PC-9 Cells











CAD$759.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | The PC-9 cell line is derived from a human lung adenocarcinoma, a subtype of non-small cell lung cancer (NSCLC). This cell line is particularly notable for harboring an activating mutation in the EGFR gene, specifically the exon 19 deletion (E746_A750del), which is a common driver mutation in NSCLC. This alteration makes PC-9 an invaluable model for studying the biology of EGFR-driven cancers and evaluating the efficacy of tyrosine kinase inhibitors (TKIs) like gefitinib and erlotinib, which specifically target this pathway. PC-9 cells have been extensively used in research focused on resistance mechanisms to EGFR TKIs, particularly the emergence of secondary mutations like T790M. These studies have informed the development of third-generation inhibitors such as osimertinib, which target both the primary EGFR mutation and resistance-associated alterations. The cell line also exhibits sensitivity to other inhibitors targeting downstream signaling pathways, including those involved in PI3K/AKT and MAPK signaling cascades, underscoring its utility in translational cancer research. In addition to its genetic and pharmacological attributes, PC-9 has been incorporated into high-throughput drug screening programs, facilitating the identification of compounds with selective activity against EGFR-mutated NSCLC. The line's well-characterized genomic landscape and consistent phenotypic behavior in vitro make it a cornerstone for both basic and applied lung cancer research, particularly in the context of targeted and combination therapy. |
|---|---|
| Organism | Human |
| Tissue | Lung |
| Disease | Lung adenocarcinoma |
| Metastatic site | Lymph node |
| Synonyms | PC9, PC-9/S1, PC-9S1 |
Characteristics
| Age | 45 years |
|---|---|
| Gender | Male |
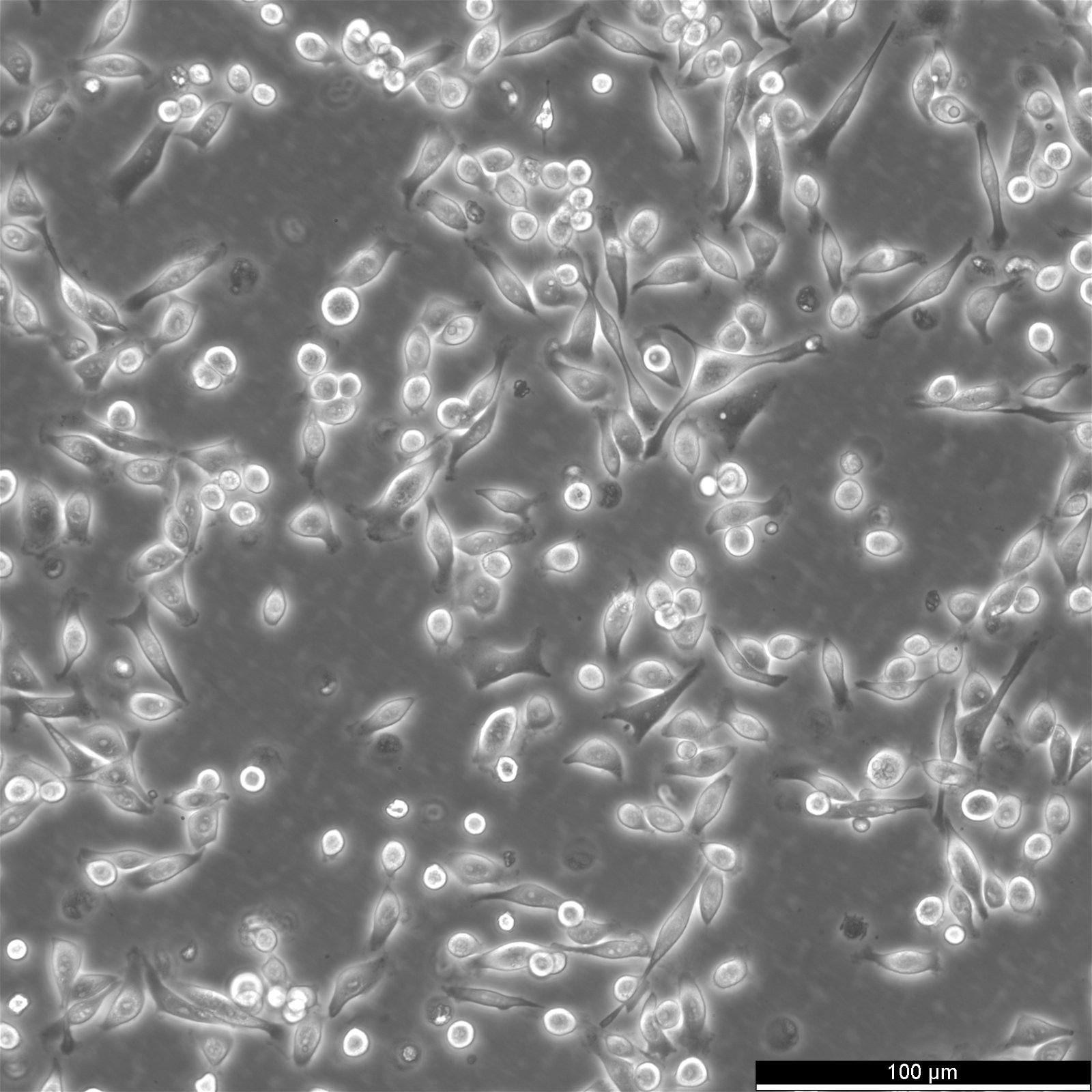
| Morphology | Heterogeneous mixture of round cells and spindle shaped cells |
| Growth properties | Adherent |
Regulatory Data
| Citation | PC-9 (Cytion catalog number 305045) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_B260 |
Biomolecular Data
| Tumorigenic | Yes |
|---|
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Subculturing | Gather the suspension cells in a 15 ml tube and gently wash the adherent cells with PBS lacking calcium and magnesium (use 3-5 ml for T25 flasks and 5-10 ml for T75 flasks). Apply Accutase (1-2 ml for T25 flasks, 2.5 ml for T75 flasks) ensuring full coverage of the cell layer. Allow the cells to incubate at 37°C for 10-15 minutes. Following incubation, combine and centrifuge both the suspension and adherent cells. After centrifugation, carefully resuspend the cell pellet and transfer the cell suspension into new flasks containing fresh medium. |
| Fluid renewal | 1 to 2 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305045-211122 | Certificate of Analysis | 23. May. 2025 | 305045 |
| 305045-110825 | Certificate of Analysis | 22. Oct. 2025 | 305045 |
