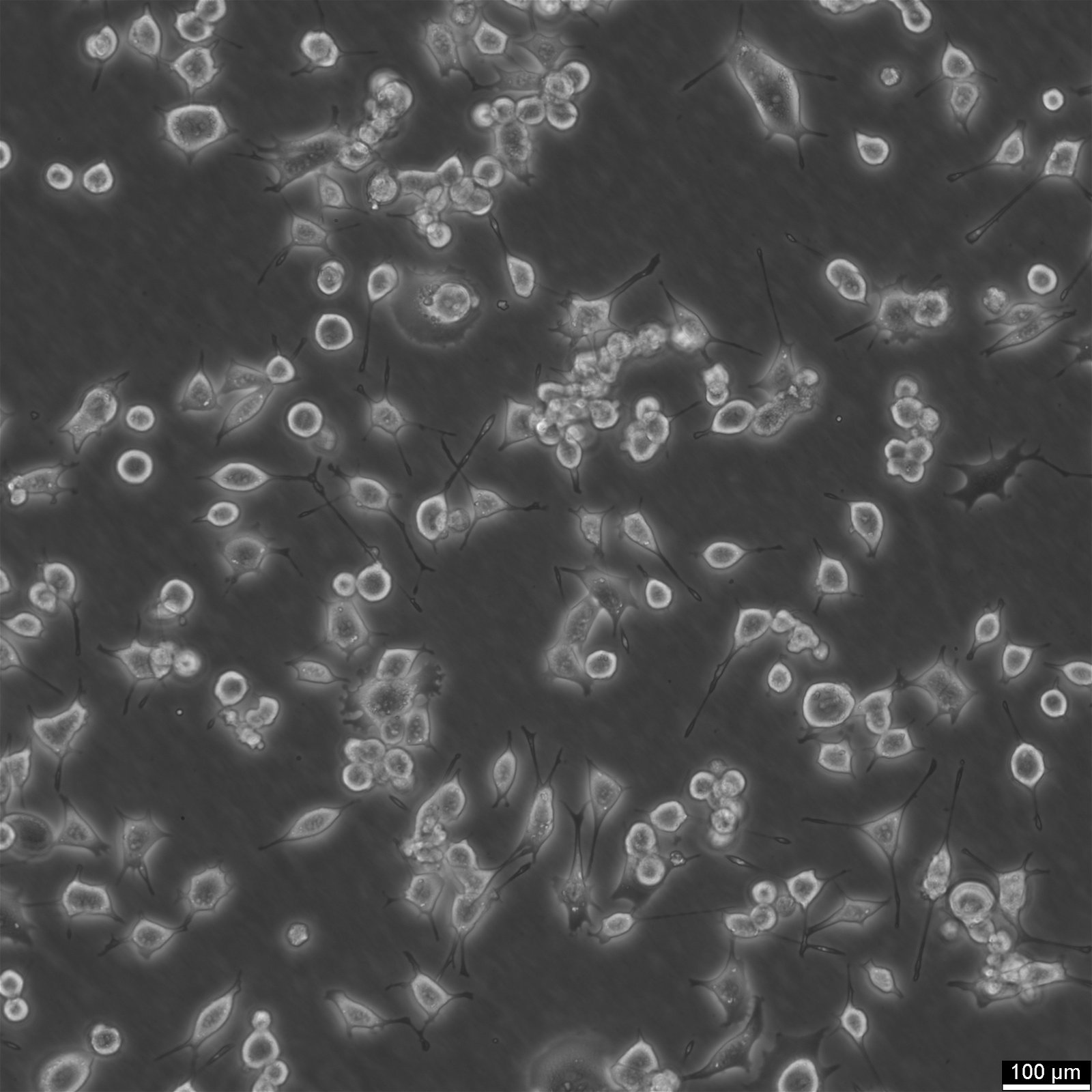
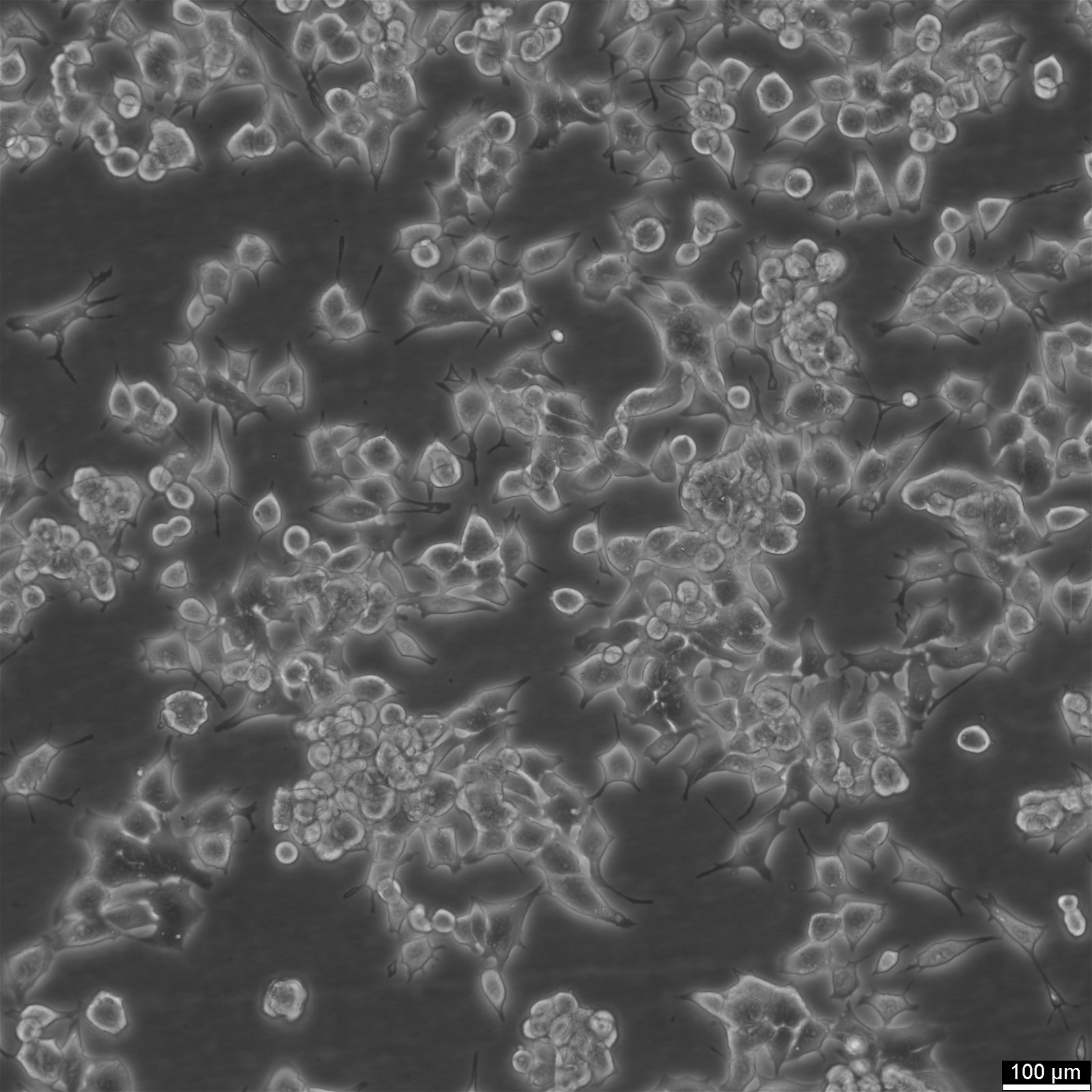
| Description | The RAG cell line is a nonreverting 8-azaguanine-resistant mutant derived from a renal adenocarcinoma of BALB/c mice. This line was developed through alternate animal-to-tissue culture passages to enrich for the tumorigenic population while eliminating normal stromal fibroblasts. RAG cells display an ameboid to epithelioid morphology with prominent cytoplasmic processes and are resistant to hypoxanthine-guanine phosphoribosyltransferase (HGPRT)-dependent selection methods due to their enzymatic deficiency. This resistance has facilitated their use in biochemical selection systems for somatic cell hybridization experiments. RAG cells are widely utilized as a parental line in somatic cell fusion studies due to their compatibility with fusion procedures using inactivated Sendai virus. When fused with other cell lines, such as LM(TK-) or WI-38, the hybrids retain marker chromosomes and exhibit biochemical complementation of metabolic deficiencies. These hybrids have been instrumental in mapping genetic regulatory elements and studying gene expression, particularly in kidney-associated enzymes like the ES-2 esterase. RAG hybrids provide insights into both inter- and intraspecific chromosomal segregation and functional genomics. In addition to their role in hybridization studies, RAG cells have served as a model to study the epigenetic regulation of gene expression. Hybrid cells involving RAG often show extinction and re-expression of specific genetic traits, depending on the retention or loss of particular chromosomes. This makes the RAG cell line a valuable tool in understanding the dynamics of genetic regulation and chromosomal stability in tumorigenic cells. |
|---|---|
| Organism | Mouse |
| Tissue | Kidney |
| Disease | Mouse kidney carcinoma |
| Synonyms | Rag |
Characteristics
| Breed/Subspecies | BALB/c |
|---|---|
| Morphology | Amoeboid |
| Growth properties | Adherent |
Regulatory Data
| Citation | RAG (Cytion catalog number 305190) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 10090 |
| CellosaurusAccession | CVCL_3575 |
Biomolecular Data
| Protein expression | Kidney specific esterase-2 (ES-2) |
|---|
Handling
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
