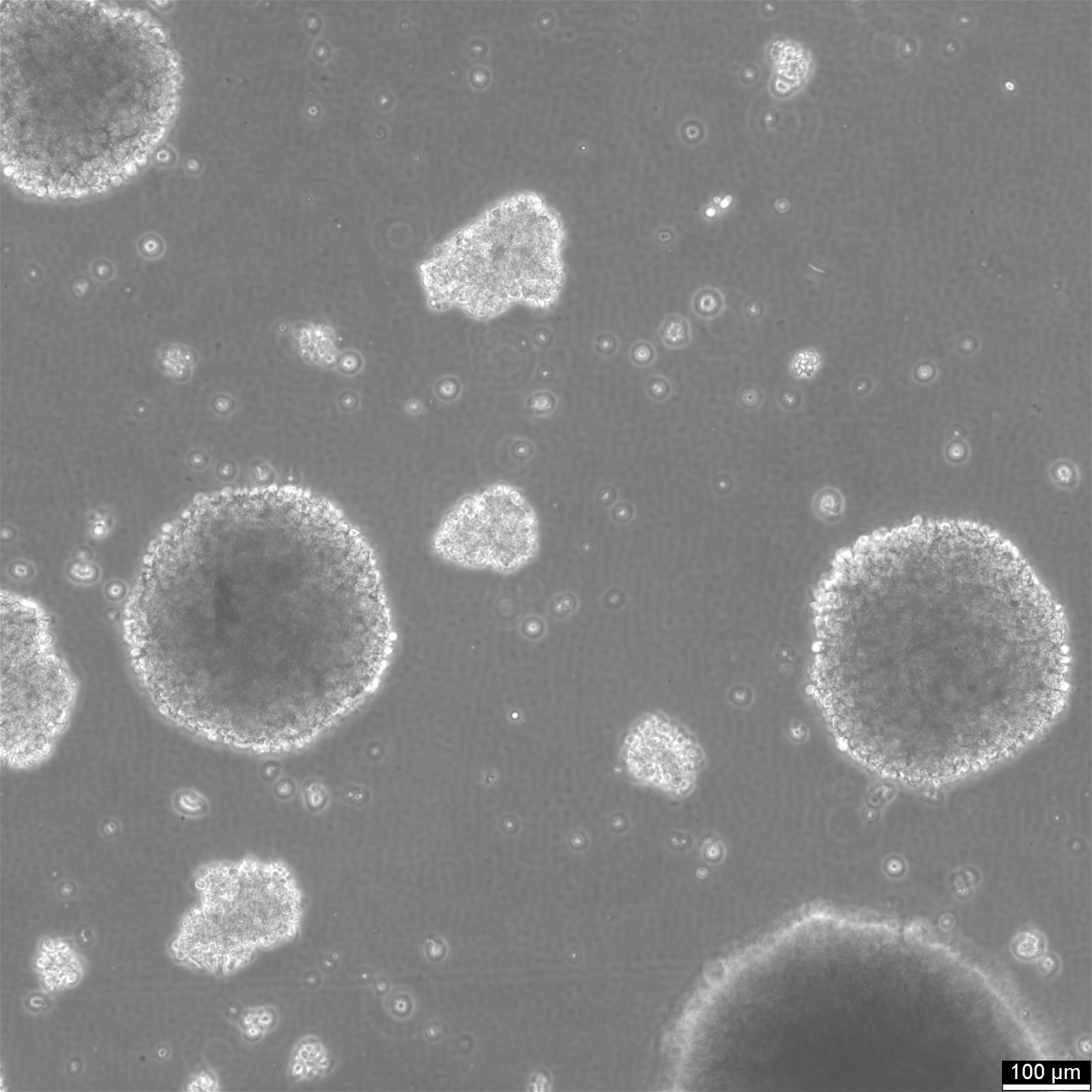
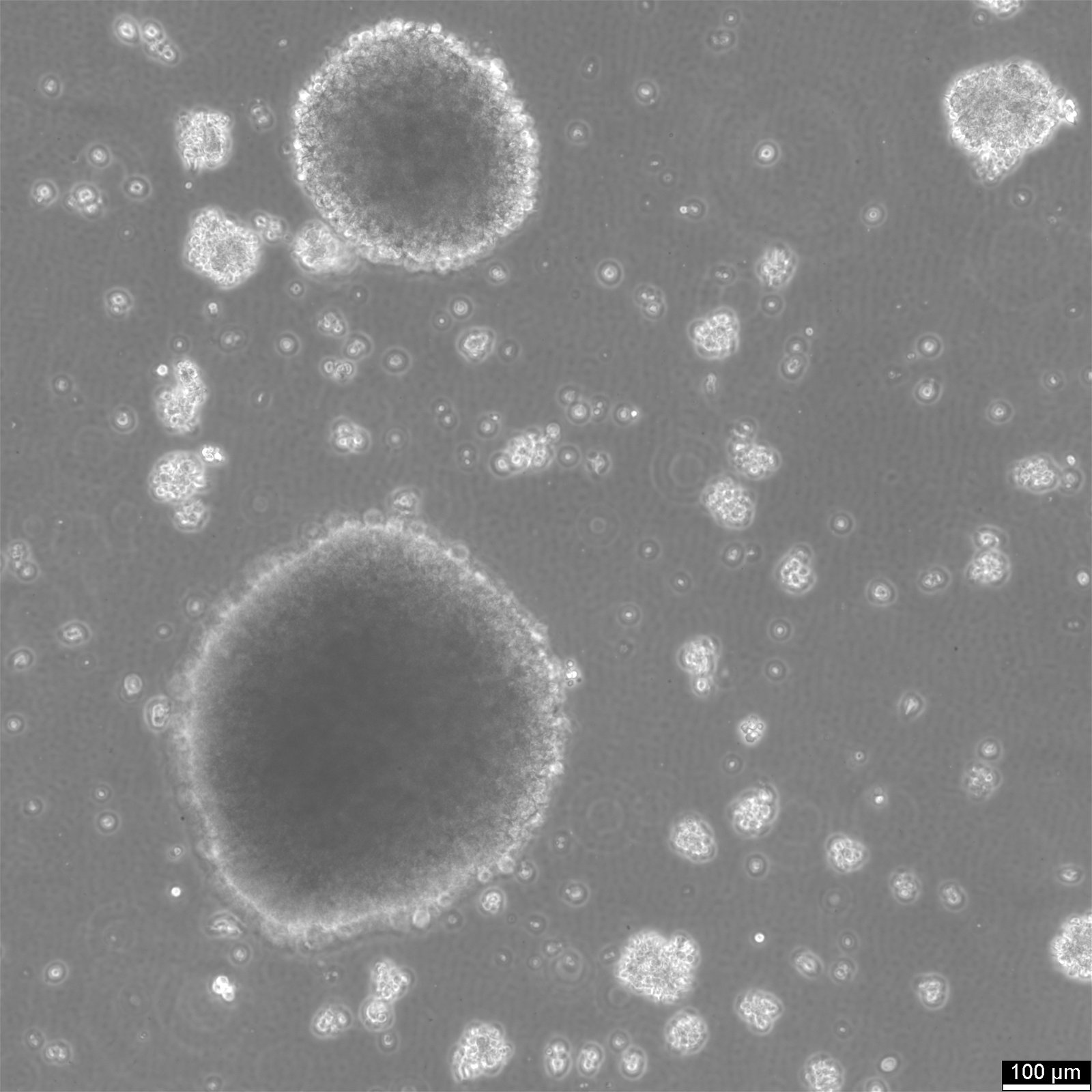
B-LCL-HROC112 Cells






CAD$1,104.00*
Products are shipped frozen on dry ice in cryotubes. Each cryotube typically contains 3 × 106 cells for adherent lines or 5 × 106 cells for suspension lines (refer to the batch CoA for details).
General information
| Description | B-LCL-HROC112 is an Epstein-Barr virus (EBV)-immortalized human B lymphoblastoid cell line established from B lymphocytes isolated from either tumor tissue or peripheral blood of an adult patient. The cells were generated by ex vivo infection with EBV-containing supernatant derived from the B95/8 marmoset cell line in the presence of cyclosporin A to suppress T- and NK-cell outgrowth. Following several weeks of culture, stable lymphoblastoid outgrowth was achieved, resulting in a continuously proliferating monoclonal or oligoclonal B-cell population suitable for long-term in vitro expansion. Immunophenotypically, B-LCL-HROC112 exhibits a mature and activated B-cell profile characterized by expression of CD19 and CD20, along with high levels of activation and maturation markers such as CD23 and CD80. Strong expression of MHC class I and class II molecules indicates preserved antigen-presenting capacity. Depending on the individual clone, variable expression of differentiation-associated markers such as CD27, CD38, or CD138 may be observed, reflecting different stages of B-cell maturation. The cells are negative for T-cell markers, confirming lineage specificity. Functionally, B-LCL-HROC112 secretes immunoglobulin of a defined isotype (e.g., IgG, IgM, or IgA), which remains stable during prolonged culture. The secreted antibodies can be collected from culture supernatants and used for downstream applications, including antigen-binding assays, tumor cell recognition studies, or identification of disease-associated antigens. As an EBV-immortalized B-cell model, B-LCL-HROC112 provides a robust in vitro platform for investigating humoral immune responses, B-cell activation and differentiation, and antibody-mediated mechanisms in the context of tumor immunology or systemic immune responses. |
|---|---|
| Organism | Human |
| Tissue | Peripheral blood |
| Disease | Carcinoma |
| Synonyms | B-LCL CO112, Bc HROC112 |
Characteristics
| Age | 80 years |
|---|---|
| Gender | Female |
| Morphology | Round cells |
| Cell type | B lymphoblast |
| Growth properties | Suspension |
Regulatory Data
| Citation | B-LCL-HROC112 (Cytion catalog number 302023) |
|---|---|
| Biosafety level | 2 |
| NCBI_TaxID | 9606 |
Biomolecular Data
| Viruses | Transformant: EBV |
|---|
Handling
| Culture Medium | RPMI 1640, w: 2.0 mM stable Glutamine, w: 2.0 g/L NaHCO3 (Cytion article number 820700a) |
|---|---|
| Supplements | Supplement the medium with 10% FBS |
| Subculturing | Gently homogenize the cell suspension in the flask by pipetting up and down, then take a representative sample to determine the cell density per ml. Dilute the suspension to achieve a cell concentration of 1 x 105 cells/ml with fresh culture medium, and aliquot the adjusted suspension into new flasks for further cultivation. |
| Freeze medium | As a cryopreservation medium, we use complete growth medium (including FBS) + 10% DMSO for adequate post-thaw viability, or CM-1 (Cytion catalog number 800100), which includes optimized osmoprotectants and metabolic stabilizers to enhance recovery and reduce cryo-induced stress. |
| Thawing and Culturing Cells |
|
| Incubation Atmosphere | 37°C, 5% CO2, humidified atmosphere. |
| Shipping Conditions | Cryopreserved cell lines are shipped on dry ice in validated, insulated packaging with sufficient refrigerant to maintain approximately −78 °C throughout transit. On receipt, inspect the container immediately and transfer vials without delay to appropriate storage. |
| Storage Conditions | For long-term preservation, place vials in vapor-phase liquid nitrogen at about −150 to −196 °C. Storage at −80 °C is acceptable only as a short interim step before transfer to liquid nitrogen. |
Quality Control & Molecular Analysis
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
