IM-9 Cells



























CAD$545.10*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
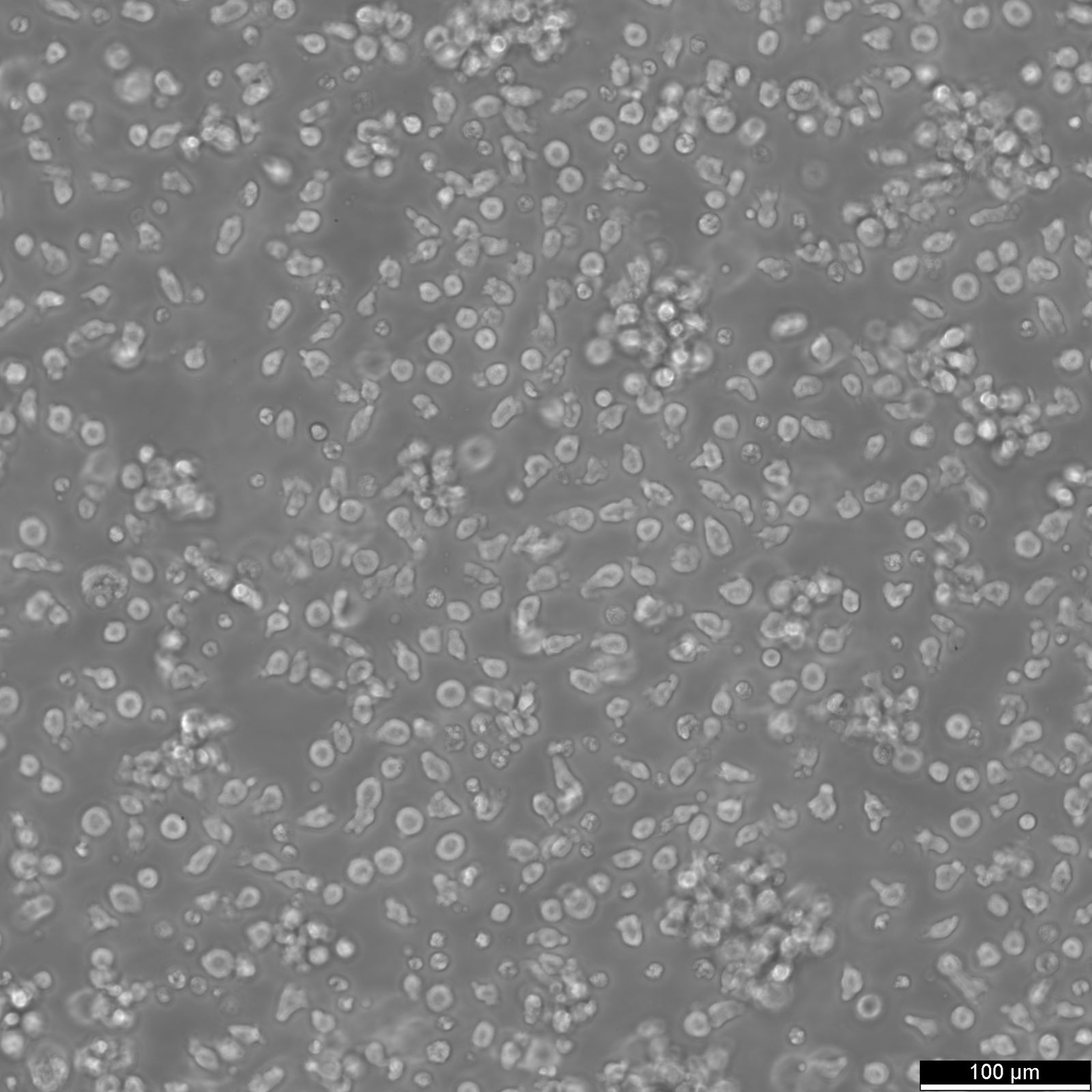
| Description | IM-9 is a human lymphoblastoid cell line established in 1967 from the bone marrow of an adult woman diagnosed with multiple myeloma. Originally believed to be derived from myeloma cells, subsequent research, including findings published by Pellat-Deceunynk et al. in 1995, has indicated that IM-9 cells are more accurately classified as Epstein-Barr Virus-positive (EBV+) B-lymphoblastoid cells rather than malignant myeloma cells. This distinction is crucial for researchers using this cell line, as it affects the interpretation of experimental outcomes related to myeloma studies. IM-9 cells have been extensively characterized in the literature and are noted for synthesizing Immunoglobulin G (IgG). They are also known to express receptors for insulin and calcitonin, making them valuable for studying hormone-receptor interactions. Additionally, these cells express BCL2 mRNA, a gene involved in regulating apoptosis, which is often studied in the context of cancer and immune cell survival. Due to their high expression of insulin receptors, IM-9 cells are frequently used in research focused on insulin signaling and metabolic disorders, providing insights into insulin resistance mechanisms. The IM-9 cell line remains a significant resource for various research applications, particularly in the fields of immunology, cancer biology, and metabolic studies. However, given the revised understanding of their origin, it is critical to use IM-9 cells with the recognition that they are not representative of malignant myeloma cells. As always, these cells are intended exclusively for in vitro research and are not suitable for therapeutic or in vivo use. |
|---|---|
| Organism | Human |
| Tissue | Bone marrow |
| Synonyms | IM 9, IM9, GM04680 |
Characteristics
| Age | Unspecified |
|---|---|
| Gender | Female |
| Ethnicity | Caucasian |
| Morphology | Round cells in cluster |
| Cell type | B lymphoblast |
| Growth properties | Suspension |
Regulatory Data
| Citation | IM-9 (Cytion catalog number 302151) |
|---|---|
| Biosafety level | 2 |
| NCBI_TaxID | 9606 |
| CellosaurusAccession | CVCL_1305 |
Biomolecular Data
| Antigen expression | CD19+, CD20+, CD23+, CD27+, CD80+, CD83+, CD138+, MHC I+, MHC II+ |
|---|---|
| Viruses | EBV+ free of human pathogenic viruses SV40, JC/BK, HBV, HCV, HIV. |
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% heat-inactivated FBS |
| Subculturing | Gently homogenize the cell suspension in the flask by pipetting up and down, then take a representative sample to determine the cell density per ml. Dilute the suspension to achieve a cell concentration of 1 x 105 cells/ml with fresh culture medium, and aliquot the adjusted suspension into new flasks for further cultivation. |
| Fluid renewal | 2 to 3 times per week |
| Post-Thaw Recovery | Fast |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
Certificate of Analysis (CoA)
| Lot Number | Certificate Type | Date | Catalog Number |
|---|---|---|---|
| 302151-270225 | Certificate of Analysis | 23. May. 2025 | 302151 |
