OK Cells












CAD$545.10*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
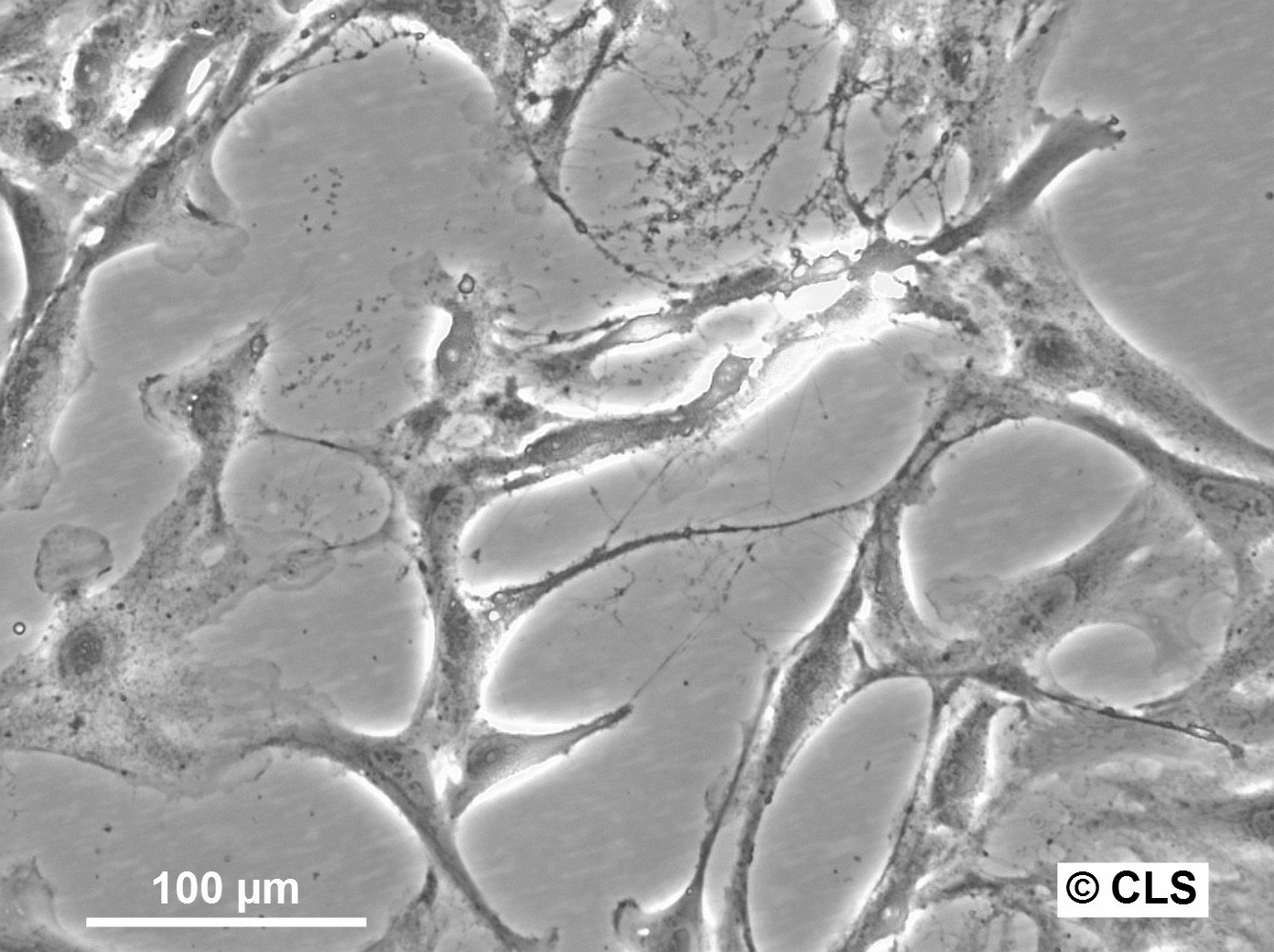
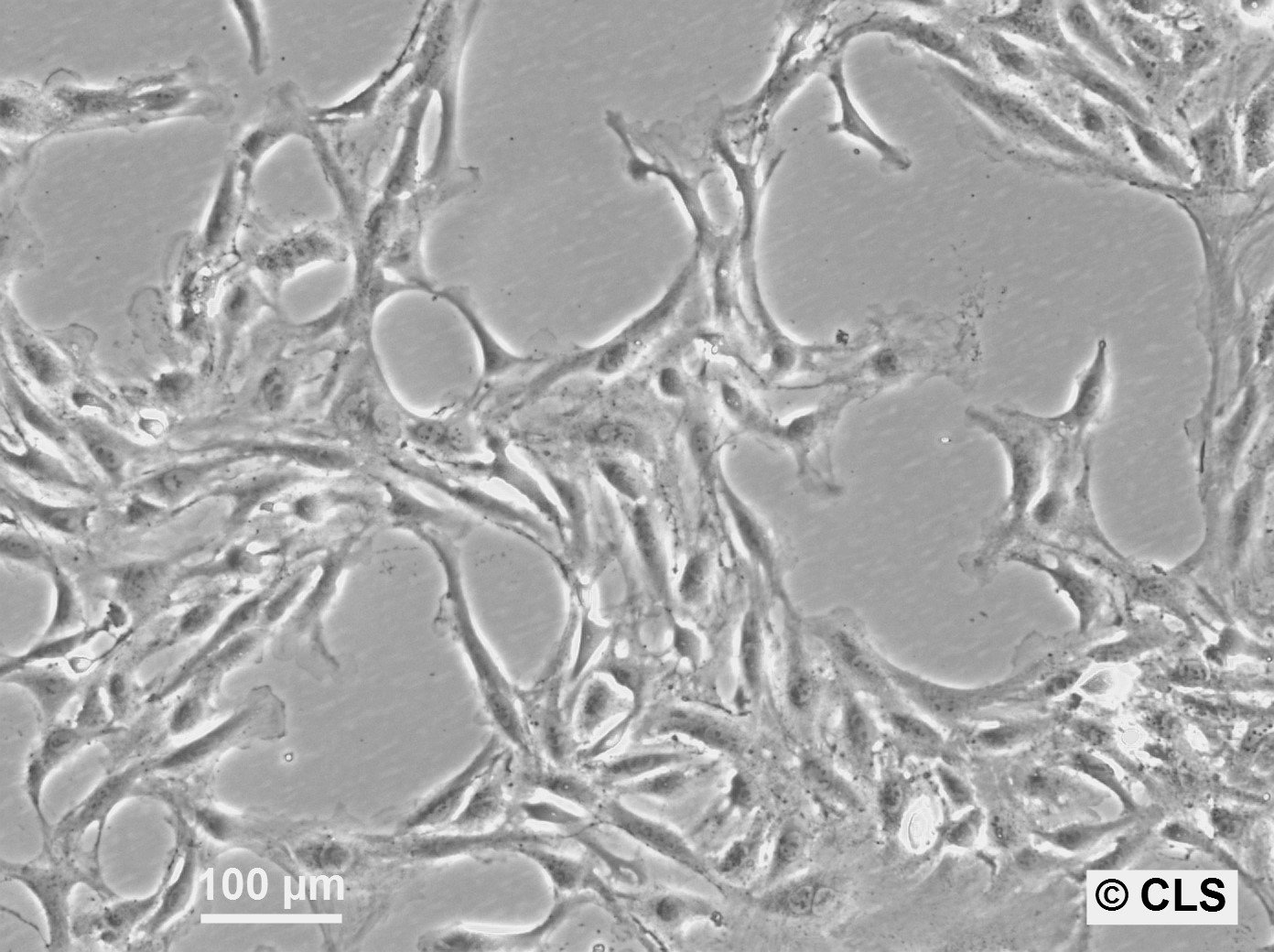
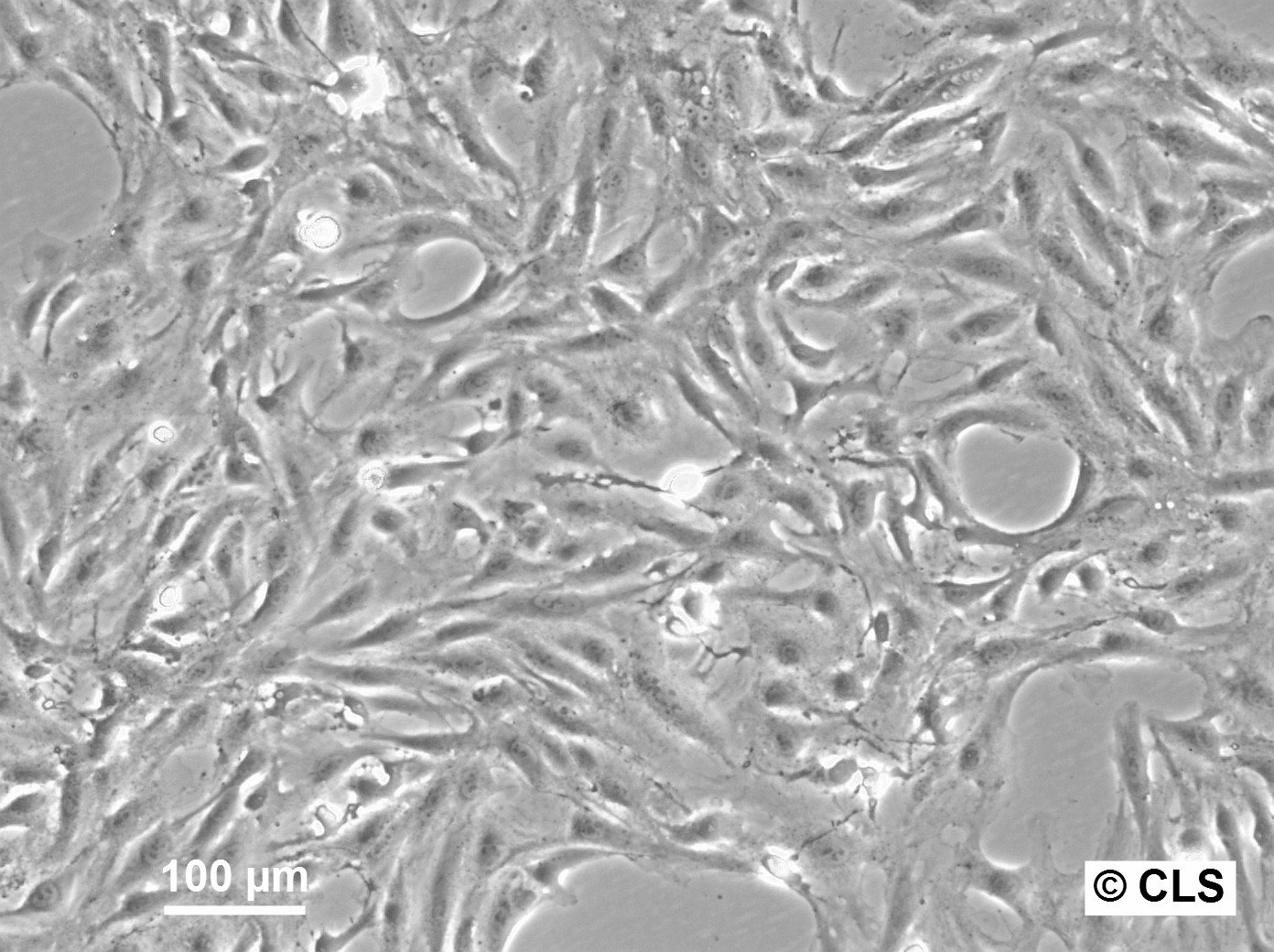
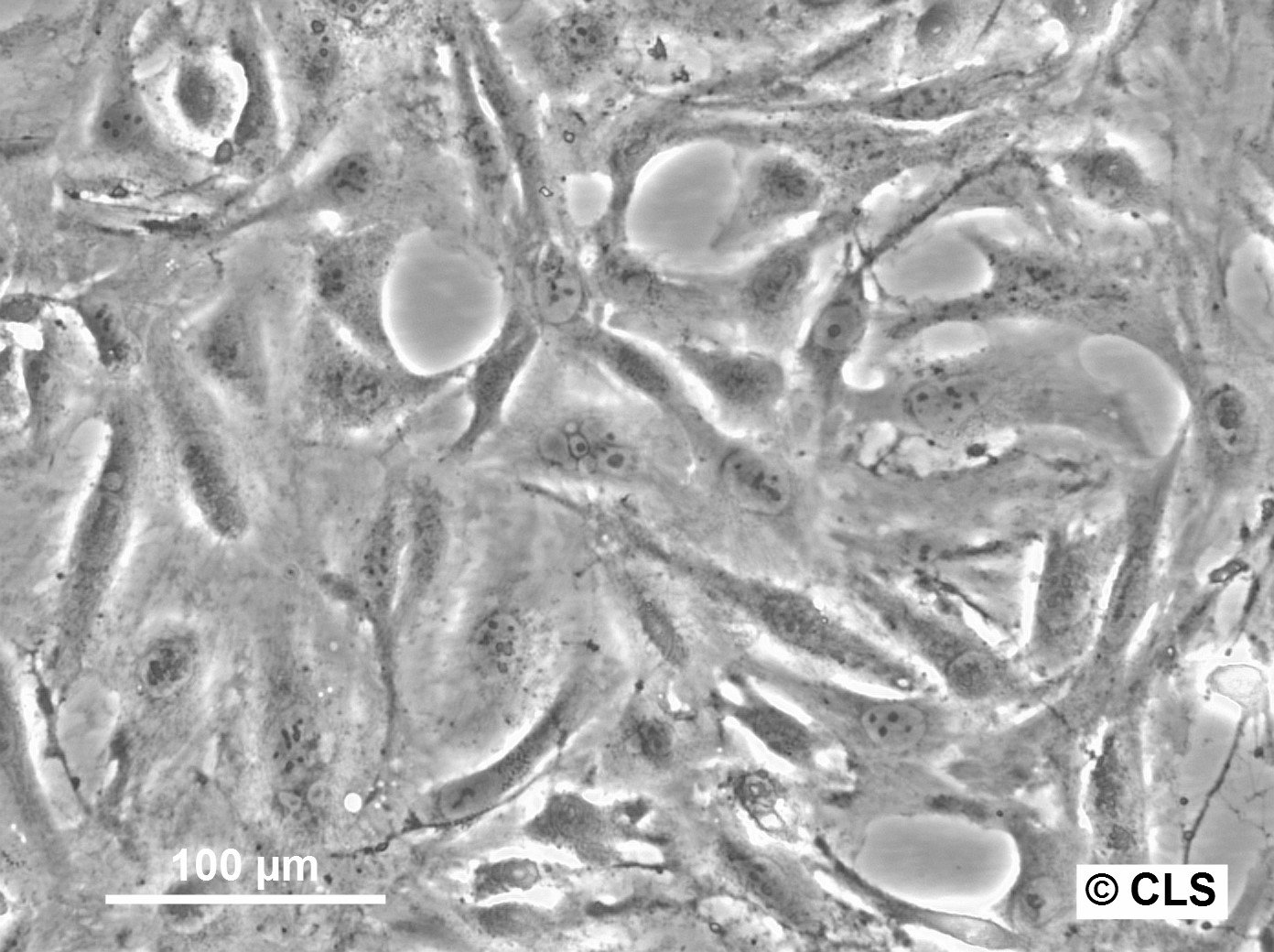
| Description | The OK cell line is a permanent epithelial-like cell culture derived from kidney tissue of an adult female American opossum (Didelphis virginiana). Established in vitro, this cell line is notable for its non-diploid chromosomal modal number of 23 and its adaptability to tissue culture conditions. Initially derived from mixed cell types, the culture evolved into predominantly epithelial cells after eight passages. The OK cell line has been extensively characterized in terms of morphology, chromosomal constitution, and growth dynamics, making it a robust model for cytogenetic and chromosome isolation studies. One of the key features of the OK cell line is its utility in chromosome studies, especially for isolating the mammalian X chromosome. The opossum X chromosome is significantly smaller (approximately 30% smaller than the smallest autosomes) and does not contain large blocks of constitutive heterochromatin, facilitating separation from autosomes through techniques like flow microfluorometry and gradient centrifugation. The stable karyotype of the OK cells, with the presence of a distinctive metacentric marker chromosome, enhances their application in genomic and chromosomal studies. The paternal X chromosome's preferential inactivation in this marsupial provides a comparative model for studying mechanisms underlying X-chromosome inactivation in mammals. OK cells have also demonstrated resilience and adaptability in various culture conditions, including serum variations and different mitotic-arresting agents like Velban (vinblastine sulfate), which is particularly effective for achieving high mitotic indices for chromosome isolation. The cell line's ability to synchronize and produce high yields of metaphase cells further underscores its suitability for detailed chromosomal analyses, including DNA content quantification and high-resolution imaging of chromosomal spreads. |
|---|---|
| Organism | Opossum |
| Tissue | Kidney, cortex, Proximal tubule |
| Synonyms | Opossum Kidney, OK-WT |
Characteristics
| Age | Adult |
|---|---|
| Gender | Female |
| Morphology | Epithelial-like |
| Growth properties | Monolayer, adherent |
Regulatory Data
| Citation | OK (Cytion catalog number 606465) |
|---|---|
| Biosafety level | 1 |
| NCBI_TaxID | 9267 |
| CellosaurusAccession | CVCL_0472 |
Biomolecular Data
| Receptors expressed | Alpha2-adrenergic, serotonin, parathyroid hormone, atrial natriuretic factor |
|---|
Handling
| Culture Medium | EMEM (MEM Eagle), w: 2 mM L-Glutamine, w: 2.2 g/L NaHCO3, w: EBSS (Cytion article number 820100a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS and 1% NEAA |
| Dissociation Reagent | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 606465-511 | Certificate of Analysis | 23. May. 2025 | 606465 |
