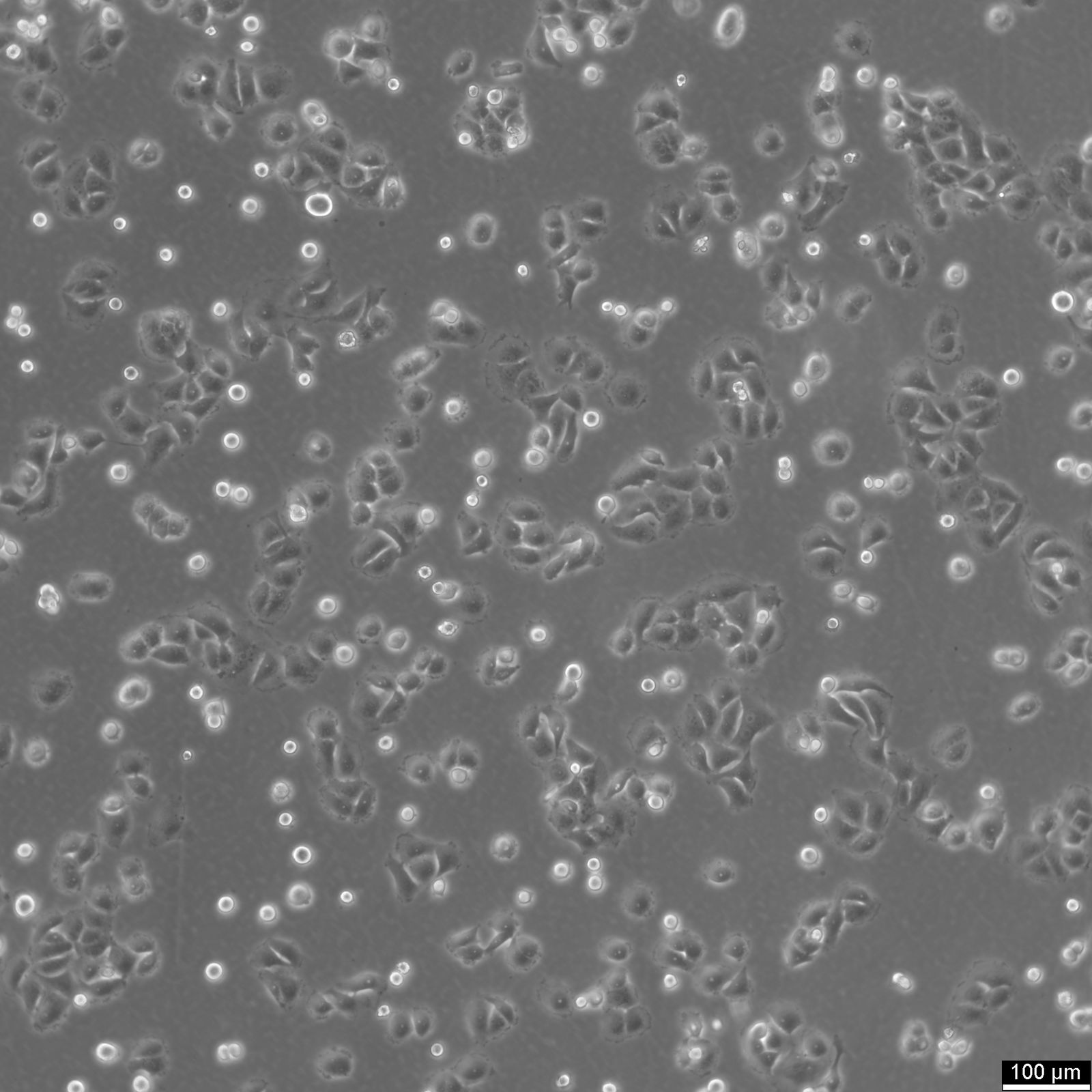
OVCAR-8 Cells















CAD$897.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | OVCAR-8 is a human ovarian carcinoma cell line established from a patient with advanced-stage ovarian adenocarcinoma. This cell line is particularly noted for its significant resistance to cisplatin and carboplatin, which were administered in high doses during the patient’s treatment. OVCAR-8 is widely utilized in research investigating mechanisms of chemoresistance in ovarian cancer, as well as in the development of strategies to overcome resistance to platinum-based chemotherapies. OVCAR-8 cells exhibit an epithelial morphology and grow adherently in culture. The cell line is characterized by molecular and phenotypic traits associated with high-grade ovarian cancers, including alterations in DNA damage repair mechanisms and other pathways contributing to tumor survival under chemotherapeutic stress. Unlike some other ovarian cancer cell lines, OVCAR-8 does not exhibit detectable expression of metallothionein, a protein thought to play a role in resistance to heavy metal-based drugs. However, this cell line demonstrates cross-resistance to cadmium and other agents, suggesting the involvement of alternative resistance mechanisms, such as increased glutathione levels and enhanced DNA repair capacity. OVCAR-8 is a valuable tool in preclinical research for screening chemotherapeutic agents, evaluating targeted therapies, and studying the biology of chemoresistance. Researchers employ this cell line to explore combinations of drugs designed to sensitize resistant tumors to standard treatments. Additionally, OVCAR-8 provides insights into the genetic and molecular adaptations of ovarian cancer cells that underlie their survival and persistence despite aggressive chemotherapy regimens. Its clinical relevance and resistance profile make it an important resource for advancing ovarian cancer research and therapy development. |
|---|---|
| Organism | Human |
| Tissue | Ovary |
| Disease | Ovarian adenocarcinoma |
| Synonyms | OVCAR 8, NIH:OVCAR-8, OVCAR8, Ovcar8, OVCAR.8, OVCA8, OVCAR-8/EGFP_LC3 |
Characteristics
| Age | 64 years |
|---|---|
| Gender | Female |
| Ethnicity | Caucasian |
| Morphology | Epithelial-like |
| Growth properties | Adherent |
Regulatory Data
| Citation | OVCAR-8 (Cytion catalog number 305383) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_1629 |
Biomolecular Data
| Mutational profile | Mutation: CTNNB1, Simple, p.Gln26Arg (c.77A>G), Heterozygous; Mutation: ERBB2, Simple, p.Gly776Val (c.2327G>T), Heterozygous; Mutation: KRAS, Simple, p.Pro121His (c.362C>A), Heterozygous; Mutation: TP53, Simple, c.376-1G>A (p.Tyr126_Lys132del, c.376_396del21), Homozygous, Splice acceptor mutation |
|---|
Handling
| Culture Medium | RPMI 1640, w: 2.1 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Dissociation Reagent | Accutase |
| Doubling time | 24-32 hours |
| Seeding density | 3-4 x 104 cells/mL |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 305383-060225 | Certificate of Analysis | 23. May. 2025 | 305383 |
| 305383-250925 | Certificate of Analysis | 05. Dec. 2025 | 305383 |
| 305383-110326 | Certificate of Analysis | 13. Apr. 2026 | 305383 |
